Abstract
Raman scattering investigations based on density functional theory (DFT) calculations were performed to explore the vibrational modes of wurtzite structured CdS nanoparticles (NPs). The calculations were performed to obtain the Raman spectra for the CdS containing 2, 4, 8 and 12 atoms to study the size dependence. Several vibrational modes indicating stretching and bending features related to Cd and S atoms were observed. Modifications of the frequency and intensity of different Raman modes with an increase in number of atoms in NPs are discussed in detail. It is found that the frequency of the CdS symmetric stretching mode of vibration shows a consistent red shift and that of CdS anti-symmetric stretching shows a consistent blue shift with the increase in the number of atoms. Hydrogen atoms were added in order to make the closed shell configuration and saturate the NPs as per the requisite for calculating the Raman spectra. This produced some additional modes of vibration related to hydrogen atoms. The SH stretching mode showed a consistent red shift and the CdH stretching mode showed a consistent blue shift with an increase in the number of atoms in NPs. The results generated are found to be in close agreement with the literature. The observed red shift in different modes is assigned to stimulated Raman stretching and blue shift is ascribed to the coherent anti-stokes Raman scattering.
1. Introduction
Nano materials have attracted great research and industrial interest in recent years because of their unique chemical, physical, optical, electrical and transport properties [1, 2]. The study of semiconductor nanoparticles has been an interesting field of research for more than two decades. This is because it gives an opportunity to understand the physical properties in low dimensions and to explore their vast potential for application, e.g., optoelectronics [3–6]. Over the past two decades research on quantum-sized semiconductor particles has increased enormously due to their novel applications in electronic and photonic devices [7–10]. The functionality of these devices has strong correlation with the electronic and structural properties of the material used. These properties depend upon the vibrational characteristics of the materials which are in turn related to the size of NPs and can be best explored using infrared and Raman scattering techniques [11–13].
CdS is a promising candidate among II-VI compound semiconductors due to its exceptional chemical and physical properties. It has been used as a window material for hetero junction solar cells to avoid the recombination of photo generated carriers, which improves the solar cells' efficiency [14]. CdS quantum dots have also been used on the substrate in the solar cell to absorb sunlight and produce more free electrons to enhance the efficiency of solar cells [15–17]. Besides using CdS in bulk form, its nanoparticles have shown exciting applications in photochemical catalysis, light emitting diodes, photo resistors and most recently in biosensors to enhance the sensitivity for the detection of the DNA hybridization [18–21]. A significant number of research groups are involved in synthesis and investigations of physical and chemical properties of CdS. The study of the size dependence of CdS NPs is an exciting research area since it provides tunable physical and chemical properties. However, few reports are available on the experimental study of vibrational dynamics of CdS [22–23] and the literature is still lacking on its counterpart using density functional theory-based calculations.
The structures of NPs can be stabilized electrostatically by attaching ligand with a negatively charged end group, thereby introducing repulsive force between the particles to keep them separated. For this purpose hydrogen may be used because it has an ambivalent nature. The study of hydrogen in crystalline semiconductors has been an active research area because of the ability of hydrogen to saturate the nanoparticles, passivate the surface states, terminate dangling bonds and remove other deleterious defects [24]. The presence of hydrogen in semiconductors is ambivalent and may therefore replace a cation or an anion, or form complexes with intrinsic impurities. Many groups have investigated the vibrational modes induced by hydrogen atoms in different semiconductors like Si [25, 26], ZnSe [27], GaAs [28, 29], etc. Although vibrational properties of hydrogenated III-V and II-VI materials have been reported [30], no report is available in the literature on hydrogen in CdS NPs to best of our knowledge. In this paper we report DFT-based Raman spectroscopy to study the vibrational modes of hydrogenated CdS NPs sized around one nanometre.
2. Computational Details
All the calculations which are described in this work are density functional theory (DFT) based and were performed in the Amsterdam density functional (ADF) program of version 2011.01 under local density approximation LDA-VWN (Vosko-Wilk and Nausir) using DZ basis sets. The ADF-GUI (ADF-graphical user interface) version was used for generating the Raman spectrum of wurtzite (WZ) structured CdS-NPs of different sizes around one nanometre. All NPs were relaxed by performing geometric optimization to obtain the stable structure with minimized energy. In the calculation, we distinguished the Cd (1s22s22p63s23p64s23d104p6) and S (1s22s22p6) as core states, and Cd (5s24d10) and S (3s23p4) as valence states. In order to provide electrostatic stabilization of the structures and saturation of the CdS-NPs to complete the outer most shell, we used hydrogen which is requisite for the calculation of Raman spectroscopy. In order to carry out a systematic study of the vibrational properties, different sized CdS-NPs of wurtzite structure (WZ) are prepared computationally. The accuracy parameter was 3.5. We performed geometry optimization of the molecules with energy convergence criteria of 10−6 Hartree, gradient convergence criteria of 10−6 Hartree/Å and radial convergence criteria of 10−2 Å. Table 1 shows bond lengths and bond angles for CdS-NPs.
Structural detail of WZ Cd-NP (all bond lengths are in picometre (10−12m) and bond angles are in degrees).

3. Results and Discussion
Figure 1 shows the Raman spectra for the WZ CdS-NPs of 2, 4, 8 and 12 atoms. The spectra, for the sake of simplicity, are divided into regions of low frequency (<600 cm−1) and high frequency (>600 cm−1). Low frequency Raman modes are in the form of a set of small peaks present in the extreme left and high frequency Raman modes are in the form of dominant peaks in the remaining part of the spectra. The high frequency Raman modes observed at a frequency of 1562 cm−1 and 2789 cm−1 for 2 atom CdS-NPs are Cd-H anti-symmetric stretching S-H anti-symmetric stretching modes respectively as per the ADF library and the literature. These Raman modes are also observed for the CdS-NPs of 4, 8 and 12 atoms (Figure 1) with their characteristic frequencies shown in Table 2.

Calculated Raman spectra for CdS-NPs of 2, 4, 8, and 12 atoms
Raman shifts for Cd-H and S-H anti-symmetric stretching in the WZ CdS-NPs of 2, 4, 8 and 12 atoms.

Note: (
The following sections describe a detailed analysis of the frequency and intensity dependence of different modes of vibration as a function of the number of atoms. Figure 2 shows such variation in frequency for Cd-H stretching and S-H stretching modes.

Variation in frequency for Cd-H stretching and S-H stretching modes as a function of number of atoms in hydrogenated CdS-NPs
3.1 Cd-H Anti-symmetric Stretching (Strong)
The Cd-H stretching mode of vibration is observed at frequencies of 1562 cm−1, 1564 cm−1, 1573 cm−1 and 1591 cm−1 for CdS-NPs of the 2, 4, 8 and 12 atoms respectively as shown in Figure 3. The reported values of characteristic frequency for Cd-H stretching mode are 1430 cm−1, 1775 cm−1 and 1836 cm−1, etc., depending upon different conditions of NPs size, pressure and temperature [30]. There is an increase in frequency (blue shift), as well as intensity with an increase in the number of atoms.

Calculated Raman spectra of CdS-NPs of 2, 4, 8 and 12 atoms. Lorentz multi peak fitting analysis is also given to deconvolute the different modes.
3.2 S-H Anti-symmetric Stretching (Strong)
The S-H anti-symmetric stretching Raman mode is observed at frequencies of 2789 cm−1, 2787 cm−1, 2779 cm−1 and 2777 cm−1 for the WZ CdS-NPs of 2, 4, 8, and 12 atoms respectively. In the literature the vibrational frequency for S-H stretching is about 2675 cm−1 [31]. The frequency of this Raman mode decreases (red shift) and intensity increases with the increase in the number of atoms as shown in Figure 3. To examine the low frequency Raman modes of vibrations we deconvulated the Raman modes using a Lorentz multi curve fitting analysis as shown in Figure 4.

Variation of (a) frequency and (b) intensity of different Raman modes of vibration with the increase in the number of atoms in CdS-NPs
In the Raman spectrum of 2-atom CdS-NPs, different modes were identified in such a way that the mode at 81 cm−1 CdH is the rocking mode, the mode at 92 cm−1 is the SH rocking mode, the mode at 260 cm−1 is the CdS symmetric stretching mode, the mode at 270 cm−1 is the CdS anti-symmetric stretching mode, the mode at 308 cm−1 SH is the rocking mode, the mode at 413 cm−1 is the SH twisting mode and the mode at 451 cm−1 is the CdH rocking Raman mode. All these Raman modes were also observed in the CdS-NPs of 4, 8 and 12 atoms with some shift in the frequency as given in Table 3.
Modes of vibration found in the low range of frequency range (0-600 nm) of Raman spectra of CdS-NPs of 2, 4, 8 and 12 atoms.
Note: (
3.3 Raman Shift
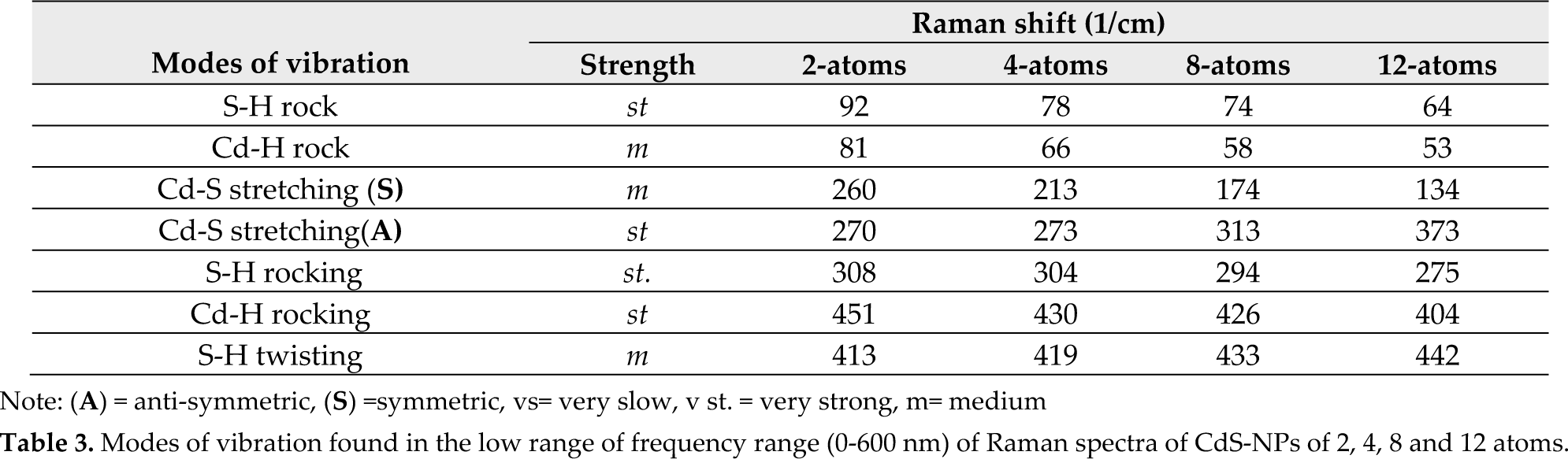
The frequency variation of the observed Raman modes plotted as a function of the number of atoms is shown in Figure 5 (a).
(a) Calculated partial vibrational density of states as a function of frequency and (b) variation of partial vibrational DOS with the increase in the number of atoms in CdS-NPs
3.3.1 Cd-H Rock (Slow)
The values of the frequency of Cd-H rocking mode were observed at 81cm−1, 66 cm−1, 58 cm−1 and 53 cm−1 for CdS-NPs of 2, 4, 8 and 12 atoms respectively. This shows red shift with an increase in the number of atoms in the NPs.
3.3.2 S-H Rock
The trend of variation in the values of the frequency for the S-H rock mode of vibration is such that it is found at 92 cm−1, 78 cm−1, 74 cm−1 and 64 cm−1 for CdS-NPs of 2, 4, 8 and 12 atoms respectively. This shift towards a shorter frequency is again a red shift as the number of atoms in NPs increases.
3.3.3 Cd-S Symmetric Stretching
The CdS symmetric stretching mode of vibration for the CdS-NPs of 2, 4, 8 and 12 atoms are observed at 260 cm−1, 213 cm−1, 174 cm−1 and 134 cm−1 respectively. There is a decrease in Raman shift as the number of atoms increases which indicates the red shift.
3.3.4 Cd-S Anti-symmetric Stretching
The Cd-S anti-symmetric stretching mode was observed at 270 cm−1, 273 cm−1, 313 cm−1 and 373 cm−1 for the NPs of 2, 4, 8 and 12 numbers of atoms respectively. This Raman mode shows the blue shift with the increase in the number of atoms in NPs.
3.3.5 S-H Rock (Strong)
The SH strong rocking is observed at 308 cm−1, 304 cm−1, 295cm−1 and 275 cm−1 in the CdS-NPs of 2, 4, 8 and 12 atoms respectively. This shows a consistent decrease in the frequency as the number of atoms increases, pointing towards red shift.
3.3.6 Cd-H Rock (Medium)
The Cd-H rocking mode was found at 451 cm−1, 430 cm−1, 426 cm−1 and 404 cm−1 for the CdS-NPs of 2, 4, 8 and 12 atoms respectively. The Raman shift for Cd-H rocking mode of vibration shifts towards a shorter frequency, i.e., red shift.
3.3.7 S-H Twisting (Strong)
The Raman shift for the S-H twisting mode was observed at 413 cm−1, 419 cm−1, 433 cm−1 and 442 cm−1 for the NPs of 2, 4, 8 and 12 atoms respectively. This shows a consistent blue shift. In the literature the vibrational frequency for the SH twisting calculated using the DFT method is 439 cm−1 [31].
The plot of Raman intensity of different modes of vibration as a function of the number of atoms in NPs is given in Figure 4 (b). The Raman intensity of almost all the modes of vibrations increases slowly, but intensity of SH stretching and CdH stretching increases rapidly with the increase in the number of atoms in the NPs. This increase in Raman intensity is reasonable because an increase in the number of atoms enhances the probability of inelastic scattering.
3.3.8 Density of States of CdS-NPs
Partial vibrational density of states of the CdS-NPs of 2, 4, 8 and 12 atoms are shown in Figure 5. All modes exhibit a similar trend of increase in intensity as a function of the number of atoms in molecules (Fig. 5b) which is an obvious effect of the increase in atoms and thus bonds.
Figure 6 shows electron density of states for the structures under study. The electronic densities of states are increasing as the number of atoms in the CdS-NPs increases. Due to the increase in the number of atoms, the number of electrons and the number of available states for the electrons increases which results in the increase of density of states.

Calculated electron density of states showing occupation of energy states of CdS-NPs for 2, 4, 8 and 12 atoms
DFT calculations are employed to determine the optimized geometries of the CdS-NPs with (CdS)n where n is the number of atoms of sulphur and cadmium n= 2, 4, 8 and 12. The highest occupied molecular orbital (HOMO) and lower unoccupied molecular orbital (LUMO) energy diagrams of the Kohn-Sham orbitals were evaluated and shown in Figure 7. HOMO consisted of S 3p, but LUMO consisted of Cd 5s and H s for 2 atom structures, Cd 5s and S p for 4 atom structures, Cd 5s for 8 atom structures and S 4s, S 3p for 12 atom CdS structures.

Molecular energy level diagram showing levels of Cd, S, H and CdH (2, 4, 8 and 12 atoms) modes in CdS-NPs
The decrease in frequency as the number of atoms increases is assigned to stimulated Raman scattering as shown in Figure 8 (a). When a photon of frequency ‘
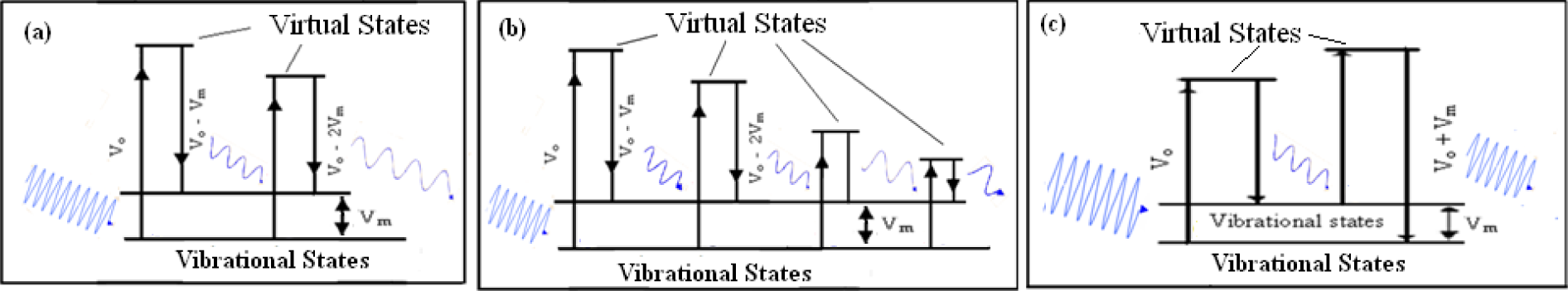
Schematic of (a) mechanism of scattering, (b) stimulated Raman scattering (SARS) and (c) coherent anti-stokes Raman scattering (CARS)
The increase in the frequency with an increase in the number of atoms is assigned to coherent anti-stokes Raman scattering (CARS) explained in Fig. 8 (c). A photon of frequency Vo is incident to promote an atom from a vibrational state to some excited state via Raman stokes scattering. The scattered photon is then incident on another atom, present already in an excited vibrational state, to go through a transition, thereby emitting a photon with higher frequency Vo + Vm. Hence CARS refers to an increase in frequency, i.e., blue shift, of photon while scattering through the material. When the number of atoms in NPs increases, the probability of the number of atoms in initially excited states increases which further enhances the CARS and consequently the blue shift.
4. Conclusion
The study aimed to explore the properties of wurtzite structured CdS nanoparticles by investigating the properties of CdS molecules consisting of 2, 4, 8 and 12 numbers of atoms. The structures were saturated by the hydrogen in the ADF-molecule user interface computational program based on density functional theory (DFT). The DFT calculations were performed to obtain the Raman spectrum for the wurtzite (WZ) CdS-NPs of 2, 4, 8 and 12 atoms and each Raman mode was investigated. It is found that the Raman shift for CdS symmetric stretching modes of vibrations shows red shift with the increase in the number of atoms due to stimulated Raman scattering and for CdS anti-symmetric stretching shows blue shift with the increase in number of atoms due to coherent anti-stokes Raman scattering (CARS). There are also some other mode vibrations which arise due to the saturation of CdS nanoparticles with hydrogen, like SH stretching, which shows red shift due to stimulated Raman scattering and CdH stretching which shows blue shift due to CARS as the number of atoms in WZ CdS-NPs are increased.
