Abstract
The thermomechanial properties of polymeric nanocomposites are related to the quality of the adhesion between matrix and nanoparticle. Since the adhesion is related to the nature of the materials and the surface available for chemical, electrostatic and mechanical interactions among these materials, weak bonding forces between alumina (inorganic) and polymer matrices (organic) were expected. Furthermore, using nanoparticles with greater diameters means that the specific surface area reduction will have an adverse impact on the adhesive process. For epoxy matrices reinforced with alumina nanoparticles, different volume fractions and sizes were observed by differential scanning calorimetry (DSC): a relation between the glass transition temperature (Tg) and the nanoparticle size. This observation was tested by dynamic mechanical analysis (DMA) and the cross-link density was calculated. In addition, the thermal stability enhanced by alumina addition to organic resins and the quality of the adhesion was observed by thermogravimetric analysis (TGA).
1. Introduction
Nanocomposite materials can be described as materials consisting of nanoparticles or nanopores embedded in a solid matrix, polymers, for example. They are considered suitable for various applications as their properties can be tuned by varying the particle material, shape, size and concentration. A growing interest in nano-sized reinforced polymers for high technological applications, such as coatings, electronic devices and adhesives, was also observed in automotive and aerospace industries [1-3].
Polymeric nanocomposites composed of an organic matrix and inorganic nanoparticles makes a useful union of the polymer's functionalities, such as low weight and malleability, with the good mechanical, thermal and electrical properties of the nanofillers [4]. In this way, nanocomposites have the advantage of adapting to the properties of the polymer according to the application of the correct nanoparticle.
The possibilities using nanoparticles to improve mechanical or thermal properties, and developing a new material, have found applications in both academia and industry. The thermal stabilization of polymer nanocomposites is considered an important factor and its study provides the required information about the period of applicability for such nanocomposites [5].
An adverse effect of the inorganic nanofillers is the strong possibility of agglomeration. Therefore, the use of surface modifiers, such as silane for example, to improve the dispersion stability of the nanoparticle and develop the interfacial interactions between inorganic nanofiller and organic matrix is well known [4].
Epoxy networks (ER) are considered the most useful thermoset polymer. It can be applied as an adhesive in the electronics industry, as a fastener component in structural joints [6]. To produce a composite material, epoxy polymer has commonly been modified by the addition of inorganic fillers. As with the combination of any materials with different chemical natures, problems related to particle adhesion have been reported [7]. Debonding and particle agglomeration are major themes in the fabrication of polymeric nanocomposites, causing the most problems in nanocomposite research [8].
The quantitative study of solid state transformation in various kinds of materials by means of differential scanning calorimetry (DSC) and dynamic mechanical analysis (DMA) has been widely discussed [9-13]. While DSC elucidates the kinetics of polymer degradation and decomposition steps [13], DMA provides a sensitive testing system for the rapid determination of thermo-mechanical properties, such as a function of frequency, temperature or time [9].
A property that has been widely used to predict a change in the mechanical properties of these materials as a function of temperature is the glass transition temperature (Tg), a second-order pseudo transition that constitutes a highly interesting parameter of amorphous and semi-crystalline materials [14]. The basis of the Tg is the onset of coordinated molecular motion in the polymer chain. In the region of Tg, the polymer softens, the modulus drops three orders of magnitude and the polymer becomes rubbery [15]. In essence, if the addition of a particle to an amorphous polymer leads to a change in the Tg, the resultant effect on the composite's properties would be considered a “nano-effect”. In addition, the temperature-dependent elastic modulus of a composite material is an important material parameter in structural analysis and design, especially for stiffness-based design. Elastic modulus represents the material stiffness and, in polymer-based composites, the temperature will significantly influence the materials' rigidity.
Epoxies are thermosetting polymers that exhibit a high glass transition temperature when compared with other polymers that can be used as a nanocomposite matrix [16]. In addition, nanoparticles presented better interfacial interactions with the matrix when compared with conventional microparticles [17]. Many studies have investigated the influence of nano-sized fillers on solid polymeric materials [18-26]. The purpose of this study is to investigate experimentally how the effective thermal-physical properties are influenced by the addition of alumina (Al2O3) nanoparticles to an epoxy matrix, in multiple volume fractions and particle sizes.
2. Experimental
2.1 Nanocomposites' Fabrication
An epoxy polymer matrix and metal oxide Al2O3 nanoparticles as fillers were used to compose the nanocomposite materials for this study.
The employed polymers were RR515 (provided by SILAEX®), and an epoxy resin (ER) based on diglycidylether of bisphenol A. This resin was polymerized by the addition of an aliphatic amine hardener in a portion of 25 phr by weight. Its properties, according to the manufacturer, are presented in Table 1.
Properties of the thermosetting polymer
The nanoparticles employed as filler were spherical alpha aluminium nanoparticles with mean diameters of 35nm, 150nm and 200nm, provided by NanoAmor®. The nanoparticles' properties, according to the manufacturer, are summarized in Table 2.
Al2O3 nanoparticles' properties

The samples were manufactured by adding different quantities of nanoparticles — from 0% to 10% of the total mixture's volume — to the liquid resin. The nanoparticles' volume fractions were calculated based on the true densities data provided by the manufacturers and the rule of mixtures. The nanoparticles were previously dried at 120ºC for 24 hours before being added to the liquid resin. Homogenization by planetary ball milling was performed for 1h at 200rpm. After mixing, the hardener was added and the resulting blends were manually homogenized and poured into the mould, which was composed of a metal frame. The specimens were cured at room temperature for 24 hours. The demoulding of the samples occurred after the first 24 hours, and the samples underwent a post-cure process at room temperature for seven days. After this period, the samples were prepared for DSC and DMA analyses.
2.2 Experimental Setup
The glass transition temperature of the manufactured nanocomposites was measured by DSC (NETZSCH®, DSC F3 MAIA) at temperatures ranging from 30–150°C, at a heating rate of 10ºC/min in a nitrogen atmosphere according to ASTM D3418 [27]. The temperature range was chosen after a previous scan of up to 500°C to observe the thermal stability and decomposition for each nanocomposite.
The glass transition temperature as defined by the endothermic change in the DSC trace indicates a significant change of viscosity, marking an alteration from a glassy solid phase to a super-cooled liquid state. In DSC measurements, Tg is recognized from the baseline change in the DSC signal.
A dynamic mechanical analyser (NETZSCH® DMA 242D) was used for measuring the temperature dependent elastic modulus of all the studied formulations of the nanocomposites. Tests were conducted with three-point bending mode and the support span was 40 mm, according to ASTM D7028 [28]. Temperature scanning from low to high was performed with a heating rate of 10°C/min, from 30°C to 180°C at an oscillation frequency of 1 Hz. The oscillation amplitude of displacement was kept at 40mm for most specimens, unless otherwise specified.
One of the main reasons to modify a polymer with oxide nanoparticles is to enhance the material's thermal and mechanical properties. DMA was frequently used in nanocomposites' characterizations since it allows the measurement of two different moduli of the nanocomposites, a storage modulus (E′) and a loss modulus (E'′). The first is related to the ability of the material to return or store mechanical energy and the second is related to the ability of the material to dissipate energy as a function of temperature.
2.3 Statistic Analysis
The glass transition temperature, storage modulus and the thermal stabilization temperatures (Tstb) have been established for the following composites: pure ER, ER/2.5% Al2O3, ER/5.0% Al2O3, ER/7.5% Al2O3 and ER/10.0% Al2O3 for each particle size, resulting in 12 different nanocomposites and 45 tests (DSC, DMA and TGA).
The results were analysed using the software Portable Statistica 8 (StatSoft Inc./DELL) which uses the analysis of variance (ANOVA) of the data with the Tg, E′ and for the nanocomposite materials was done with the objective of analysing the influence of the matrix/particle interactions, the volume fraction of the nanoparticle and the mean diameter of the nanoparticle.
3. Results and Discussion
3.1 Thermomechanical Analyses
As observed by Tanaka et al. [16], the organic-inorganic bonding force seems to be relatively weak in epoxy/alumina nanocomposites. Some authors, such as Zhang and Singh [17], are studying the addition of silane as a treatment to improve the particle (inorganic)/matrix (organic) adhesion. The glass transition data for nanocomposites with epoxy resins shown in Table 4 are according to what have apparently been obtained recently for nanocomposites without silane treatment.
Al2O3/ER Nanocomposites' DSC results

DMA results

The larger the particle diameter used, the smaller the increase in the Tg is expected, since the adhesion forces between the nanoparticles and the matrix are directly proportional to the specific surface area.
As it is seen in Table 3, the Tg of the nanocomposites varied with the particles' diameters. The increase in Tg is related to the decreased mobility chain of the matrix, and for these nanocomposites, better mechanical and thermal properties are expected, as a result of the Van der Waals forces and electrostact forces between the epoxy matrix and alumina nanoparticles.
In general, the addition of alumina in ER polymers promotes an increase in the glass transition temperature. Only the sample nano-Al2O3/ER 5% v/v did not present a result consistent with the literature and the responses of the other samples. This is probably because during demoulding and milling for the manufacture of DSC samples, the presence of bubbles inside the nanocomposite was observed. Such bubbles can be regarded as points of concentration of alumina which interfere adversely in DSC analysis.
Moreover, Table 3 showed no variation in the glass transition temperature on the addition of different volume fractions of nanofiller. The higher Tg for nano-Al2O3 (A)/ER 2.5% v/v can be attributed to overall bonding forces (Van der Waals and possible electrostatic interactions) increasing [29].
The effect of nanofillers in epoxy and polyester resins on dynamic mechanical properties, the glass transition temperature of the nanocomposite and each neat matrix were observed using a DMA analyser.
As observed by Zabihi et al. [30], a strong compositional effect on Tg, as measured by DMA, was observed as a result of the nanoparticles' addition in epoxy polymer, resulting in higher Tg values than those obtained in DSC analyses. The same behaviour was observed for the polyester matrix.
The effect of Al2O3 nanoparticles on the viscoelastic properties of cross-linked epoxy nanocomposites has been investigated by DMA, estimating the effect of each nanoparticle in each matrix by calculating the cross-link density using the equation below [30]:

where υe is the cross-link density, Er is equal to the rubber modulus at Tr, R is the universal gas constant and Tr = Tg + 30.
The result of alumina nanoparticles' effect on the viscoelastic properties of cross-linked epoxy composites was observed by DMA, as can be seen in Fig.1.

Storage modulus versus temperature curves for Al2O3 A (a), Al2O3 B (b) and Al2O3 C (c) nanocomposites
It was expected that the chain motion would change due to the interactions in the interface area around the nanoparticle. As can be noticed in Fig. 1, which shows the effect of the nanoparticle additions in volume fractions for Al2O3 A (a), Al2O3 B (b) and Al2O3 C (c), the nanofiller has an adverse effect on storage modulus and this effect appears to be different according to the particle size. The amplitude of storage modulus in the rubbery plateau's region does not vary significantly on the addition of nanoparticles. This unexpected result can be explained by reference to the weak adhesion forces between the nanofiller and the polymer matrix, since inorganic-organic interactions are very weak without the use of some inorganic surface modifier, as silane.
Table 4 shows the results obtained using DMA for pure epoxy and its alumina systems. The results show that the nanoparticle with a lower SSA has the best results of υe and that for all nanocomposites the concentration of nanofiller has a quadratic effect on cross-link density.
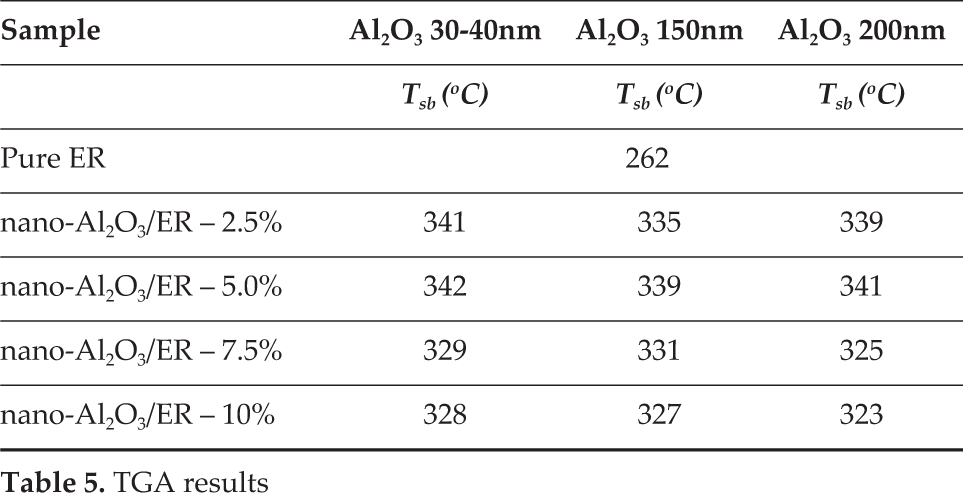
To conclude, the thermogravimetry showed that the alumina nanoparticle addition increased the onset degradation temperature in approximately 70°C. Although alumina and epoxy resin presented weak bonding forces, the interaction between polymer chains and metal oxide nanoparticles also contributed to the high value of reside for the nanocomposites, as can be seen in Table 5.
TGA results
3.2 Statistical Analysis
The glass transition temperature, storage modulus and thermal stabilization temperatures have been established for each nanocomposite, resulting in 28 different nanocomposites and 240 tests (DSC, DMA and TGA).
The statistical analyses were carried out to analyse the influence of the matrix/particle interactions, volume fraction of the nanoparticle and mean diameter of the nanoparticle on Tg, E′ and Tsb. The mean values of each parameter and each sample were compared by applying one-way analysis of variance ANOVA with a 0.05 significance level.
The ANOVA for Tg measured by DSC confirmed the quality of the results in normality distribution and homogeneity of variance in the Shapiro-Wilks and Cochran-Bartlet tests, respectively. For this variable, a strong influence of the nanoparticle's size was observed, while the effect of the filler's volume fraction was the least important of the mean effects (Fig.2)

Statistical analysis of DSC results
The second dependent variable analysed by ANOVA was the storage modulus of the nanocomposites manufactured. As demonstrated in the section above, the adverse effect of the addition of filler was observed as a mean effect. However, in Figure 3, statistic analyses showed that the nanoparticle volume fraction had no significant effect.
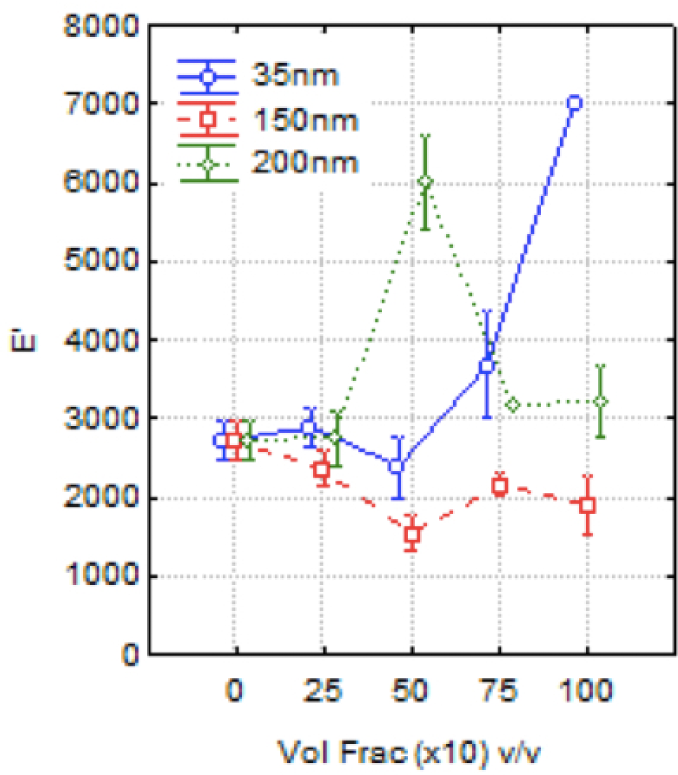
Statistical analysis of DMA-E′ results
Finally, for the stabilization temperature measured by TGA, Tstb statistical analysis showed no significant effect of the size of the nanoparticle, even for the volume fraction used to produce the nanocomposite, as displayed in Figure 4.

Statistical analysis of TGA results
4. Conclusions
The influence on thermo-mechanical properties by alumina nanoparticles' addition in epoxy resin was observed using DSC and DMA analyses. The results indicate that different interactions exist for different matrix/nanoparticle size combinations. The influence of the nanoparticle size on thermo-physical properties of alumina/epoxy was demonstrated as the mean factor in this kind of composite. The cross-link density calculated by DMA data has a quadratic behaviour with the nanofiller volume fraction increase. The DSC analyses showed different impacts on glass transition temperature according to the nanoparticle's size, indicating that the SSA is an important factor in organic-inorganic interactions and the adhesion forces between nanoparticles and the matrix. Finally, the nanofillers improved the thermal resistance of the neat epoxy.
Footnotes
5. Acknowledgements
The financial support of Rio de Janeiro State Funding, FAPERJ, the Research and Teaching National Council, CNPq, the Coordination for the Improvement of Higher Level Personnel, CAPES and CEFET/RJ is acknowledged with gratitude.
