Abstract
Purpose
Bevacizumab, a humanized, anti-vascular endothelial growth factor-A monoclonal antibody, has shown efficacy in a number of cancers. However, its use in metastatic breast cancer (MBC) remains controversial.
Methods
A literature review using the PubMed database was performed to update the currently available clinical trials evidence on bevacizumab in the first-line treatment of breast cancer. In addition, the proceedings of selected oncology annual meetings were searched for relevant presentations.
Results
This article reviews the available evidence for bevacizumab as first-line therapy for MBC and discusses its current and future applicability in the management of MBC. Three phase III trials (ECOG-2100, AVADO, RIBBON-1) demonstrated that the addition of bevacizumab to chemotherapy is well-tolerated and improves progression-free survival and objective response rates in the first-line setting. These findings were supported by a large clinical practice-based study (ATHENA) and a recent clinical trial in which bevacizumab added to paclitaxel showed notable activity in triple-negative MBC. However, bevacizumab has thus far not demonstrated a significant benefit in overall survival.
Conclusions
The addition of bevacizumab to chemotherapy is well-tolerated and produces substantial improvements in overall response rate and progression-free survival, compared with chemotherapy alone, in advanced HER2-negative breast cancer. Nevertheless, it has thus far not demonstrated a significant benefit in overall survival. Whether prolongation of progression-free survival is enough to consider bevacizumab efficacious is unclear. Based on the available clinical trials results, bevacizumab is a part of the complex therapeutic strategy of advanced HER2-negative breast cancer.
Keywords
Introduction
Breast cancer (BC) is recognized as a complex and heterogeneous disease due to the diversity of its molecular features, range of multiple morphologic subtypes, and the differences seen in response to therapy and patient outcomes (1). Current treatment for metastatic BC (MBC) includes hormonal therapy or chemotherapy. Taxanes and anthracyclines are the most active chemotherapy agents in BC (1). Furthermore, approaches combining antiangiogenic agents, such as bevacizumab, with standard chemotherapy have been shown in clinical trials to prolong progression-free survival (PFS) without causing unacceptable toxicity.
There is a firm basis to support the rationale of employing antiangiogenic agents in the treatment of cancer. It is well-documented that organ growth and repair is dependent on angiogenesis, the physiologic process leading to the formation of new blood vessels. Disturbance of the tightly regulated physiologic balance underlying angiogenesis is an important factor in the development of a number of pathologic processes, such as inflammatory conditions, malignancies, and vascular and immune disorders (2). Tumor growth and the development of metastatic disease is dependent on angiogenesis. Vascular endothelial growth factor (VEGF) is thought to play a critical role in tumor angiogenesis (3) by stimulating the proliferation and migration of vascular endothelial cells, inhibiting apoptosis and antigen-presenting dendritic cells, inducing extracellular matrix remodeling, and increasing vascular permeability and vasodilation (4). Among various isoforms of VEGF, VEGF-A has been demonstrated to be the most potent in determining vasodilation and tumor angiogenesis, and its function is mediated by binding to VEGF receptors 1 and 2 (VEGFR-1 and VEGFR-2) (5).
A hypothesis generated by Folkman (6) in 1971 proposed that tumor progression could be prevented by interference with tumor angiogenesis, causing inhibition of new blood vessel formation and stimulating the loss of existing vessels. Furthermore, as proposed by Jain (7), tumor vessels appear to be structurally and functionally abnormal and are tightly associated with stabilizing pericytes and less dependent than normal vessels on VEGF for survival. So, unlike the counterpart of embryogenically derived blood vessels, the vasculature of tumors is chaotic and poorly organized, with evidence of hyperpermeable, irregularly shaped, tortuous, dilated, and saccular blood vessels characterized by restricted blood flow (7). Vascular endothelial growth factor inhibition causes these immature blood vessels to be “normalized.” Thus, following anti-VEGF therapy, a higher percentage of pericyte-associated blood vessels in the tumor vasculature are able to function more efficiently, at least transiently. The associated improvement in blood flow within the tumor enhances the delivery of cytotoxic chemotherapy (8). Besides, antiangiogenic therapy results in the reduction of interstitial pressure and thereby facilitates tissue penetration of chemotherapy (9). Consequently, VEGF blockade with bevacizumab or another antiangiogenic agent may enhance the antitumor effect of chemotherapy (5, 6). Furthermore, levels of VEGF also affect response to endocrine therapy, regardless of steroid hormone receptor status.
In advanced BC, the presence of a high tumor VEGF level was found to be an independent marker predicting a poor response to first-line endocrine therapy (10) or to both first-line endocrine therapy (tamoxifen) and first-line chemotherapy (11).
Bevacizumab (Genentech Inc., South San Francisco, CA, USA) is a humanized monoclonal antibody against all VEGF-A isoforms, and it prevents the binding of VEGF to receptors on vascular endothelial cells, leading to the inhibition of angiogenesis and tumor growth. It has been shown to be effective in colorectal cancer, non-small-cell lung cancer, renal cell carcinoma, ovarian carcinoma, cervical cancer, and glioblastoma multiforme (12-13-14-15-16-17). However, to date, in BC, bevacizumab has generated more controversies than any other targeted therapy.
In February 2008, the US Food and Drug Administration (FDA), based on promising results of the Eastern Cooperative Oncology Group (ECOG) 2100 trial (18), granted accelerated approval to bevacizumab in combination with paclitaxel for the first-line treatment of HER2-negative MBC. However, on July 20, 2010, the Oncologic Drugs Advisory Committee of the FDA's Center for Drug Evaluation and Research voted against the combination of bevacizumab with chemotherapy for the first-line treatment of MBC (19). A definitive announcement by the FDA that revoked approval of bevacizumab for this indication followed (20).
Bevacizumab is still approved in several countries as first-line treatment of advanced BC. European regulatory authorities came to a different conclusion than the FDA, and bevacizumab not only continues to be an accepted option for MBC in Europe but has been approved by the European Medicines Agency's Committee for Medicinal Products for Human Use in an extended indication: MBC in combination with capecitabine as first-line therapy when a taxane/anthracycline combination cannot be used.
The difference in the regulatory status of bevacizumab in the United States and Europe has been the subject of debate, and the regulatory authorities reached different conclusions regarding its clinical benefit. This article reviews the available data on the use of bevacizumab in the first-line treatment of MBC and examines these findings in the context of the FDA's revocation of its approval in the United States.
Methods
A literature search using the PubMed database identified English-language reports of clinical trials of bevacizumab in the first-line treatment of BC. In addition, the proceedings of the San Antonio Breast Cancer Symposium, European Society of Medical Oncology, and American Society of Clinical Oncology annual meetings were searched for presented abstracts using the following terms: bevacizumab and breast cancer or chemotherapy or efficacy or safety or cost. Each study was reviewed and phase III trials in patients with locally recurrent BC or MBC who had never been treated for metastatic disease and who received bevacizumab in combination with standard chemotherapy or broad community-based population were evaluated (Fig. 1).
Selection process for clinical trials of bevacizumab in first-line HER2-negative metastatic breast cancer included in the analysis.
Results
Three phase III trials tested the addition of bevacizumab to chemotherapy in the first-line setting (Tab. I).
Phase III studies of bevacizumab plus chemotherapy as first-line therapy for advanced breast cancer
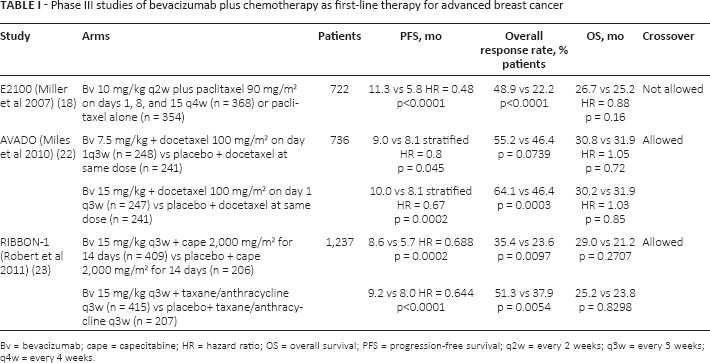
Bv = bevacizumab; cape = capecitabine; HR = hazard ratio; OS = overall survival; PFS = progression-free survival; q2w = every 2 weeks; q3w = every 3 weeks; q4w = every 4 weeks.
ECOG 2100
The ECOG 2100 trial compared the efficacy and safety of weekly paclitaxel plus bevacizumab with paclitaxel alone in 722 patients with MBC (18). The primary endpoint of the study was PFS. Patients were randomly assigned to receive weekly paclitaxel (90 mg/m2) on days 1, 8, and 15 every 28 days either alone or in combination with bevacizumab (10 mg/kg) on days 1 and 15. Patients continued therapy in the absence of disease progression or prohibitive toxicity. Median PFS was 11.8 months for the combination of paclitaxel/bevacizumab versus 5.9 months for paclitaxel alone (hazard ratio [HR] 0.6; p<0.001). Overall, adding bevacizumab to paclitaxel significantly improved the objective response rate (ORR) (36.9% vs 21.2%; p<0.001). Combined therapy increased the 1-year survival rate (81.2% vs 73.4%; p = 0.01); however, the median overall survival (OS) rate (26.7 vs 25.2 months, HR 0.88; p = 0.16) was similar between the 2 groups. These results were also validated by an independent review (21). The combination of bevacizumab with weekly paclitaxel proved a tolerable regimen, with similar low rates of hematologic, gastrointestinal, and musculoskeletal adverse events in each group. Rates of grade 3 or 4 neuropathy (23.6% vs 17.6%; p = 0.03), infection (9.3% vs 2.9%; p<0.001), and fatigue (8.5% vs 4.9%; p = 0.04) were higher in the combination group. Grade 3-4 hypertension was more common in patients receiving bevacizumab (14.8% vs 0%; p<0.001). Proteinuria was also more common in the experimental arm, but was rarely clinically significant (grade 3 proteinuria 2.7% vs 0%, respectively; p<0.001). No differences in quality of life were observed between the 2 groups. Interestingly, an exploratory analysis of the ECOG 2100 data showed that patients who had received prior taxane-based adjuvant therapy also benefitted from the addition of bevacizumab (18).
AVADO
The AVADO (Avastin and docetaxel) trial, a randomized, double-blind, placebo-controlled trial, investigated the efficacy of docetaxel alone or with bevacizumab at a dose of 7.5 or 15 mg/kg as first-line therapy in patients with HER2-negative MBC (22). The primary endpoint was PFS. This study, however, was not designed to detect a statistically significant difference between bevacizumab doses. Patients (n = 736) were randomized to either docetaxel (100 mg/m2) every 21 days with bevacizumab at a dose of 7.5 mg/kg or 15 mg/kg or docetaxel with placebo, for a maximum of 9 cycles of docetaxel. Earlier discontinuation was allowed for drug intolerability. Two dose reductions, to 75 mg/m2 and/or 60 mg/m2, were also allowed. Continuation on bevacizumab or placebo as maintenance therapy until disease progression was permitted following discontinuation of docetaxel. The majority of patients (75%) had received prior anthracycline- and/or taxane-based adjuvant chemotherapy. After progression, all patients were given the option to receive bevacizumab in combination with second-line regimens. The association of bevacizumab with docetaxel showed superior PFS compared to placebo plus docetaxel, at both 7.5 mg/kg (9.0 vs 8.1 months, stratified HR 0.8; p = 0.045) and 15 mg/kg (10.0 vs 8.1 months, stratified HR 0.67; p<0.001). Both doses of bevacizumab were also associated with significantly higher ORRs (7.5 mg/kg: 55.2% vs 46.4%; p = 0.07; 15 mg/kg: 64.1% vs 46.4%; p<0.001). However, as in the ECOG 2100 study, the rate of OS was similar across all 3 arms, with median values of 31 months (HR 1.05; p = 0.72 for bevacizumab at 7.5 mg/kg; HR 1.03; p = 0.85 for bevacizumab at 15 mg/kg). The addition of bevacizumab to docetaxel had limited impact on the known toxicity profile of docetaxel.
RIBBON-1
The Regimens in Bevacizumab for Breast Oncology (RIBBON)-1 trial was an international, multicenter, phase III, placebo-controlled trial. Patients (n = 1,237) were randomly assigned 2:1 to receive either chemotherapy (capecitabine, taxane-based, or anthracycline-based chemotherapy) plus bevacizumab or chemotherapy plus placebo, as first-line treatment for HER2-negative MBC (23). The primary endpoint was PFS. Secondary endpoints included ORR, OS, and 1-year survival rate. The combination of bevacizumab and chemotherapy as first-line therapy resulted in a statistically significant improvement in PFS. In the taxane or anthracycline cohort, median PFS resulted in 9.2 versus 8.0 months (HR 0.64; 95% confidence interval [CI] 0.52-0.80; p<0.001). Similarly, capecitabine plus bevacizumab significantly improved PFS compared with capecitabine alone (8.6 vs 5.7 months; HR 0.69; CI 0.56-0.84; p<0.001). However, there was no statistically significant improvement in OS or the 1-year survival rate between treatment arms for either cohort. For the cohort of patients who received capecitabine, the estimated HR for OS was 0.85 (95% CI 0.63-1.14; p = 0.27), and there was a trend for improved 1-year survival in the bevacizumab-containing arm (81.0% vs 74.4%; p = 0.076). For patients who received anthracyclines or taxanes (with or without bevacizumab), the stratified HR for OS was 1.03 (95% CI 0.77-1.38; log-rank p = 0.83), and the 1-year survival rate was 83.2% versus 80.7% (p = 0.44).
Meta-analyses show improved PFS but not OS
A meta-analysis performed by Miles et al (24) (n = 2,447) determined that bevacizumab increased PFS compared with non-bevacizumab therapy (HR 0.64, 95% CI 0.57-0.71; median PFS 9.2 months vs 6.7 months) and also improved the ORR (49% vs 32%). However, there was no significant difference in OS between the bevacizumab and non-bevacizumab cohorts (HR 0.97, 95% CI 0.86-1.08; median OS 26.7 vs 26.4 months). One-year survival rate was significantly increased in the bevacizumab arms (77% vs 82%; p = 0.003). Furthermore, bevacizumab in poor-prognosis patients with triple-negative MBC prolonged PFS (HR 0.63; 95% CI 0.52-0.76) without statistically significant improvement in OS (HR 0.96; 95% CI 0.79-1.16). The median PFS was 8.1 months with bevacizumab versus 5.4 months with chemotherapy alone, while the median OS was 18.9 versus 17.5 months, and the 1-year OS rates were 71% versus 65%.
These results were similar to those of previously published meta-analyses that evaluated the efficacy of bevacizumab in combination with chemotherapy for the first-line treatment of MBC. A meta-analysis by Valachis et al (25) pooled data from the 3 randomized studies E2100, AVADO, and RIBBON-1, a phase II study, and a trial with bevacizumab after first-line chemotherapy for MBC, and found that the addition of bevacizumab first-line MBC significantly improved PFS, with a global HR of 0.70 (95% CI 0.60-0.82; p<0.001). This benefit in PFS was seen across different subgroups and independently from sites of metastases, hormone receptor status, or prior adjuvant taxane treatment. The addition of bevacizumab to chemotherapy was also related to a significant improvement in ORR (p<0.001). However, no significant increases in OS were observed in the bevacizumab-containing arms, with a global HR of 0.90 (95% CI 0.80-1.03; p = 0.119).
Lee et al (26) evaluated 4 studies involving a total of 2,860 patients and reported improvement in PFS for patients treated in bevacizumab-containing arms (pooled HR 0.69, 95% CI 0.58-0.81), but not OS (HR 0.92, 95% CI 0.82-1.03) or ORR (HR 1.53, 95% CI 1.37-1.71).
Rossari et al (27) performed a meta-analysis on the E2100, AVADO, and RIBBON-1 studies. They found that adding bevacizumab to chemotherapy in the first-line treatment of MBC significantly improved PFS, with an overall HR of 0.70 (95% CI 0.57-0.86; p = 0.0006). The PFS was assessed according to hormone receptor status (positive or negative), prior adjuvant chemotherapy (yes or no), age (<65 years vs ≥65 years), prior use of taxanes (yes or no), and disease-free interval (DFI) (short vs long). It was noted that the analysis by DFI was different among the trials, with the AVADO and E2100 trials stratifying DFI by ≤24 months versus >24 months, whereas RIBBON-1 stratified by ≤12 months versus >12 months. However, adding bevacizumab to chemotherapy provided a consistent benefit in PFS in all the patient subgroups analyzed. Nevertheless, none of the individual studies showed a significant OS benefit of combining bevacizumab with chemotherapy and, globally, no statistically significant OS benefit (HR 0.95, 95% CI 0.85 to 1.06; p = 0.65) was reported.
ATHENA real-world study
ATHENA was an open-label, single-arm, international study conducted in a real-world oncology practice setting and included a total of 2,251 patients with MBC who received first-line therapy with bevacizumab plus taxane monotherapy or combination chemotherapy. ATHENA was the largest study of patients with MBC treated with bevacizumab-containing chemotherapy regimens to be carried out in a broad community-based population (28) more closely representative of routine clinical oncology practice. The primary objective was safety. Secondary endpoints were time to progression (TTP) and OS.
Patients in this study received, at the discretion of treating oncologists, bevacizumab 10 mg/kg every 2 weeks or 15 mg/kg every 3 weeks plus taxane-based chemotherapy or other non-anthracycline regimen until disease progression, unacceptable toxicity, or patient withdrawal (28). The majority of patients had primarily estrogen receptor-positive BC (66.1%) and more than 3 metastatic sites (64%). Chemotherapeutic agents more frequently combined with bevacizumab were single-agent paclitaxel (35%) or docetaxel (33%). The paclitaxel administration was weekly in 17% of cases, versus 13% every 3 weeks or other schedule in 6%. A further 10% of patients received taxane-based combination regimens, commonly with carboplatin and gemcitabine. The non-taxane chemotherapies most frequently utilized were capecitabine (5%) and vinorelbine (3%). The median treatment duration of treatment was 6.2 months for bevacizumab (range 0.0-29.7) and 4.2 months for chemotherapy (range 0.0-29.5).
Safety and efficacy data were consistent with the findings of the previous reported randomized clinical trials of chemotherapy evaluating bevacizumab in combination with first-line taxane-based regimens for locally recurrent BC or MBC. First-line bevacizumab in combination with taxane-based chemotherapy was well-tolerated, with a low incidence of severe adverse events (SAEs) or specific adverse events (AEs) in a broad population of BC patients (28). The most frequent SAEs were febrile neutropenia (5.1%), neutropenia (3.6%), and pyrexia (1.5%). The incidence of grade 3-4 hypertension was 4.4%, lower than that reported in E2100 (16%), but similar to AVADO (4.5%). Furthermore, other bevacizumab-related SAEs of grade 3 or higher were uncommon and consisted of arterial/venous thromboembolism (3.3%), proteinuria (1.7%), gastrointestinal perforation (0.26%), and congestive heart failure (0.44%). The overall median TTP was 9.5 months (95% CI 9.1-9.9). The ORR in the intent-to-treat (ITT) population was 52% and a further 33% of patients achieved disease stabilization as their best response.
Final survival data from the ATHENA study after a median follow-up duration of 20.1 months showed that OS was 25.2 months in patients receiving bevacizumab in combination with standard first-line chemotherapy (29). Furthermore, for patients who continued bevacizumab monotherapy after stopping chemotherapy, median TTP was 11.6 months and median OS 30.0 months (29).
Interestingly, a subanalysis in the elderly subpopulation of the ATHENA study was published, comprising 175 patients aged ≥70 years (30). Approximately half of these patients (46%) received a paclitaxel regimen in combination with bevacizumab. The combination was well-tolerated by elderly patients. The median TTP in this subpopulation was 10.4 months (95% CI 8.8-11.8 months), compared with 9.5 months (95% CI 9.1-9.9) in the overall ITT population. The ORR was 42% for the elderly patients, compared with 52% in the overall ITT population. Final survival data in this patient group showed a median OS of 20.4 months. Moreover, the overall incidence of bevacizumab-related SAEs was similar to that reported in younger patients (<70 years). The only grade ≥3 SAEs more commonly reported in older versus younger patients were hypertension (6.9% vs 4.2%) and proteinuria (4.0% vs 1.5%). The incidences of grade 3 or higher arterial/venous thromboembolic events (2.9%vs 3.3%), CNS hemorrhage (<0.1%), and cardiac events (0.6 vs 0.4%) were similar to younger patients.
Efficacy of bevacizumab plus paclitaxel
A recent study confirmed that the addition of bevacizumab to a paclitaxel-based regimen was an active and well-tolerated first-line treatment for MBC (31). In 220 patients with previously untreated MBC, the combination of bevacizumab plus paclitaxel resulted in a median PFS of 9.3 months (95% CI 7.8-10.8) and a 1-year survival rate of 68%. This regimen also presented notable activity in patients with triple-negative BC (TNBC; n = 106) with a median PFS of 8.3 months (95% CI 7.8-8.8). Severe AEs were consistent with the established safety profile of bevacizumab plus paclitaxel.
Discussion
The combination of bevacizumab with taxane-based regimens has been associated with benefits in ORR and PFS in the first-line treatment of patients with locally recurrent BC or MBC (32). However, to date, no significant OS advantage has been observed in randomized clinical trials. The RIBBON-2 study showed a trend towards a benefit in OS with bevacizumab, even though the benefit in OS was not statistically significant (33). The open-label, single-arm, international, real-world ATHENA trial also reported a notable OS in patients receiving bevacizumab in combination with standard first-line chemotherapy, also including elderly population, and, above all, for patients who received maintenance with bevacizumab monotherapy after stopping chemotherapy (29). The RIBBON-2 trial only included pretreated patients, and ATHENA was in patients not previously treated for metastatic disease. Although the benefit was persistent in the TNBC and elderly subgroups, no clear benefit on OS was demonstrated (30, 34). Furthermore, the single-arm ATHENA trial does not allow us to define the impact of the addition of bevacizumab to chemotherapy. Nevertheless, its large sample size (n = 2,251) could provide valuable information on outcomes in clinically important subgroups, including patients with TNBC and the elderly. The results of a multicenter national French observational study, based on a large-scale real-life setting database, the Epidemiological Strategy and Medical Economics program, were presented at the recent American Society of Clinical Oncology annual meeting (35). Significantly better OS was reported in the 2,127 patients with HER2-negative MBC who received paclitaxel plus bevacizumab, compared with the 1,299 patients receiving paclitaxel alone (HR 0.672 [95% CI 0.601, 0.752]; median survival time, 27.7 vs 19.8 months). Also, PFS was better for patients receiving paclitaxel plus bevacizumab versus paclitaxel (HR 0.739 [0.672, 0.813]; 8.1 vs 6.4 months). All data were consistent across all supportive and sensitivity analyses, as well as in triple-negative and estrogen receptor-positive subgroups. Concerning this, it is worth noting that a robust methodology was applied. However, as the same authors concluded, despite the robust methodology, real-world data should be interpreted with caution.
To be thorough, no benefits were reported in patients with earlier stage BC with the addition of bevacizumab in the adjuvant setting (36, 37). However, this is not a subject of this review.
In MBC, due to the lack of a clear survival benefit associated with bevacizumab, the FDA recommended against bevacizumab in 2010 (19, 20). The FDA revocation resulted from concerns about safety, considering that bevacizumab did not improve OS in different randomized studies.
One of the major problems has been its lack of an OS benefit. However, there is no unanimous opinion on the appropriateness of OS as a sole primary endpoint in first-line MBC clinical trials, which has been the subject of lively debate. Interestingly, Cortés et al (38) reported that, even though from a regulatory standpoint gain in OS is the gold standard for drug approval, PFS could be a valid surrogate endpoint in some diseases and in specific treatment settings.
The value of PFS in the context of MBC remains controversial. However, PFS has several important advantages in MBC trials. To date, patients with MBC usually receive multiple lines of subsequent therapy (Tab. II), and PFS/TTP should be considered true endpoints since they are not affected by subsequent lines of chemotherapy. In MBC, a strikingly small proportion of all trials (12%), and even fewer first-line trials (4.8%), demonstrated OS gain. According to the model proposed by Broglio and Berry (39) on post-progression survival (PPS), OS would be an appropriate primary endpoint when there is a short median PPS interval. On the other hand, when the PPS is >12 months, subsequent lines of chemotherapy would dilute the effect of the study medication on OS. In MBC, the ability of first-line phase III trials to demonstrate a treatment effect upon OS depends on the duration of PPS (40, 41). Indeed, OS may be a more appropriate endpoint for second- or third-line trials, when PPS is shorter. This is illustrated by the recent EMBRACE trial in heavily pretreated patients with anthracycline- and taxane-refractory MBC who were randomized to monotherapy with the microtubule inhibitor eribulin or treatment of physician's choice. Eribulin significantly improved OS (median 13.1 months, 95% CI 11.8-14.3) compared with the real-life standard treatment choices (10.6 months, 95% CI 9.3-12.5; HR 0.81, 95% CI 0.66-0.99; p = 0.041) (42).
Data were not available from the E2100 study.
Moreover, these clinical trials did not collect data on post-progression therapies. Without details on subsequent treatments, it would be impossible to determine the impact of different post-trial therapies patients received in the bevacizumab and non-bevacizumab arms. Furthermore, the lack of statistically significant OS benefit could be due to the cross-over to bevacizumab allowed to patients in non-bevacizumab arms at the time of progression (22, 23).
Several issues arise over the FDA's removal of the BC indication for bevacizumab. If this decision was based on a lack of OS benefit, it is unclear why the indications for bevacizumab in other cancers remain, particularly as a clear OS survival benefit has not been established for the majority of cytotoxic chemotherapy combinations in current use in MBC. Even the most well-established cytotoxic regimens show at best only marginally significant OS benefits (43, 44), which makes the FDA's decision appear at least controversial, if not arbitrary.
The FDA cited primarily safety-related concerns for its reasons to remove the MBC indication for bevacizumab (45). These toxicities include hypertension, proteinuria, thrombosis, and hemorrhage (46). However, as previously reported in available MBC phase III safety data (AVF2119g, E2100, AVADO, RIBBON-1, and RIBBON-2), bevacizumab generally appeared to be well-tolerated (18, 22, 23, 33, 47). Typically, SAEs occurring during treatment with bevacizumab are predictable, mild to moderate in severity, and/or manageable with standard medication, and usually do not necessitate discontinuation of therapy. In general, bevacizumab has been shown to have a favorable tolerability profile when used in various combination therapy approaches, including in special populations such as the elderly and patients with poor performance status (48). However, AEs such as arterial thrombosis and cardiac toxicity, although uncommon with bevacizumab, are more serious and require appropriate clinical awareness and prudent monitoring.
Regarding cost, bevacizumab seems expensive and not cost-effective as a first-line treatment for MBC. The cost-effectiveness of bevacizumab in combination with paclitaxel-based chemotherapy has been assessed using a decision-analytical model incorporating efficacy and adverse events data from the ECOG 2100 trial (49-50-51). This is not only related to bevacizumab of course, but it is common for almost all new drugs now employed in the treatment of cancer (breast or other). Most of the combinations with new agents recently approved for the treatment of cancer did not demonstrate a clear advantage in OS, including expensive combinations of anti-HER2 agents with endocrine therapy for MBC (52, 53) or the most expensive drugs employed in other cancer types, for example trabectedin in ovarian cancer (54, 55). Nevertheless, physicians who have access to expensive interventions have a responsibility to consider the cost-effectiveness of cancer drugs as part of their role as overseers of finite health care resources (56, 57). Taking into consideration very limited healthcare budgets, alternative solutions such as risk-sharing schemes should be considered. To this end, risk-sharing schemes for pharmaceuticals can be regarded as agreements between payers and pharmaceutical companies with the aim of reducing the impact of new and existing medicines on the payer's budget where there is uncertainty regarding the value of the medicine and/or the need to work within finite budgets.
Strategies to accelerate reimbursement and improve access for newer and potentially innovative medicines, particularly where limited data are available at the time of marketing approval, have recently been initiated by the Italian Medicines Agency (AIFA), using 2 different approaches: variations on patient access schemes and performance-based/outcome contracts (where the pharmaceutical company reimburses the cost if the medicine is shown to be ineffective and the National Health Service pays for effective medicines) (58-59-60). The AIFA Oncologic Working Group proposed 2 risk-sharing arrangements for new anticancer drugs to enhance their reimbursement potential according to the following criteria: “epidemiological data for the disease; the possibility of clearly defining a subset of the population responsive to the treatment; and results from clinical trials” (60). Bevacizumab—for metastatic carcinoma of the colon or rectum, BC, non-small-cell lung cancer, and advanced and/or metastatic renal cell cancer—was included in this scheme.
Conclusion
The addition of bevacizumab to chemotherapy has produced substantial improvements in ORR and PFS, compared with chemotherapy alone, in advanced HER2-negative BC. However, no significant OS improvement has been observed so far, and further follow-up is needed. Still to be answered from currently available and emerging data is whether prolongation of PFS is in itself enough to consider bevacizumab efficacious in MBC. Moreover, it should be noted that bevacizumab was well-tolerated in studies in patients with MBC, and AEs were fairly manageable, with remarkably similar frequencies of bevacizumab-related AEs when combined with a number of different anticancer drugs. Patients with TNBC seem particularly well-suited to bevacizumab treatment. Finally, the cost of treatment with bevacizumab is consistent with the costs of other generally accepted new anticancer treatments, and should therefore not be considered a deterrent to its use in MBC.
To date, there is no role for bevacizumab for the treatment of early-stage BC. On the other hand, bevacizumab, based on the available clinical trials results, is a part of the complex therapeutic context of MBC. The identification of biomarkers would be useful to optimize its use in MBC.
Footnotes
Acknowledgment
The authors thank Health Publishing & Services, Milan, Italy, for editing assistance.
Financial support: No financial support was received for this submission.
Conflict of interest: P. Marchetti has received grants from AstraZeneca, Novartis, Roche, Bristol Myers Squibb, and GlaxoSmithKline. None of the other authors has conflict of interest with this submission.

