Abstract
Nanocrystalline zirconia powders stabilized with varying concentration of yttria (YSZ) were synthesized following urea solution combustion route. Thermogravimetric analysis combined with differential scanning calorimetry was carried out for the intermediate gel for analyzing reaction kinetics. The final products were characterized by X-ray diffractometry, transmission electron microscopy and specific surface area measurements. Sintering behavior of theses powders were studied. The powder quality is poor in respect of surface area and sintered density.
1. Introduction
Yttria-stabilized zirconia (YSZ) having excellent chemical resistance, refractory character, oxygen ionic conductivity and polymorphous nature finds many structural and electronic applications such as gas sensors, electrolyte and anode in solid oxide fuel cell (SOFC) [1, 2]. For these applications several synthetic strategies have been developed such as hydrothermal, solvothermal, aqueous synthesis, organic precursor route, homogeneous precipitation, coprecipitation, spray drying, sol-gel, plasma spray, flame spray pyrolyosis, molecular decomposition and spray-ICP for preparation of YSZ nano- powders [3]. Combustion synthesis is another promising route for the synthesis of nanopowder. This synthesis route is simple and faster method. This process starts at low temperature, with the help of an external heating source, followed by subsequent exothermic reaction between the oxidiser and fuel; this exothermic the reaction provides necessary heat to further carry out the reaction in the forward direction to produce nanocrystalline powders as the final product. Several organic fuels such as urea, glycine, oxalyl dihydrazide, sucrose, glycine-nitrate, urea-formaldehyde have been tried to synthesize nano-ceramic powders [4–8].
In this work, nanocrystalline cubic zirconia powders stabilized with varying concentration of yittria have been synthesized by solution combustion route using urea as fuel. The reaction process has been established using thermogravimetric analysis combined with differential scanning calorimetry (DSC-TGA). Phase formation of the powders has been verified by x-ray diffraction (XRD) technique; lattice parameter was calculated using precise lattice parameter method. The final products have also been characterized by transmission electron microscopy (TEM) and specific surface area measurement (BET method). Sintering behavior of the powders was studied. The aim of this paper is to analyze the effects of concentration of yttria on the properties of YSZs powders.
2. Experimental
Zirconium oxychloride octahydrate, ZrOCl2.8H2O (99%, Indian Rare Earth Ltd., Mumbai, India], Yttria, Y2O3 (99.9%, Indian Rare Earth Ltd.), urea (Thomas baker (Chemicals) Ltd., Mumbai, India) and nitric acid (Merck, Mumbai, India) used were of analytical reagent grade. Flow chart for the process has been shown in Fig.1. 8–12 mole% yttria stabilized zirconia (8 – 12YSZ) powders were synthesized with the step size of 2. Zirconium nitrate and yttrium nitrate were synthesized following some standard procedures mentioned in several literatures [9, 10].

Process Flow Chart
Both the solutions were mixed stoichiometrically. Fuel was added to this composite nitrate solution in a proportion of two moles per mole of metal ion. The fuel-nitrate solution was kept in the muffle furnace at 500 °C to carry out combustion inside the furnace [11].
Differential scanning calorimetry (DSC) along with thermogravimetric analysis (TGA) (model: Setsys 16, SETARAM, France) of the dried gel was carried out at a heating rate of 10 °C /minute in air atmosphere to know the combustion initiation temperature as well as combustion behavior and the weight loss regime.
The phase formation of the powders were studied by X-ray diffractometry (XRD) (Xpert MPD, PANalytical B.V., Almelo, the Netherlands) using CuKα radiation (1.5406Å) in the angular region of 2θ = 20° - 80°. The observed XRD data was processed to obtain the actual peak positions to determine lattice parameter ‘

Where Δ2θ is the deviation from the observed angle, 2θobs, the three constants, a1, a2 and a3 were calculated from the data obtained using silicon standard sample. The crystallite size from the XRD data was calculated using Scherrer formula [12]:

where t is the crystallite size in nm, β is broadening of diffraction line at full width at half maxima (FWHM), θ was obtained from corrected peak position, 2θ and λ is the wavelength of CuKα radiation. Instrumental broadening was eliminated using standard procedure [12]. Transmission electron microscopy (TEM) (Philips USA) was carried out for all the YSZ powders to confirm crystallite size of the powders. Specific surface area analysis (Sorptomatic 1990, Thermo Fennigan, Radano (MI), Italy) of the calcined powders was carried out by Brunauer-Emmett-Teller (BET) method.
Sintering behaviour of the powders were studied varying temperatures for 30 minute soaking time.
3. Results and discussion
TGA curve shows a progressive weight loss from around 70 °C and it became a bit faster up to 105 °C, which corresponds to endothermic effect at around 100 °C in the DSC curve (Fig.2). This weight loss may be due to dehydration of nitrates and partial decomposition of urea. Sudden charring of urea occurs, as evidenced by a strong exothermic peak at around 160 °C in the DSC curve [13]. This reaction leads to the faster weight loss, as evidenced in TGA curve. The exothermic peak appearing at 275 °C in DSC curve is due to the combustion of urea by the nitrates. The heat emission keeps on at higher temperature, as indicated by the exothermal broad peak above 270 °C. This exothermal broad peak indicates that urea combustion occurs in a complex way [13]. The strong exothermal combustion reaction raises the temperature locally, thus promoting the formation of YSZ face centered cubic (FCC) phase during the combustion.

TGA-DSC Plot of Gel
The XRD patterns are shown in Fig.3. The plots reveal the powder to be a single phase one with FCC structure in each case. The lattice parameter of the FCC unit cell calculated by least squares method using XRD data was found to be 5.1175, 5.1198 and 5.1219

XRD Plots of YSZ Powders
The TEM micrograph of 8 mole% yttria stabilized zirconia powder is shown in Fig. 4. The TEM images indicate that the particles are of uniform size with average size approximately 17 nm. The results confirmed the value of crystallite size determined from xrd data.

A Representative TEM Image of 8YSZ Powder
Sintering kinetics of YSZ ceramics was measured as density vs. temperature for 30 minute soaking. It was observed that density increased with sintering temperature up to 1550 °C and thereafter showed a decreasing trend. Maximum sintered density of 92.68 % of x-ray density was achieved at 1550 °C.
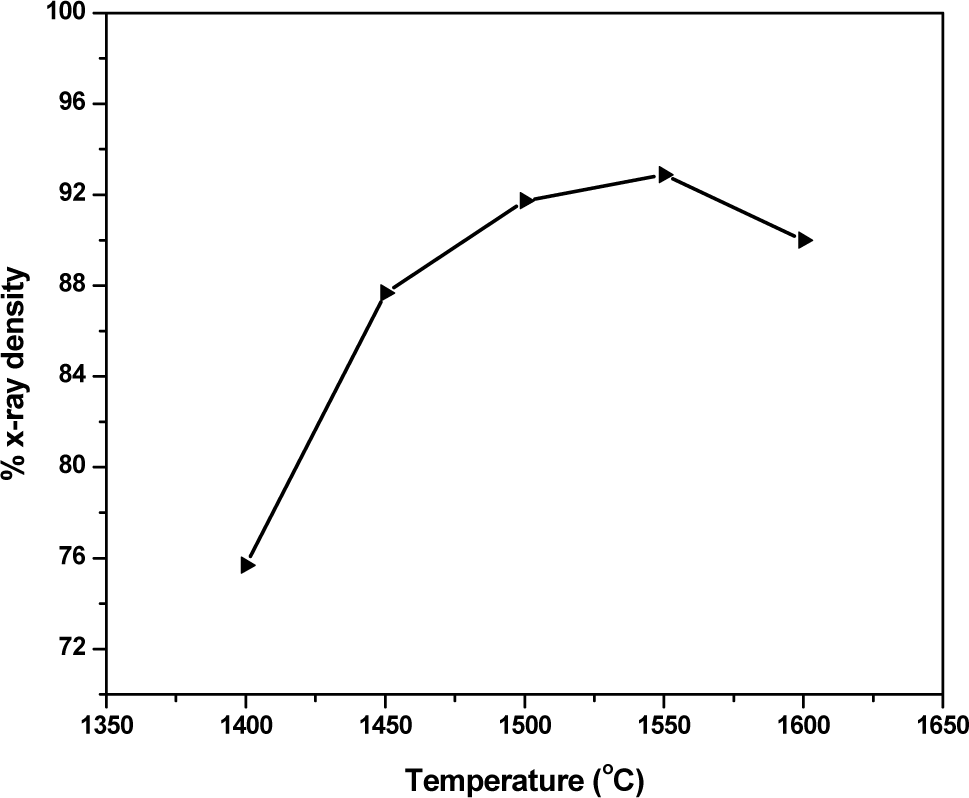
Sintered Density of 10YSZ Pellet

Sintered pellet of 10YSZ
4. Conclusion
Face centered cubic nanocrystalline YSZ powder can be prepared by urea solution combustion synthesis route. The DSC study showed that the combustion occurred in a complex manner which resulted in low surface area. The average crystallite size of the powder was 17 nm. Lattice parameter was observed to increase with yttria concentration from 5.1175 to 5.1219 nm for 8 to 12 mol% yttria in zirconia. Study of sintering showed that maximum of 92.68% x-ray density can be achieved at 1550°C for 30 minutes.
Footnotes
5. Acknowledgments
The author is grateful to Dr. J.N. Das, Director, NMRL and Dr. S.C. Sharma, Head of Ceramics Division and the colleagues of Ceramics Division of NMRL for their contribution in this work.
