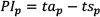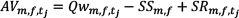Abstract
Materials management is an important issue for healthcare systems because it influences clinical and financial outcomes. Before selecting, adapting and implementing leading or optimized practices, a good understanding of processes and activities has to be developed.
In real applications, the information flows and business strategies involved are different from hospital to hospital, depending on context, culture and available resources; it is therefore difficult to find a comprehensive and exhaustive description of processes, even more so a clear formalization of them.
The objective of this paper is twofold. First, it proposes an integrated and detailed analysis and description model for hospital materials management data and tasks, which is able to tackle information from patient requirements to usage, from replenishment requests to supplying and handling activities. The model takes account of medical risk reduction, traceability and streamlined processes perspectives. Second, the paper translates this information into a business process model and mathematical formalization.
The study provides a useful guide to the various relevant technology-related, management and business issues, laying the foundations of an efficient reengineering of the supply chain to reduce healthcare costs and improve the quality of care.
Keywords
1. Introduction
Progressive reduction in public resources – and the subsequent need to restore budgets – means governments are responsible for finding solutions to achieve more operational efficiency in hospital processes. Drug expenditure, in particular, is a relevant factor in the profit and loss accounts of healthcare systems ([1, 2]); hospital pharmacy management is called upon to adopt policies to reduce drug inventory costs and maximize the cost-effective use of personnel and resources [3–5].
Awareness of logistics is becoming more widespread and many initiatives and studies dealing with supply chain integration have been undertaken (for example, supply outsourcing strategies [6, 7]). However, the internal supply chain (vs. external, i.e., beyond the boundaries of an organization) “remains the sore spot or weak link” [8] in process integration and optimization. This lack of systemic approach to internal supply chain management is reflected in huge costs in materials management and low service quality delivered to patients [9].
Hospital materials management (HMM) also involves the clinical sphere of healthcare service performance. Clinical errors in drug prescription and administration, for example, are always possible, depending on human factors and procedural issues.
Regarding resource use optimization, the difficulties of transferring manufacturing best practice directly to the hospital environment are evident [10], although the first sector of intervention is inventory management (see, for example, [8]). It is conceivable to suppose that the main cause of inefficiency is the existence of hidden stocks to avoid stock-outs [10], which would be more politics- and experience- rather than data-driven [6].
It is clear that a significant percentage of a pharmacist's time is consumed by order entry, verification, clarification, and follow-up activities [11]; the same applies for nurses with prescription transcriptions, stock level control and administration.
In order to achieve a comprehensive image of the HMM process and potential ways of exploiting IT to enable an efficient reengineering of this supply chain, reducing healthcare costs without affecting the quality of care [1], it is fundamental to consider the previously presented logistical and clinical perspectives as both sides of the same coin.
In the literature, there are many collateral references on how materials management works; in real cases analysed, information systems are usually built for fragmented applications, and much information is lost or is not recorded when it flows through processes. This implies losses in traceability and increases in clinical risk, while inventory management techniques and logistics are hardly performed, causing high inventory costs.
The objective of this paper is twofold. First, on the basis of a literature review, analysis of real cases and international guidelines, the paper fills a research gap, providing an integrated and detailed analysis and description of hospital materials management data and tasks (able to tackle information from patient requirements to usage, from replenishment requests to supplying and handling activities) in relation to medical risk reduction, traceability and streamlined processes perspectives. Secondly, the paper translates this knowledge into a business process model and a mathematical formalization, showing the integrated information and physical flows in order to trace and share data among actors with the aim of reducing clinical risk and time-consuming tasks while enabling requirement programmability and, more in general, knowledge management.
The paper is structured as follows: after the materials under examination are defined, the materials, processes and actors are described in section 2. Details about process modelling and formalization are given in section 3, and in section 4 the performance results of a case study are presented. Finally, conclusions and suggestions for possible further research are presented.
2. HMM elements: materials, processes and actors
The first step in the hospital pharmacy “micro-world” reengineering process is to identify the behaviour of this system, to establish for example what to manage in terms of materials (par. 2.1), processes (par 2.2) and actors (par. 2.3), taking into account information and legal constraints.
2.1 Materials and places
Management of materials in healthcare involves two kinds of item clusters: drugs (or medicines) and medical devices, subjected to different regulations harmonized by countries according to international guidelines. The properties of medicines in a hospital information system may be mandatory or optional depending on the contextual workflow [12]. A fundamental “identifier” is the ATC (Anatomic – Therapeutic – Chemical) classification, internationally accepted and maintained by the World Health Organization. In addition to commercial drugs, drug administrations can also refer to galenics, such as personalized medicines prepared as a “mixture” of commercialized products at the bedside, in hospital pharmacy or in another defined medical unit. In parallel, medical devices, such as surgical kit and apparatus, can be managed as single or grouped items by pharmacists. The item list (in other words, the set of medicines or medical devices that can be administered/dispensed to or implanted in patients in a healthcare system) changes from hospital to hospital, depending not only on the healthcare services managed, but also on the physician's expertise and preferences, and following pharmaco-economics principles [13].
Regarding places of materials management and dispensing, it is possible to distinguish between clinical and managerial perspectives. From a clinical point of view, the usefulness of having centralized patient-oriented pharmacy services to deliver professional services to the patient has been recognized [14, 15]. From a managerial perspective, many authors state the possibility of reducing logistics costs using satellite pharmacy systems [16]; numerous studies have suggested the benefits of having central inventory control rather than allowing departments or regions to deal with suppliers individually (O'Hagan 1995, cited in [17]), particularly in terms of drug waste reduction [16]. In addition to a centralized point of distribution, however, it is necessary to have a decentralized materials management warehouse close to the point of delivery [18] to allow nurses to quickly reach stocks in the proximity of places (beds) where services (administrations) are delivered. The same holds for medical devices, particularly in the case of vendor-managed inventories for surgical interventions. For these reasons, the inventory system is typically characterized by centralized and decentralized stores [10] (hub and spoke model), with two types of warehouses: medical unit and hospital pharmacy. The first has to deliver materials to the patients of the facility; the second is in charge of managing items for inside (to the medical unit) and outside dispensing (to patients).
2.2 Processes
Businesses should not be analysed in terms of the functions to which they can be deconstructed or in terms of the output they produce, but taking into account the key processes they perform [19]. The hospital medication workflow is triggered by patients needing drugs and medical devices. In this perspective, the Technical Framework was developed for the pharmacy domain by the initiative Integrating the Healthcare Enterprise (IHE) [12], in order to stimulate the integration of healthcare information systems operating with different standards (such as DICOM, HL7, etc.). In the proposed pharmacy interoperability model [13], the care path (clinical perspective) described is orthogonally combined with the supply path (logistical perspective) in the phase of distribution but, as explicitly stated, the supply chain of ordering/delivering medication and stock management are out of the IHE's scope.
In this paper we adopt and refine the IHE pharmacy interoperability model as an “information track” to classify and describe all the elements depending on logistics decisions in the hospital medication workflow. We readapt that model to define in more detail different care paths, distinguishing between the types of medicine or medical devices employed; moreover, we explore the management area, formalizing the involved processes.
Analysing hospital cases, the literature review and international guidelines, and considering the clinical/logistical perspective, we define hospital materials management as composed of several main macro-processes, which are fully described, together with their sub-processes (illustrated in Figure 1), in the next sections:
Process Framework of Materials Management in a Hospital (the notation used is explained in section 3) Patient Management, Medical Unit inventory Management, Centralized inventory Management.
2.3 Actors
According to the macro-processes classification, the actors participating in the HMM can be divided into three categories:
Medical Unit actors: including the patient, physician (who delivers the care), nurse (who dispenses the care) and nurse manger (who supervisions the dispense of care); Pharmacy Unit actors: usually composed of hospital pharmacists, storekeepers and transporters; Other organizational functions also take part in the process: the Medical Director, the Superintendence and Treasurer's Office, and the Accounting Office.
3. HMM process model
It is accepted worldwide that business process management analysis plays “a major role in the perception and understanding of business processes” [19], spanning from organizational/managerial issues to information systems and even social problems [20]. Trkman [20] states that understanding and analysing a business process “helps to recognize the sources of problems and ensure that they are not repeated in the new process”, thus providing a measure of value for the proposed changes.
Many authors have provided frameworks for presenting and classifying different business process modelling techniques. Using the classification proposed by Vergidis et al. [19], which applies to Diagrammatic, Formal/Mathematical and Business Process Language models, we use one technique from the last area. The technique permits “tackling the complexity of the formal models [while] retaining their consistency and potential for further analysis”, the Business Process Modelling Language (BPML), being the most distinctive.
BPML, defined by the Business Process Modelling Initiative, is also an XML-based language that encodes the flow of a business process in an executable form. BPML is accompanied by Business Process Modelling Notation (BPMN), a graphical flowchart language that is able to represent a business process in an intuitive visual form. Each BPML process has a name, a set of activities, a handler and also supports sub-processes.
In what follows, the three processes previously mentioned (a, b, c) are described and represented by the BPMN. Their interdependences are shown in Figure 1; as regards the drugs to be administered, they are examined in depth in Figure 2 and Figure 4.
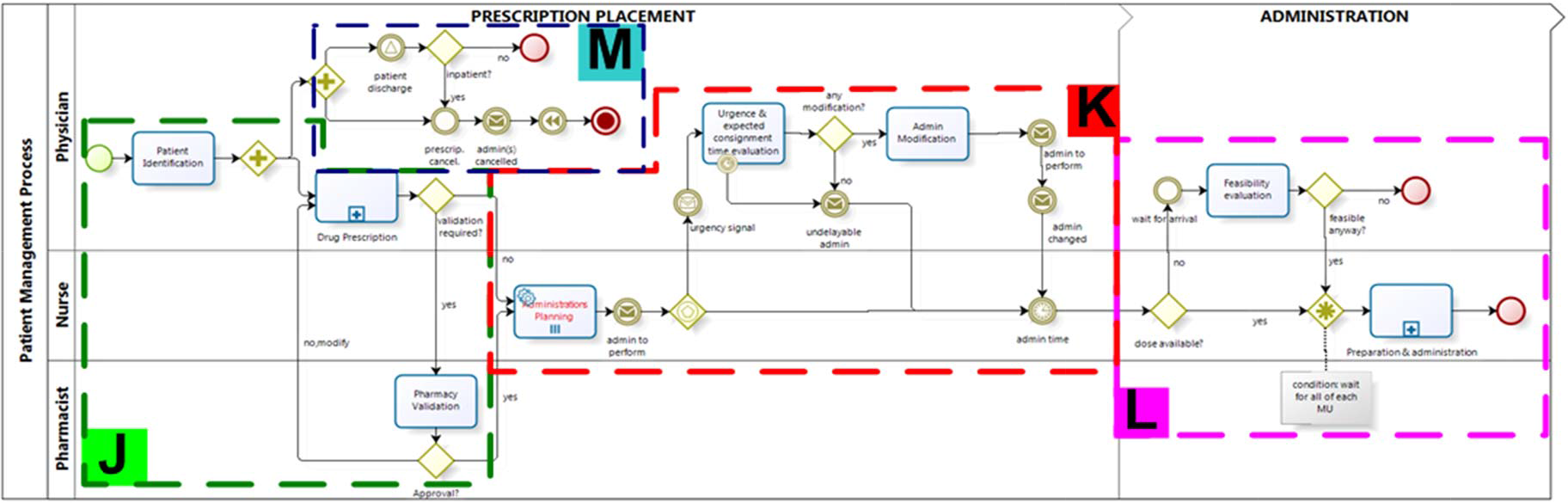
Patient Management Process frames (J, K, M and L)
3.1 Patient Management process (a)
The first macro-process is the patient management process, since the need for care (and then the hospital medication workflow) triggers materials requirements.
When it comes to a patient's admission (to an emergency or outpatient department), a physician states a diagnostic hypothesis, attributing (with or without confirmation) one or more diseases to the patient.
Depending on resource availability (beds, laboratories capacity, etc.) and urgency, (s)he also assigns a diagnostic and/or therapeutic pathway to be followed in the hospital or at home.
Hospitalization in a medical unit can be immediate or delayed according to a queue list (for Inpatient access, Day Hospital, Day Surgery, etc.); hospitalization or not, a patient's anamnesis and a previous and current therapy record are always required to know whether the patient is compatible with the respective pathway.
The patient management process (a), in which physician, nurse and pharmacist take part, is composed of three subprocesses, as shown in Figure 2:
Prescription:
Drug prescription or surgical intervention plan,
Exam prescription;
Pharmacy prescription validation;
Materials delivery to patient:
Preparation, administration or implant (inside dispense),
Dispense to patient (outside dispense),
Examination accomplishment,
Low cost and genericgoods usage.
3.1.1 Drugs Prescription or surgical intervention plan (a.1.1)
The process entity that flows throughout the system is the drug prescription. According to clinical risk reduction, in the J frame of Figure 2, patient identification is carried out by the physician before the prescription, who is also provided with diagnosis, anamnesis and double reconciliation (processes (a.1) and (a.2)).
In addition to pharmaceutical validations (if the prescription has to be checked) a drug prescription is also the input of nurses' instructions for administering the drug to the hospitalized patient or, in the case of outside dispense, for patient home care. Variations in the content of prescriptions can occur, depending on routines and country regulation, responsibilities and standards; however, on the basis of governmental laws, IHE statements [12] and interviews, the medical prescription data set can be said to be composed, among other elements, by actor ID (Patient and Physician), reason for prescribing, active substance and brand, dosage, frequency of intake, and quantity. Alerts about prescription restrictions and potential Intolerance, Contra-indication and Allergies (ICA) should always be consulted.
The active substance(s), as stated above, is usually a key element because it can permit evaluations about pharmaco-economics and availability (for example, to distribute the cheapest brand name stored in satellite or central pharmacies or to prescribe what is stored in the local warehouse), but the clinical consideration can imply a more rigid selection.
In the case of surgical intervention, some materials have to be explicitly prescribed (for example, an orthopaedic prosthesis and its size or surgical kit).
Considering the data related to inventory management activities, the output of this phase is one or a list of records of prescriptions (p) for each patient, formalized as follows.
The time base unit of administration schedule and evaluation of requirements is the time buckets (tj), with T = {t1, …, tj, …, tj] being a given observation time (T).
In particular, having defined:
f εF, drug with the attributes:
Nf: number of units contained in a package of f LTf: supplier lead time, time between the ordering and the delivery of a supply order
m εM, Medical Unit belonging to a hospital with the attribute:
TTm: time needed to transport orders from the pharmacy to the medical unit m.
The classical prescription record for a patient is composed of:
p εP, hospital prescription for a single drug f, with: tsp: time bucket at which the physician states the prescription p
tap: scheduled administration time bucket for the prescription p DRp: drug prescribed f, chosen from F Qp: administration quantity MUp: medical unit where the patient is hospitalized m, belonging to M.
3.1.2 Exams prescription (a.1.2)
The physician can prescribe some diagnostic examinations to patients, such as laboratory exams (e.g., blood, RX) or other activities that require materials.
3.1.3 Pharmacy Prescription Validation (a.2)
After a prescription, prescription information may be made available to pharmacists for a pharmacy validation. It can be advisable but not mandatory, so that many organizations tend to jump this step. This implies no double reviews and, hence, no possibility of medical error reduction [13].
A detected problem can be a supply issue (suspended medication, out-of-stock, etc.), a legal issue (medication recalled or not allowed under certain conditions), or a medical issue (redundancy, interaction, contra-indication, ICA, etc.).
3.1.4 Materials delivery to patient (a.3)
In the K frame of Figure 2, the administrations dependent by the prescriptions placed are evaluated in terms of dose availability at the administration time tap and possibly activate the (b) process.
The possibility of managing urgent deliveries caused by unavailability of administration materials is also presented. Delivery urgency means lead time (TTm or LTf) compression and, consequently, higher delivery costs, not excluding being behind schedule. However, deliveries may be easily evaluated by the physician before being activated. Indeed, a time can be given to the physician to confirm the feasibility of the administration behind the schedule on the basis of the delivery scheduled time.
In the M frame of Figure 2, the cancellation of a prescription or the discharge of an inpatient, are transmitted to process (b).
While Figure 2 deals with drug administration to a hospitalized patient, four events can imply materials delivery to patients:
Preparation, administration or implant (inside dispense), Dispense to patient (outside dispense), Examination accomplishment, Low cost and genericgoods usage.
Preparation, administration or implant (a.3.1)
This refers to the delivery of materials inside the hospital at the tap occurrence, depending on a prescription and carried out by a nurse.
Many hospitals do not have an information tool or do not have an information system at all, so nurses are in charge of transcribing prescriptions on provided diaries, checking them to know when to administer. This implies risk of errors in transcription and administration execution (right time, person, etc.).
Preparation is the phase in which nurses take drugs from the stock and prepare them to be administered to patients.
Galenics are a special case because the preparation, depending on the organization, may involve pharmacists and requires an appropriate recording of all actions and items used (information about drug ID, lot number, quantity used, etc.) for traceability reasons. For example, while an intravenous injection of cortisone diluted in 0.9% sodium chloride solution can be prepared at a patient's bed, oncological galenics are usually made in a dedicated medical unit and later transported. Labelling with patient and galenic ID is the right way to reduce clinical risk while ensuring traceability. According to IHE suggestions [12], the assignment of a medicine to a specific patient is a “dispense”; a galenics dispense can be considered as the instant in which labelling occurs.
All the medications are usually collected in a trolley, or an already-filled trolley is used, with a number of drawers containing the most frequently used drugs and other drawers for specific patients' needs (this is the case described in [21]). Moreover, some medical devices such as syringes, gloves, roller bandages, etc., have to be added to the trolley.
Updated inventory information is crucial, because previous checks should be done in order to ensure that each patient has his own medication available at tap. Together with the quantity availability (the L frame of Figure 2 provides an administration feasibility evaluation of a drug made available only after the tap), expiration date control has to be made and the package with the closest expiration date should be taken.
Administration follows. The nurse goes to the patient's bed and carries out the 7 “rights” checks (right patient, right drug, right dose, right route, right time, with the right information and documentation), administers the medication and records all the connected information, according to traceability and medical risk reduction objectives. Dispense (if done) or administration coincide with the recording of the consumption of the medicines used from medical unit virtual stock, so that the warehouse management system is always up to date.
In case of surgical intervention, medical devices, kits or apparatus have to be used. After it, information about these materials has to be recorded as indicated before for drug administered, in order to satisfy clinical, traceability and inventory management requirements.
Dispense to patient (a.3.2)
In some healthcare organizations, the hospital pharmacy can also be charged with materials dispense for home care (by outpatient departments or discharged patients). These materials almost overlapping with those used by inpatients, but instead of tap, a dispensing period bigger than a time bucket is fixed by the physician.
The outside dispense process can be likened to medical unit dispense, because the prescription is checked, material is taken from the warehouse, is recorded and given to the patient.
Exam accomplishment (a.3.3)
Differently from the low cost and genericgoods usage explained later, the consumption of some medical devices can be directly attributable to one patient (a bill of materials may exist). Inventory levels are usually updated when the examination is performed (back-flushing method).
Low cost and generic goods usage (a.3.4)
This process concerns medical devices utilization that is neither planned nor directly attributable to one patient's care. This distinction is trivial because it is always possible to record each activity performed for one patient and the requirements in terms of materials; this can be time consuming without any practical advantage, and most accountability/information systems do not elaborate on this function. For this reason, the medical devices belonging to this process are considered genericgoods for hospitals (examples are gloves, elastic bandages).
3.1.5 Traceability and requirements programmability
Materials traceability, from their admission to hospital till their consumption by a patient, is extremely important for clinical reasons (all information about medical processes should be recorded and available for checks), accountability reasons (to monitor expenditure and its attributions) but also for managerial motivations. A prescription, i.e., the step before materials delivery to the patient in the most part of cases, can be used in scheduling replenishments or orders on the basis of patient needs. Materials requirements programmability may have an enormous impact on inventory management techniques, but it has not really been developed because of the lack of managerial culture in the healthcare environment, together with the difficulties in implementing accurate and usable information systems.
Materials requirements programmability as a function of materials consumption traceability is illustrated in Figure 3.

Programmability of hospital materials requirements as a function of traceability of consumption

Centralized Inventory Management Process frames (N, O, P and Q)
In particular, events involving materials consumption are ranked from the least traceable to the most (the independent variable) as follows: low cost and genericgoods usage (by definition untraceable), materials employed for examinations (traceable if the information system provides this feature), materials dispense (not traceable when it comes to administration because it is performed at the patient's home) and materials administration/implant (traceable until the end of the process, because the usage of an identified material, with its characteristics, expiration date, batch number, etc., can be fully recorded). The other axis shows the materials requirements programmability, which goes from the possibility only to forecast the consumption of some materials in a period of time, to the highest probability that a particular consumption is going to take place in a specific moment. The diagram shows activities concerning patient care.
Given that “a plan must cover a period at least equal to the time required to accomplish it” [22], the programming interval PIp is fundamental data related to prescriptions (when a batch consignment for each medical unit is chosen, PI is the maximum number of time buckets available for medical unit replenishments or supply activities):
On the other hand, genericgoods usage is predictable with low uncertainty, as, because of the big volumes, it is easily forecasted by time series techniques. Almost the same applies for examinations demand. Outpatient dispense is in the middle of this plane because it concerns a prescription kept by the pharmacy for a given period while the tap is not known.
3.2 Medical Unit Inventory Management process (b)
The second identified macro-process deals with the materials management at the medical unit level. The process starts when an administration is planned. This passage is particularly important because, without defining a provisioning politics, it is able to introduce the real requirements data in the definition of an order, allowing the medical unit (and hospital pharmacy, as a consequence) to manage its stocks facing patient requirements when they emerge.
The actors involved in this process are the nurse manager and the pharmacist, and the two main activities are:
Medical unit stock management and replenishment (handling of incoming and wasted materials, dispensing and keeping the warehouse management system up-to-date, defining replenishment requests), Pharmacy requirements assessment.
The first element to consider when modelling the (b) process (and later (c) too) is the management policy adopted. Basically, the two fields of intervention which need to be globally optimized are medical unit replenishments and pharmacy supplies. Look-back (for example Re-Order Level, Re-Order Cycle, Just in Time), look-ahead (for instance Material Requirements Planning) or mixed (for example Vendor Managed Inventory) approaches can be used.
As Nicholson et al. [6] claim, the most traditional servicing approach in the pharmacy management field is the periodic review par level (or order-up-to level), that requires setting the review interval and the optimal security stock (base stock level). While the second depends on therapeutic and medical constraints set by taking into account demand variability, the first has to be defined according to the involved resources.
Some examples of re-ordination strategies are given by Kalmeijer et al. [23], who promote the extensive use of information systems to manage requirements considering the default medication database as the local stock. Not-stock items are automatically ordered from pharmacy, instead.
In general, the look-back approach is more popular than the others, but brings higher inventory levels. Moreover, forecasts on aggregate data about consumptions recorded by pharmacy are influenced by medical unit management techniques [6]. Look-ahead methods, instead, usually need careful and punctual information about requirements forecasts.
While the BPMN representation of this process is a subset of the one shown for the process (c), the mathematical formalization of the involved inventory management procedures is as follows:
Our integrated approach to the HMM process, oriented to the information management and traceability, allows the adoption of Materials Requirement Planning (MRP) techniques [22]. In particular, starting from the patient prescriptions collected in sub-process (a), it is possible to plan medical unit replenishment actions and supplier order releases.
The following is first defined:
SSm,f: safety stock quantity for drug f at the medical unit m (expressed in units of dose)
Qwm,f,tj: quantity of the f drugstored in the warehouse of the medical unit m at the beginning of the time bucket tj (expressed in units of dose)
GRm,f,tj: gross requirement, that is the total quantity of drug f required during the tj time bucket at the m medical unit.
SRm,f,tj: scheduled receipts, which give the quantity of drug f (defined according to a chosen policy) that is planned to be delivered to the medical unit m at the beginning of the time bucket tj
AVm,f,tj: availability, which is the quantity of drug f to be retained for administrations at the medical unit m at the beginning of the time bucket tj
NRm,f,tj: net requirements, i.e., quantity of drug f required and not available for administrations at the medical unit m at the beginning of the time bucket tj
The replenishment policy adopted depends, among other things, on [24]:
lot sizing policy (lot for lot, Economic Order Quantity, Fixed Order Period, etc.),
lead time offsetting (at least equal to TTm).
Consequently, the decision maker (usually the medical unit nurse manager) can release an order to the hospital pharmacy for the transportation quantity (PORm,f,tj, planned order releases for the medical unit m, the drug f at the time buckettj) to satisfy the requirements of the medical unit m at the kth time bucket (where k ≥ tj + TTm), dependent on the policy and at least equal to the net requirement rounded to the nearest integer of Nf.
The aggregate information may be directly used by the centralized inventory management process in order to plan medical unit distributions and supplier orders, freeing medical unit staff from managerial activities and making more aggregated and powerful data available about medical unit requirements and hospital pharmacy availability over the time.
3.3 Centralized Inventory Management (c)
The centralized Inventory Management process (Figure 4) is triggered by a medical unit requirement and can be interpreted as substitutive with respect to the medical unit inventory management, or in line with it, as already explained in the previous section.
The involved sub-processes are:
Pharmacy stock management, order disposition and supplying activities, Internal distribution, Materials admission, quality control and payment.
3.2.1 Pharmacy stock management, order dispositions and supplying activities (c.1)
Based on pharmacist order assessment, the stock management and supplying activities take place. Operationally speaking, the tasks carried out by pharmacy unit actors are (N frame of Figure 4):
incoming and expired materials handling, warehouse management system updating, budget reconciliation assessment (each cost centre/medical unit typically has its own budget to manage for each expenditure class; the same goes for materials belonging to tenders, which have their specification budget. Materials have to be transferred according to these; otherwise, a budget integration has to be requested from the Superintendence and Treasurer's office reporting quantities and the amount of extra funding needed), stock levels control and authorization to dispense, supplying activities.
As with medical unit stock management, different inventory management policies can be implemented in order to take frequency/quantity supplying decisions.
This “Central of Purchasing” collects the orders and forwards them to the suppliers, feed backing information such as order acceptance or delivery due date to the ordering point, with the exception of life-saving drug stock-out, which is managed by urgent procedures.
Finally, the O frame of Figure 4 illustrates the design of the urgent deliveries management process described in the sub-process (a).
In terms of order releases, the same considerations and formalization already expressed for the process (b) can be repeated. In this case, the medical unit under examination is the central pharmacy, with the exception of the Gross requirement. This is given by:
This time, the results of the calculations are the array of orders to suppliers, which are decided, again, according to the policy chosen and the feasibility evaluation of transportation to the medical unit (in this last case, some urgent procedures may be needed in order to avoid stock-outs).
3.2.2 Internal distribution (c.2)
The internal distribution process begins with the pharmacist's authorization triggered by medical unit replenishments, and is eventually completed with the supply order (also transient materials), as shown in the Q frame of Figure 4, which is composed of order preparation and delivery to medical units.
Inside hospital pharmacy delivery systems have a key role in hospital service quality [25]. The diffusion of the multi-echelon inventory problem as opposed to scheduling-oriented ones [7] has been often dealt with in the literature, and different transportation solutions for optimized routing problems (see, e.g., [21]) have been developed, in which transportation costs and resources (both personnel and physical resources) are some of the main variables taken into account.
The order preparation, performed by storekeepers, consists of printing (also virtually) the materials list released by pharmacists for a medical unit (if the list exists and no refilling method is applied), labelling and filling a bag/receptacle with all materials listed. FIFO logic should be adopted, considering materials' expiration date.
The second task, delivery to the medical unit, can be carried out immediately or according to a schedule by transporters (these may be pharmacy personnel or the medical unit's nursing staff).
Finally, materials admission, quality control and payment (c.3) are carried out by pharmacy personnel (P frame of Figure 4).
4. Expected performances of HMM: case studies
The proposed HMM has been tested both for usability (par. 4.1) and economic convenience (par. 4.2).
4.1 Usability
First of all, we test usability and completeness in the clinical aspect of the BPM. During the study we made an executable version, implementing the user interfaces and the underlying database by means of the software Bizagi©. We subjected one medical unit and the pharmacy personnel of an Italian acute care hospital with 200 beds and 16 specialist medical areas to this prototype. Specifically, the following were involved:
Medical unit (cardiology) personnel:
2 physicians,
1 nurse manager,
1 nurse;
Hospital pharmacy personnel:
hospital pharmacy director,
2 pharmacists,
1 storekeeper.
The subjects feed-backed improvements to the modelling, helped better characterize user requirements and realize customizations (to be adapted to physical resources and the type of available job positions).
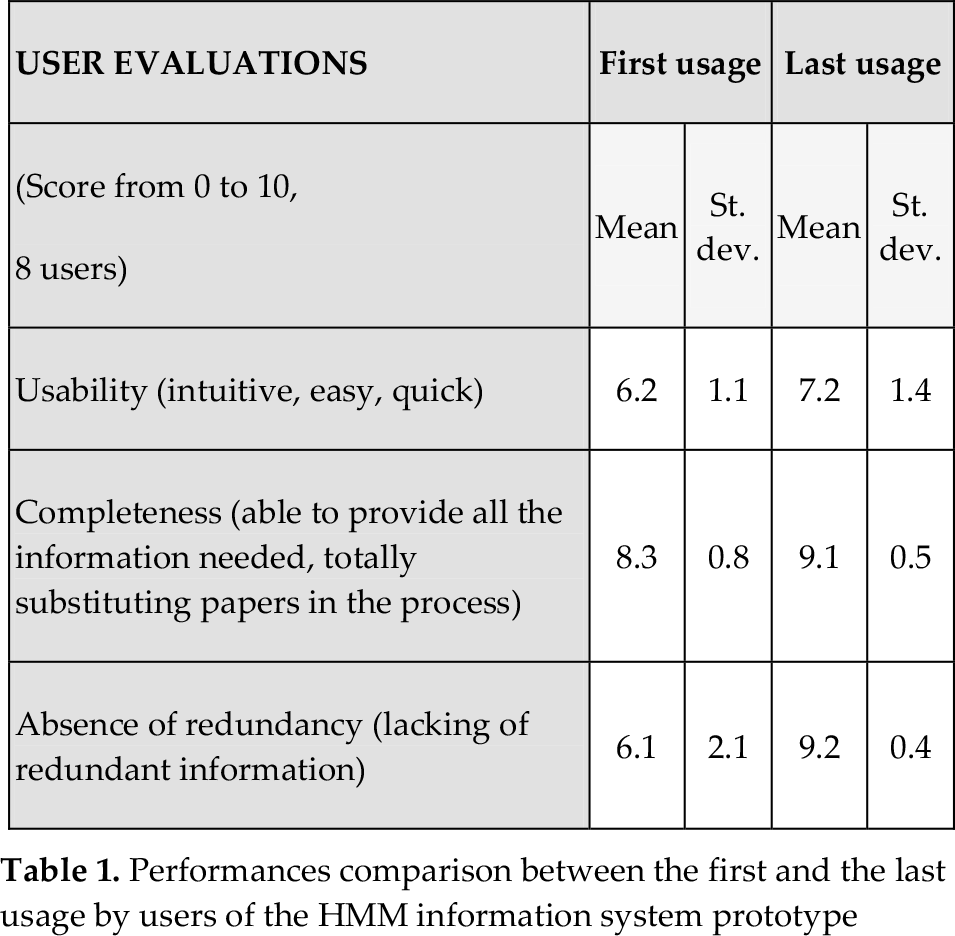
Overall, we recorded great satisfaction in the adoption of the model, as shown in Table 1, which compares the score obtained by users during the first (after the first implementation) and the last (at the end of the study, when the prototype included all the suggested improvements) prototype experimentation.
Performances comparison between the first and the last usage by users of the HMM information system prototype
4.2 Economic convenience
In this paragraph we analyse the feasibility and the profitability of the MRP technique presented in par. 3.1.5 and formalized in equations 1 to 5.
This step is particularly critical for hospital managers and it is obviously not advisable to adopt it without previous performance forecasts and analysis. The choice was to design a simulation model able to represent the main physical characteristics and boundaries of the system by means of input variables, and collecting and summarizing performances by measuring well-defined output variables.
With this aim, we developed a detailed discrete event simulation model (using Arena Rockwell©) able to compare two scenarios:
the “AS IS” scenario, (the “traditional way to manage hospital materials”), a periodic review par level servicing approach, set on the basis of past consumption, keeping distributed (among ward warehouses) safety stocks to prevent stocks-out, the “TO BE” scenario, which involves a delivery scheduling based on patient requirements (MRP technique).
The performance evaluations are carried out through an inventory cost function (C) that takes into account the overall cost sustained by a hospital in adopting an inventory management policy; in particular:

The simulation model has been validated and verified using data from an Italian regional university acute hospital with 700 beds and 26 specialist medical areas.
The
The comparison between the two scenarios and its positive first results (a saving of 10%) are summarized in Table 2 (input data), Table 3 (example of input data related to drugs), Figure 5 (inventory build-up diagram of a drug managed by a medical unit in the two scenarios), Figure 6 (inventory build-up diagram of a drug managed by the hospital pharmacy in the two scenarios) and Table 4 (outputs).
Configurations of the input parameters for the two scenarios

For more details, see Table 2
Demand distribution data for the first 10 drugs used in a medical unit 1 (m = 1)
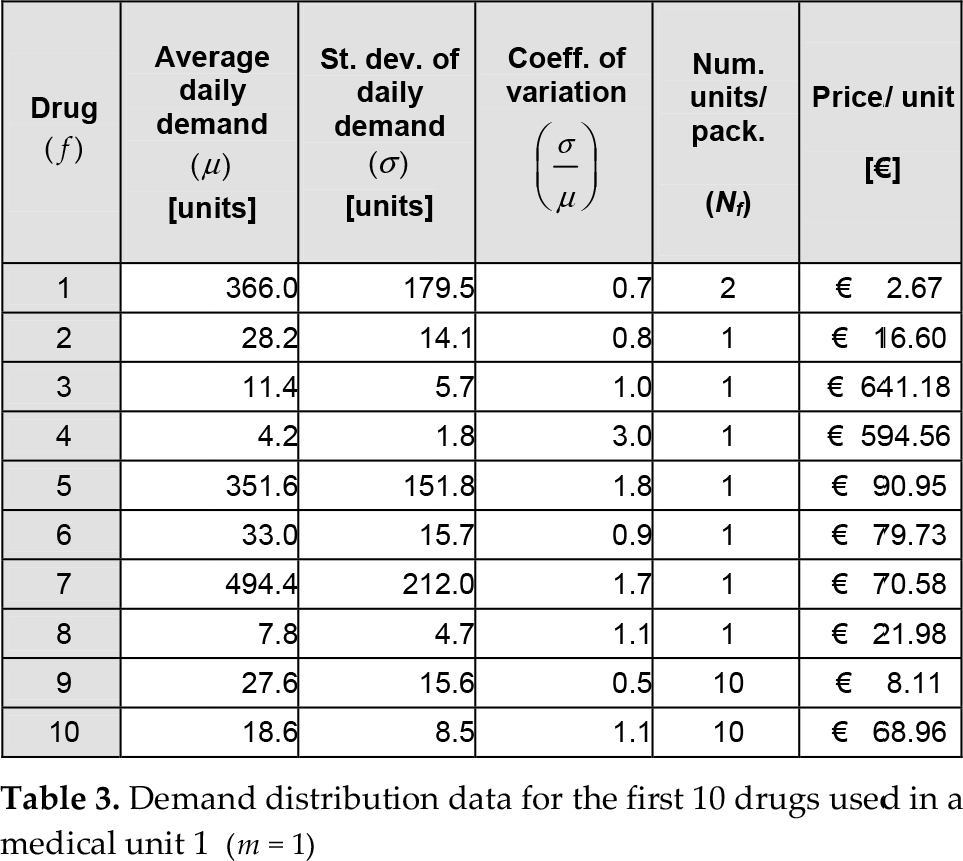
Comparisons of the two scenarios on the basis of the cost function

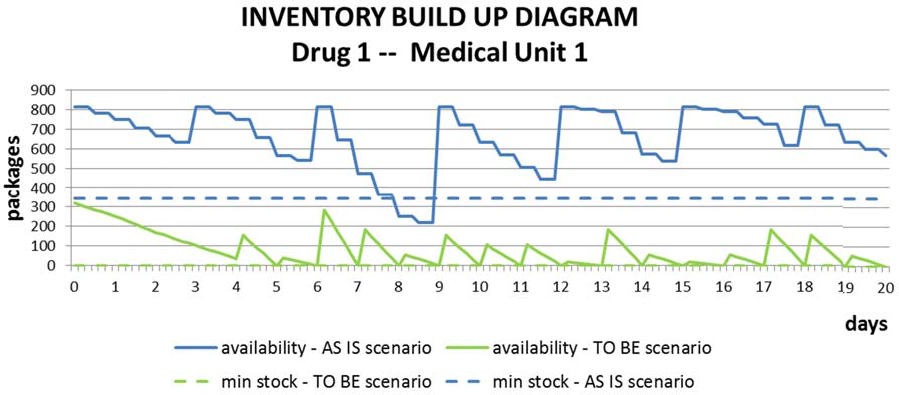
Inventory build up diagram of a drug (f = 1) in a medical unit (m = 1) in the two scenarios.

Inventory build-up diagram of a drug (f = 1) in the hospital pharmacy (m = h) in the two scenarios.
The simulation study is ongoing in order to complete analysis of the effects of multiple factors.
5. Conclusions
This paper proposes an integrated and detailed analysis of the hospital materials management processes and translates this into a Business Process Model, merging clinical and managerial objectives. From the clinical perspective, it allows the enhancing of patient safety through clinical risk reductions while limiting, among other things, time-consuming tasks of all actors; this allows readiness in case of recall and the efficient managing of drug expiration dates. From the management point of view, the model lays the foundations for optimizing the HMM, because an exhaustive, robust and flexible basis of information can allow (as an option of the model) the execution of all logistical activities by a centralized function (the hospital pharmacy), allowing medical staff to devote their time to clinical activities and programming replenishment and supply tasks on the basis of the materials requirements of patients.
Moreover, the concept behind business process languages (such as the BPMN used here) is to make a process executable and hence amenable to quantitative analysis (in primis the simulation, as the most frequently proposed in the literature [19]), being a useful method to evaluate the expected organizational impact on performances of the HMM. This feature was exploited in the first testing step to assess the usability and the completeness of the model by testing the information system prototype in a medium-size hospital.
Finally, a clear mathematical formalization of main requirements for the inventory management, according to an MRP technique, allows the easy and innovative managing of all requirements for materials coming from patient prescriptions. A simulation is carried out (using data from a big university hospital), showing a cost saving of 10% compared to a “traditional” policy.
The complexity and variety of the hospital processes and management policies make this study limited due to the small number of case studies analysed.
Nevertheless, modelling the data sharing and the integration and coordination among actors and activities, the proposed HMM gives the guidelines for the design of a common and distributed information platform that can be also be used to collect data, identify key performance indicators and compare them in different management technique scenarios, carrying out wider performance evaluations.