Abstract
Prognostication after cardiac arrest often depends primarily on neurological function, and characterizing the extent of neurological injury hinges on neurophysiological testing and clinical neurological examination. The presence of early posthypoxic myoclonus (PHM) following cardiac arrest had been invariably associated with poor outcome, but more recent studies have shown that those with early PHM may survive with good neurological function. Electroencephalographic patterns suggestive of severe brain injury may be more valuable than the presence of PHM itself in portending poor functional status, and phenotyping PHM may also be useful in delineating benign and malignant forms. Patients with early PHM should be evaluated similarly to others who suffer cardiac arrest by using a multimodal approach in determining prognosis until further studies are performed that better characterize early PHM subtypes and their outcomes.
Neurologists are often consulted to assist critical care providers in determining prognosis in those who have suffered anoxic brain injury after cardiorespiratory arrest (CRA). Neurophysiological testing and neurological examination have been well studied as useful indicators of outcomes (1, 2). One particular examination finding of prognostic importance is early posthypoxic myoclonus (PHM). Early PHM is defined clinically by focal, multifocal, or generalized myoclonic movements of the face, trunk, extremities, and sternocleidomastoid and trapezius muscles that begin within 72 hours of CRA (3–8). Early PHM arises due to brain injury and subsequent increased neuronal hyperactivity (3, 5, 6). Previous guidelines on prognostication after CRA from the American Academy of Neurology (AAN) concluded that early PHM represents no hope for meaningful recovery (9). This consensus was primarily based on a small study of 40 patients (6) and one other retrospective analysis (10) that demonstrated a 0% false positive rate of early PHM predicting poor outcomes. Other studies around that time also confirmed a poor prognosis in those with early PHM (11, 12). Though more recent studies have reaffirmed the 0% false positive rate in determining poor outcome due to myoclonus (13–19), there have been a number of publications that demonstrate a higher false positive rate (3–5, 20–32), and have since challenged the way we view early PHM (33).
In this article, we review early PHM and its value in prognostication after CRA. We will explain why there are differences in outcomes noted in different studies on PHM, propose a new way in which to view PHM and its utility in prognostication after CRA, highlight the importance of ancillary testing, and lay out goals of future study.
Historical Views of PHM
Early PHM has previously been termed status myoclonus, reticular reflex myoclonus, or myoclonus status epilepticus (MSE; 4). The use of “status” in the terminology is based on the presence of continuous diffuse jerking that lasts a minimum of 30 minutes (9). However, the definition of MSE has become less clear, given its extended use to encompass continuous PHM of shorter and longer durations as well as electrographic status epilepticus following CRA with coincident clinical myoclonus (23). We recommend referring to MSE in its strictest clinical terms, as described in Wijdicks et al. (9) and avoiding the inclusion of EEG characteristics. We will conform to this definition of MSE in this paper.
Previous definitions of PHM relied upon the timing of onset of myoclonus with respect to CRA given that this appeared to be most helpful in determining outcomes (3). Early or “acute” PHM, or MSE, has been differentiated from “chronic” PHM, or Lance-Adams syndrome (LAS), which has been defined as PHM occurring after the acute post-arrest period and portends a better outcome (3). However, cases of chronic PHM may occur within 5 to 8 hours of CRA, therefore making the distinction by time-of-onset less helpful, particularly acutely after CRA (3, 5, 23). The delay in diagnosing some cases of LAS is confounded by the use of sedatives to facilitate ventilator-assisted breathing shortly after CRA, which might mask LAS (3, 23, 31). As a result, patients may be diagnosed with MSE when in fact they have LAS (3, 5, 20), leading to false diagnosis and inaccurate prognostication.
Some authors have tried to localize PHM to a cortical or subcortical generator to differentiate forms of PHM (8, 34). Generalized PHM is believed to originate from subcortical areas (34), which may be consistent with the proximal limb myoclonus often seen in these cases (27). However, a more recent study has demonstrated that localization based on the clinical exam is not consistent (4). Though studies have used neurophysiological testing to determine the origin of subtypes of PHM (3, 27, 35), they have been flawed due to the inconsistent use of EEG–EMG polygraphy and somatosensory evoked potentials (SSEP), both of which have been shown to be the most reliable means of diagnosing cortical myoclonus (3). Therefore, defining cortical and subcortical myoclonus using neurophysiological studies in evaluating early PHM needs further study.
Recent Studies of Early PHM Show Variability in Outcomes
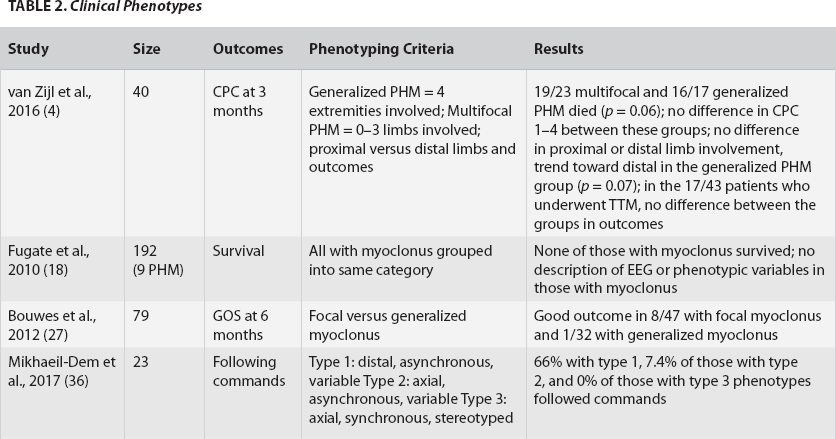
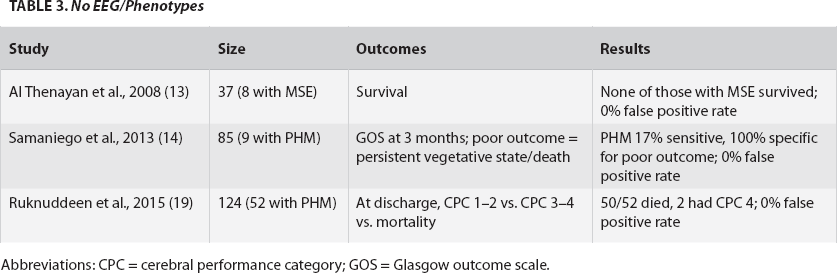
Since the publication of the 2006 AAN guidelines on neurological prognostication after CRA and other studies of PHM (6, 11, 12), a number of case reports, retrospective studies, and systematic reviews have been published demonstrating survival and good neurological outcomes in patients who developed early PHM (3–5, 20–32, 36). Some of these studies demonstrated that there are different subtypes of early PHM and that defining early PHM by neurophysiological and clinical measures may enable prognostication of outcomes (Tables 1–3).
EEG Characteristics

Clinical Phenotypes
No EEG/Phenotypes
Abbreviations: CPC = cerebral performance category; GOS = Glasgow outcome scale.
Neurophysiological Studies May Help Differentiate “Benign” and “Malignant” Forms of Early PHM
There are several malignant EEG patterns seen in patients after CRA, including burst suppression, nonreactive background, alpha coma, generalized status epilepticus, and lateralized (LPDs) or generalized periodic discharges (GPDs; 4, 5, 17, 24, 29, 37). Multiple studies have demonstrated that these EEG patterns, as well as others that signify severe brain injury, predict worse outcomes when found in those with PHM after CRA (4, 5, 21, 24–26, 29). This was also described in a review by Gupta et al. that included 37 articles, of which a number are included in this review (4, 11, 12, 24, 27, 29, and a number of cases of LAS also described in Freund et al. [3]). Other studies have demonstrated the importance of malignant EEG patterns in determining poor prognosis in early PHM but failed to include patients with PHM and more benign neurophysiological findings for comparison (6, 11, 12, 15, 17). Two of these studies together (11, 12) cited nine older studies that reported these patterns typically appeared shortly after CRA in PHM. Other studies have demonstrated the presence of less concerning EEG patterns in some patients with early PHM and subsequently better outcomes (5, 21, 24, 26, 30, 32). Therefore, many of the studies portending very poor outcome in early PHM are likely biased due to the over-representation of patients with malignant EEG patterns, representing graver neurological injury following CRA than in those without PHM (2). This has been supported in one study that showed similar outcomes between those with and without PHM following CRA if EEGs demonstrate GPDs, bilateral independent periodic discharges (BIPDs), or LPDs (32). This suggests that neurophysiological testing may add more to prognostication than the presence of PHM itself, particularly when differentiating between malignant and benign EEG patterns in these patients. Though extremely rare, there may be some who do survive with good outcomes despite EEGs detecting burst-suppression (28), non-reactive background activity (27), and status epilepticus (20). On the other hand, up to 15% of those with poor outcomes may demonstrate reactive or continuous background activity (24, 32), similar to those with good outcomes in one study (32), though all patients described had GPDs or BIPDs detected on EEG which may have confounded this finding.
Phenotyping PHM May Assist in Diagnosing Forms of PHM
Varying degrees of neurological injury can be clinically demonstrated by the presence of generalized versus focal/multifocal PHM (27). Generalized PHM, which is likely associated with more widespread brain damage, may involve axial and proximal limb musculature and may appear more symmetric than does multifocal/focal PHM (4, 36). Worse outcomes have been demonstrated in studies including only those with generalized PHM with 0% false positive rates (6, 11, 13, 14, 16), further suggesting that more generalized forms of PHM portend poorer prognoses. Those with generalized PHM involving axial structures and appearing bilateral and synchronized may have a graver prognosis than those without these features (36). Other studies set out to differentiate PHM by clinical phenotype (4, 27), but their methods were empiric, possibly resulting in little prognostic difference among the groups studied.
Future Directions in Studying PHM
Determining the localization of myoclonus clinically—paying particular attention to involvement of neck, facial, proximal versus distal limb musculature, and limb versus axial musculature, as well as the extent of synchrony of bilateral myoclonic movements—appears to show some promise in understanding clinical phenotyping of early PHM and its importance in outcomes (36). Correlating phenotypes with EEG characteristics would also be useful, as EEG provides valuable information early after CRA in those with and without PHM. Further delineating malignant from benign EEG patterns and understanding their meaning in prognosis in patients with early PHM may hold the key.
In future studies, the timing of EEG should be included in the analysis given that there is variability in EEG findings over time in those with early PHM (5). The assessment of early EEG and its utility in prognostication in CRA, particularly within 24 hours of CRA, has been performed previously but did not focus on those with PHM, and may have been biased by withdrawal of care after treatment-resistant PHM (2).
Conclusions
Since the 2006 AAN guidelines were published, studies of early PHM have been biased by a self-fulfilling prophecy of withdrawal of life support when early PHM arises. However, early PHM does not always represent severe neurological injury. PHM subtypes may be differentiated by clinical features and EEG, and each case requires careful clinical and EEG evaluation to avoid a falsely pessimistic prognosis. In particular, EEG patterns themselves may hold more prognostic value than the development of early PHM. Though recent studies have focused on evaluating clinical and neurophysiological features in early PHM and their utility in predicting outcomes, there remains questions as to how to define the various forms of PHM. Until early PHM subtypes are better characterized, prognosis or medical decision-making should not be based alone on its presence.
Highlights
Early PHM does not invariably predict poor outcomes;
Early PHM subtypes that portend worse prognosis can be identified by EEG patterns suggestive of severe neurological injury including isoelectric activity, burst-suppression, GPDs, status epilepticus, alpha coma, and nonreactive background activity;
Clinical phenotyping of early PHM may be of value in prognostication;
Patients with early PHM should be evaluated using a multimodal approach in determining prognosis, similar to others who present following CRA.
