Abstract

Evidence of an inhibitory restraint of seizure activity in humans.
Schevon CA, Weiss SA, McKhann Jr G, Goodman RR, Yuste R, Emerson RG, Trevelyan AJ. Nat Commun 2012;3:1060. doi: 10.1038/ncomms2056
The location and trajectory of seizure activity is of great importance, yet our ability to map such activity remains primitive. Recently, development of multi-electrode arrays for use in humans has provided new levels of temporal and spatial resolution for recording seizures. Here, we show that there is a sharp delineation between areas, showing intense hypersynchronous firing indicative of recruitment to the seizure, and adjacent territories where there is only low-level, unstructured firing. Thus, there is a core territory of recruited neurons and a surrounding “ictal penumbra.” The defining feature of the ictal penumbra is the contrast between the large amplitude EEG signals and the low-level firing there. Our human recordings bear striking similarities with animal studies of an inhibitory restraint, indicating they can be readily understood in terms of this mechanism. These findings have important implications for how we localize seizure activity and map its spread.
Single-Neuron Dynamics in Human Focal Epilepsy.
Truccolo W, Donoghue JA, Hochberg LR, Eskandar EN, Madsen JR, Anderson WS, Brown EN, Halgren E, Cash SS. Nat Neurosci 2011;14:635–641.
Epileptic seizures are traditionally characterized as the ultimate expression of monolithic, hypersynchronous neuronal activity arising from unbalanced runaway excitation. Here, we report the first examination of spike-train patterns in large ensembles of single neurons during seizures in persons with epilepsy. Contrary to the traditional view, neuronal spiking activity during seizure initiation and spread was highly heterogeneous, not hypersynchronous, suggesting complex interactions among different neuronal groups, even at the spatial scale of small cortical patches. In contrast to earlier stages, seizure termination is a nearly homogenous phenomenon followed by an almost complete cessation of spiking across recorded neuronal ensembles. Notably, even neurons outside the region of seizure onset showed significant changes in activity minutes before the seizure. These findings suggest a revision of current thinking about seizure mechanisms and point to the possibility of seizure prevention based on spiking activity in neocortical neurons.
Commentary
Epileptic seizures propagate extensively across the brain. The associated electrophysiological mass phenomena can be captured by scalp or intracranial electroencephalography. Recent evidence shows the initiating mechanisms of seizures operate at the smaller scale of neuronal ensembles, producing phenomena such as high-frequency oscillations (1, 2) and microseizures (3, 4) that are best detected at a tighter spatial resolution of local field potentials recorded by microelectrodes. More recently, researchers have started using microelectrodes to analyze the activity of large numbers of individual neurons during seizures in neocortex (5) and hippocampus (6) and to relate this activity to the corresponding macroscopic EEG recordings. The main focus of this commentary is the most recent of these studies by Schevon et al., which provides potential solutions to puzzles arising from previous neocortical work.
The idea that seizures represent “hypersynchrony” goes back at least to the 1950s (7). However, until recently, recordings from people with epilepsy suggested that neuronal firing is not hypersynchronous, despite the presence of large rhythmic—arguably hypersynchronous—field potentials. Early studies with single microelectrodes (8, 9) found no evidence for extensive or synchronous neuronal firing during seizures. The advent of dense multi-electrode arrays allows sampling of the activity of many neurons within local regions in the brain. Schevon et al. and Trucculo et al. both used microelectrode (Utah) arrays in the neocortex of epileptic patients. The arrays sampled 96 sites at a depth of 1 mm within a 4 mm × 4 mm area. Closely spaced penetrating electrodes are likely to cause more damage to the tissue than the depth microelectrodes used by Bower et al. (6) in the medial temporal lobe. This means the arrays must be targeted to areas likely to be resected. Fortunately, this is exactly where they need to be to study the initiation of seizures.
All three studies found recordings consistent with earlier evidence of heterogeneous changes in neuronal firing during electrographic seizure activity, both increases and decreases in firing rate with many units not changing (one-third in Trucculo et al. and two-thirds in (6)). In Schevon et al., this activity was found to be unrelated to the field potentials recorded in the same areas; this is surprising because the simplest model would have local neurons driving the synaptic currents that generate most of the field potentials or responding to those synaptic currents.
Results of Schevon et al. differ from those of Truccolo et al. in finding a second class of activity: propagating areas of hypersynchronous neuronal firing at rates > 30 × baseline. This class of activity can also be found in microwire seizure recordings from human medial temporal lobe (Figure 1). Interpretation of the relationship between these two fundamentally different states is based on evidence presented on epileptiform activity in normal mouse cortex in vitro when exposed to a convulsant medium (with lowered magnesium ion concentration). A combination of live-cell imaging and patch clamp recording revealed the epileptiform activity depended on rapid and synchronous firing of clusters of neurons that propagated slowly across the cortical slice. Crucially, areas ahead of the advancing region of neuronal firing experienced a barrage of excitatory and inhibitory synaptic activity, with inhibition dominating and restraining propagation. When the epileptic front had passed, neurons fired briskly and synchronously with phase delays revealing coupling much faster than the epileptic propagation velocity.
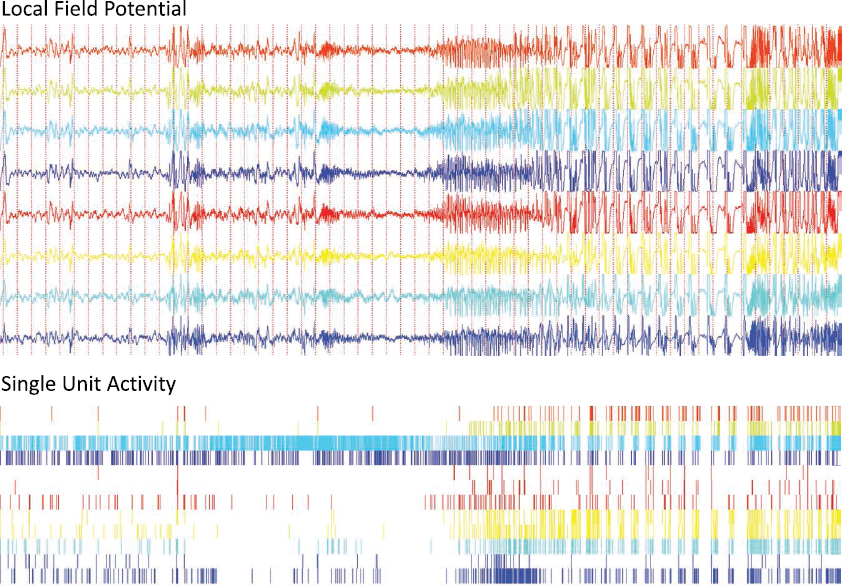
Microwire recording of a medial temporal lobe seizure acquired at the Dept. of Epileptology, University of Bonn. Local field potentials and unit activity from a bundle of eight microwires in the hippocampus are displayed in corresponding colors.
Human recordings by Schevon et al. revealed patterns of firing and relationships with local field potentials very similar to those in the mouse model. They failed to place their arrays over the sites of seizure initiation (although the good outcomes for surgery suggest they were not far off). The advancing wave of hypersynchronous neuronal hyperactivity was preceded by large rhythmic field potentials that were not synchronized with neuronal firing. The human recordings also resembled the mouse model in three ways: a sharp boundary between these two classes of activity, slow propagation velocity of hyperactivity, and much faster synchronization during the period of rapid firing. The experimental and clinical parts of this study provide a good example of how well the two approaches can complement each other. Further, the clinical evidence of hypersynchronous neuronal hyperactivity is also encouraging for animal models of epilepsy where it usually features prominently.
Unit recordings can provide new insights into the process of transition to seizure and seizure prediction. Truccolo et al. observed significant changes in firing rates during the 3-min interval before a seizure from those during the previous 30 min. Based on the number of neurons exhibiting these changes— averaged across different seizures from a given patient and thus corresponding to an algorithmic sensitivity in the range of 50%—they assessed the specificity by sampling random 3-min segments from interictal control epochs away from the seizure. Specificity values for these control epochs ranged between 44% and 94%, thus failing to reach statistical significance for any of the control epochs (p > 0.06). Nevertheless, as the authors correctly state in their cautious interpretation of these findings, these are promising preliminary results that warrant further research.
Another interesting aspect of the work of Trucculo et al. is that seizure termination was accompanied by a prominent decrease in neuronal firing activity, observed both for putative principal cells and interneurons (classified electrographically). In light of results by Schevon et al., this shutdown of activity could possibly be caused by feedforward inhibition generated by a homogeneous activation of inhibitory neurons at a different location. The varying degrees of stereotypical firing patterns between seizures in the same patient (6) might likewise depend on whether or not neurons are actually being recruited by the propagating wave of seizure spread (Figure 1).
One implication from these studies is that the core of the seizure-generating zone is spatially tight, elusive for 4 mm square arrays of microelectrodes. This is interesting scientifically but also has practical implications: The location of the first signs of electrographic seizure activity will be related to the epileptogenic zone, but the length of synaptic projections from the neurons in this zone means that the spatial resolution of field potential recordings is compromised. Perhaps the most striking aspect of these findings is the clear dissociation between the high-amplitude rhythmic field potentials in the lower part of the frequency spectrum (2–50 Hz) thought to reflect synaptic input activity, and the multiunit firing activity reflecting the output of these neurons. While these rhythmic field potentials are traditionally considered the hallmark of propagating seizure activity, they appear to reflect the spatially extended synaptic inputs elicited from a much more focal region of synchronously firing neurons located elsewhere. Actual recruitment of neurons in areas exhibiting electrographic signs of seizure activity may or may not occur; currently, this can be determined only by monitoring the neurons’ output firing (Figure 1; Figure 4 in Schevon et al.).
