Abstract

Baud MO, Kleen JK, Mirro EA, Andrechak JC, King-Stephens D, Chang EF, Rao VR. Nat Commun 2018;9:88.
Epilepsy is defined by the seemingly random occurrence of spontaneous seizures. The ability to anticipate seizures would enable preventative treatment strategies. A central but unresolved question concerns the relationship of seizure timing to fluctuating rates of interictal epileptiform discharges (here termed interictal epileptiform activity, IEA), a marker of brain irritability observed between seizures by electroencephalography (EEG). Here, in 37 subjects with an implanted brain stimulation device that detects IEA and seizures over years, we find that IEA oscillates with circadian and subject-specific multidien (multi-day) periods. Multidien periodicities, most commonly 20–30 days in duration, are robust and relatively stable for up to 10 years in men and women. We show that seizures occur preferentially during the rising phase of multidien IEA rhythms. Combining phase information from circadian and multidien IEA rhythms provides a novel biomarker for determining relative seizure risk with a large effect size in most subjects.
Commentary
Open your favorite cell phone app and look for your sleep statistics in a day, in week, in a month. A wave on a wave, we are surrounded by rhythms. Their fractal anatomy is nothing novel, certainly not for neuroscience. Circadian fluctuations influence behavior and brain electrical activity, which are organized from infra-slow (0.01–0.1 Hz) to ultra-fast frequencies (250–800 Hz) passing through delta (1–4 Hz), theta (4–12 Hz), beta (10–30 Hz), gamma (30–90), and ripples (100–250 Hz; 1). Strikingly, these rhythms nest one inside another from fast to slow in a hierarchy. Brain activity is a harmonious dance. And epilepsy is a disease of brain activity. So, is there anything harmonious about epileptiform activities? A recent paper by Baud and colleagues, published in Nature Communication, suggests that periods of 15 to 30 days may influence seizures.
The idea has old roots. In spite of controversy, the moon and epilepsy have been considered a dance partner since the annals of medicine. In Baud et al., there was no such perfect astronomical alignment but instead variable multiple-day periods of interictal epileptiform activities (IEAs) across subjects: 15, 20, 26 days. Similar clustering was noted before both in animal models and human epilepsies (2–4). What is new?
Two major conceptual advances are introduced from this paper, results of the unique opportunity to examine intracranial recordings obtained with the use of NeuroPace systems over years. First, a subject-specific interaction emerged between circadian and multidien oscillations when accounting for the temporal structure of IEAs. Second, for each individual, seizure timing exhibited striking regularities in a narrow phase range. Importantly, an elegant analysis identified the highest risk for seizures at the intersection between the circadian and multidien peaks of IEAs. Some caution is worth noting, though: the possible influence of neurostimulation and a methodological requirement of a large number of events for a confident circadian–multidien joint-risk assessment.
Slow brain oscillations of 0.5–1 Hz reflect coordinated fluctuations of neuronal excitability in which neurons switch from hyperpolarized to depolarized membrane potentials (5). The slow wave sweeps the cortex, the hippocampus, and the thalamus to influence faster rhythms during sleep, such as cortical delta waves, thalamocortical spindles, and hippocampal ripples (5, 6). The transition from hyperpolarized to depolarized potentials triggers or facilitates emergence of faster events within each of these microcircuits. During active exploration, a similar slow modulatory rhythm interacts with hippocampal theta oscillations (7), which in turn modulate the amplitude of gamma activities (8). Intracellularly, that reflects the interplay between synaptic and membrane potential fluctuations, bringing neurons closer or further away from their firing threshold at a shorter time scale (9). Above all, an infra-slow rhythm (0.02–0.2 Hz) modulates global excitability over minutes (10).
How does it work? Imagine the day and the year as a torus (Figure a). Excitability fluctuates slowly from morning to afternoon, a loop around the torus surface, while time is passing to another day, pushing your loop ahead. Day after day, circadian fluctuations evolve in a multidien rhythm. But if you look closely within a day, an infra-slow oscillation is there too. It is as though the torus surface were not flat but actually wavy or corrugated (Figure b). Then, if you focus on one of these infra-slow oscillation cycles, you find still more waves. Waves upon waves of fluctuating excitability. If one considers all these fluctuations, there will be a moment when all conspires.
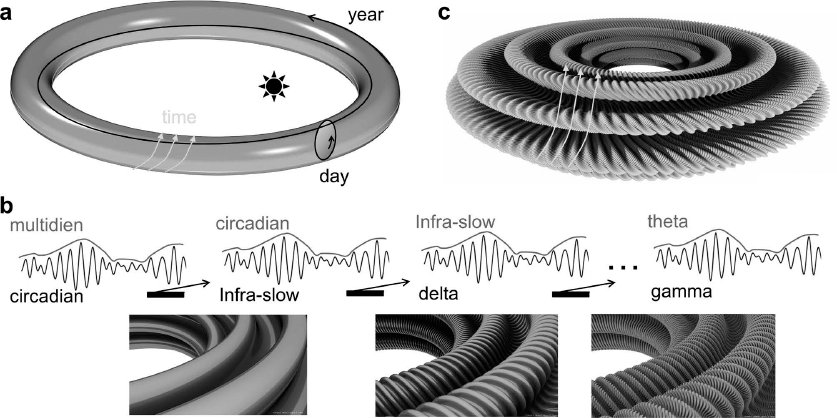
(
For people suffering from epilepsy, any fluctuating changes of excitability represent an opportunity for seizure initiation. Occurrence of seizures and IEAs have been found related to instances of high-frequency oscillations or fast ripples, gamma, theta, slow, and infra-low oscillations (3, 4, 10–12). Possibly, such a nested structure of IEAs and seizures always points downward to the excitability level of single cells, reaching the firing threshold for an uncontrolled coordinated activation. Data by Baud and colleagues suggest that any pre-ictal period reported so far is just reflecting a window of opportunity of enhanced excitability (13) and that relying on such a period for forecasting seizures without incorporating modulatory influences must necessarily be suboptimal. This should have a profound impact on future detection algorithms for seizure anticipation.
However, the egg question is mandatory: What came first? Is there a mechanism (or several) to nest fast brain rhythms into slower oscillations? Many suggested 1-Hz oscillations reflect pacing from subcortical modulatory inputs (5), similar to how suprachiasmatic clocks set circadian oscillations (14). This could imply a causal flow from slow to fast rhythms so that, by interfering upstream, one can gain control. Another possibility is that processes triggered by faster brain rhythms converge to leak over slower time scales. For instance, diverse forms of plasticity can be triggered by high-frequency firing to last seconds, minutes, hours, or days. Slower rhythms may well reflect a resonant phenomenon generated by faster oscillations driven by uncorrelated noise (15). Or perhaps one can imagine a multidien rhythmicity emerging from a slightly drifting circadian dance that resets at some point. Interestingly, the seizure risk analysis by Baud and colleagues suggests no asymmetry in the phase space between circadian and multidien oscillations. Possibly epilepsy is an oscillatory disease, a rhythmopathy in which one or several of the mechanisms responsible for setting the dance tempo are impaired (16, 17). A lunatic dance after all.
