Abstract
Edema is a common complication of numerous renal disease. In the recent past several aspects of the pathophysiology of this condition have been elucidated. We herein present a case of nephrotic syndrome in a 30 year-old men. The discussion revolves around the following key questions on fluid accumulation in renal disease:
What is edema? What diseases can cause edema? What are the mechanisms of edema in nephrotic syndrome?
The “underfill” theory The “overfill” theory Tubulointerstitial inflammation Vascular permeability What are the mechanisms of edema in nephritic syndrome? How can the volume status be assessed in patients with nephrotic syndrome? What are therapeutic strategies for edema management? What are the factors affecting response to diuretics? How can we overcome the diuretics resistance?
Effective doses of loop diuretics Combined diuretic therapy Intravenous administration of diuretics Albumin infusions Alternative methods of edema management Conclusion.
Case presentation
A 30-year-old male patient was referred to the nephrology clinic with massive periorbital, upper and lower extremities and scrotal edema, 10 kg weight gain, foamy urine and reduction of the urine output. His edema was resistant to oral furosemid administration (80 mg/day). His blood pressure was 110/70 mmHg. The past medical history was unremarkable. One month before admission minimal proteinuria was found.
Physical examination did not reveal any significant signs and symptoms beyond edema.
Urinalysis demonstrated nephrotic range proteinuria (10 g/day) without any abnormalities of the urinary sediment. Total blood count showed hemoglobin 12.5 g/dL, an erythrocyte sedimentation rate of 40 mm/h, hematocrit 54%. The main biochemical data are summarized in Table I.
The biochemical data of the patient with nephrotic syndrome
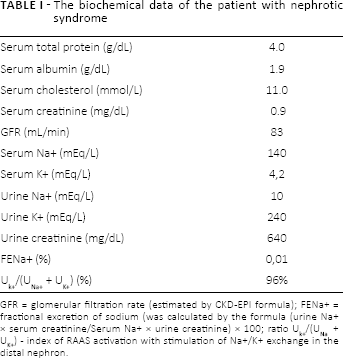
GFR = glomerular filtration rate (estimated by CKD-EPI formula); FENa+ = fractional excretion of sodium (was calculated by the formula (urine Na+ × serum creatinine/Serum Na+ × urine creatinine) × 100; ratio Uk+/(UNa + UK+) - index of RAAS activation with stimulation of Na+/K+ exchange in the distal nephron.
No monoclonal component was found either in the serum or the urine. Thromboembolic events were not observed. Ultrasonography revealed two normal-sized kidneys and presence of ascites. Chest plain radiography revealed bilateral hydrothorax.
Limitation of the oral sodium intake to 2.5 g daily was recommended. The intravascular volume was corrected by intravenous infusions of 20% albumin (2 mL/min for 1–1.5 hours). Intravenous infusions of furosemide (initial bolus −60 mg, followed by continuous infusions - 60 mg/h) were started. The restriction of sodium intake and the diuretic strategy resulted in a positive natriuresis with moderate reduction of edema and a body-weight loss of 900–1000 mg/day.
A kidney biopsy was performed and on light microscopy normal glomeruli were found. Immunofluorescent microscopy did not reveal any immune complex deposits. Electron microscopy confirmed the absence of electron-dense deposits and demonstrated hypertrophy and vacuolization of some podocytes and foot process effacement. Based on these findings minimal change disease was diagnosed.
The patient was started with oral prednisone 60 mg/day (1 mg/kg), while the diuretic therapy was discontinued. A urinary output of 3 L/day was observed in a week after the initiation of steroids. Two weeks later remission of NS was achieved. The full dose of prednisone was continued for the next two weeks. After achieving a complete remission, it was tapered slowly over 6 months.
What is edema? What diseases can cause edema?
Edema is defined as an abnormal accumulation of fluid in the interstitial space of the body.
Edema is the reason for visiting the doctor, and for a careful differential diagnosis. Various diseases (renal, cardiac, hepatic, thyroid glands and others) may be causative (summarized in Tab. II).
List of the diseases, presenting with edema

ADH = antidiuretic hormone.
The leading causes of renal edema are nephrotic and nephritic syndromes (Tab. II).
Confirmation of the renal nature of the edema requires a precise diagnosis of the renal disease with an assessment of its clinical and morphological activity. All this information is important for defining the treatment strategy, including the administration of diuretics.
What are the mechanisms of edema in nephrotic syndrome?
Edema is one of the cardinal clinical features of nephrotic syndrome (NS), and may range from localized puffiness to massive anasarca.
The pathophysiological mechanisms of edema formation in NS were discussed over several decades. Abnormal accumulation of interstitial fluid results from the combination of:
abnormal renal sodium retention (primary and secondary)
increased capillary wall permeability (related to the release of vascular permeability factor and other cytokines).
Given the wide spectrum of renal diseases leading to the NS, more than one single mechanism may be responsible for the renal salt retention observed in these diverse conditions (1–6).
The “underfill” theory
According to classic theory, also called the “underfill” hypothesis, sodium retention in NS is secondary to hypovolemia. Urinary protein losses lead to the decrease of plasma albumin concentration and lowering of the plasma oncotic pressure resulting in imbalance of the Starling forces (Fig. 1). These events lead to fluid redistribution from the intravascular space towards the interstitial space, causing the decrease in effective arterial blood volume. The plasma volume contraction triggers adaptive neurohumoral responses through activation of the sympathetic nervous system (particularly efferent renal nerves), stimulation of the renin-angiotensin-aldosterone system (RAAS), an increase of antidiuretic hormone (ADH) release, renal sodium and water retention. The decrease in atrial natriuretic peptide (ANP) secretion facilitates salt retention and subsequent edema formation (Fig. 1).
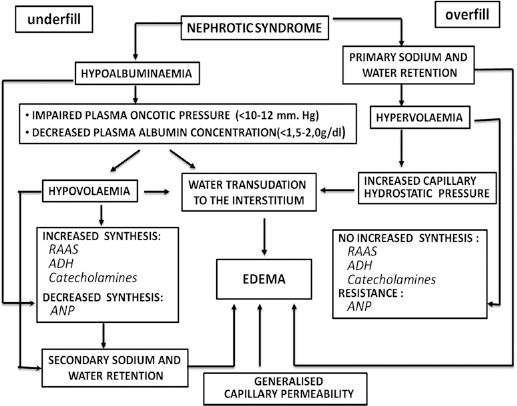
Pathophysiological mechanism of edema formation in nephrotic syndrome.
However, there are many arguments against this theory.
Clinical observations indicate that most of the edematous patients with the different histological forms of NS have either a normal or even expanded intravascular volume, whereas only a minority of nephrotic patients have a depleted intravascular volume (7, 8). So, some patients with early phases of severe NS (usually in rapidly developing relapse of minimal changes disease (9–11)) demonstrate a temporary hypovolemia, because the mobilization of interstitial albumin to the blood is not sufficiently fast to prevent it, there is disequilibrium between plasma and extravascular albumin stores. Later, when the albumin redistribution becomes complete, a new equilibrium is established, during which blood volume is maintained.
There is increasing experimental and clinical evidence that hypoalbuminemia cannot explain the development of edema in NS (for reviews see (1, 2)). In particular, it has been shown that young analbuminemic Nagase rats do not develop edema despite low plasma oncotic pressure (12). This phenomenon has also been demonstrated in humans with NS (13,14).
Furthermore, decrease in plasma oncotic pressure does not affect the volume of the intravascular space in NS (15, 16), and intravenous administration of albumin to induce volume expansion promotes only mild natriuresis (12).
These data suggest that there might have been sodium retention in patients with NS, not mediated by volume depletion.
The “overfill” theory
Unlike the “underfill” hypothesis, the “overfill” concept presumes a primary intrinsic defect of salt and water excretion in the nephrotic kidney (1, 2). This theory postulates that there are also consequences of the proteinuria other than just hypoalbuminemia, which lead to enhanced tubular sodium reabsorption, progressive extracellular fluid volume expansion and edema formation by an overflow mechanism (Fig. 1).
The cellular mechanisms of this primary sodium retention in NS are associated with:
activation of the expression of Na+/H+exchanger3 (NHE3) in the apical membrane of the proximal tubules by the tubular albumin (17–19)
proteinuria-induced stimulation of the basolateral Na, K-ATPase pump in the collecting ducts (20–22)
activation of the apical epithelial amiloride-sensitive sodium channels (ENa+C) in the collecting ducts induced by urine proteases (plasminogen/plasmin) (2, 23–26)
tubular resistance to ANP (the result of an increased cyclic guanosine monophosphate (cGMP) phosphodiesterase activity and depletion of cGMP, which is the ANP second messenger) (2, 27–29).
Tubulointerstitial inflammation
There is also strong evidence that tubulointerstitial inflammation and glomerulo-interstitial cross-talks are involved in sodium retention and nephrotic edema formation (30). It has been demonstrated that proteinuria induces infiltration of the interstitium by inflammatory cells (T-cells and monocyte/macrophages). Production of vasoactive mediators by these cells (increase of angiotensin II and decrease of nitric oxide) can cause a reduction in glomerular ultrafiltration coefficient and single nephron GFR (30, 31) as well as a decline in sodium filtration with an increase of tubular reabsorption, resulting in primary sodium retention (Fig. 2). Interstitial inflammation in longstanding NS, stimulating tubulointerstitial fibrosis and atubular glomeruli formation, can lead to a more permanent decrease in glomerular filtration rate (GFR) and thus to sodium retention.
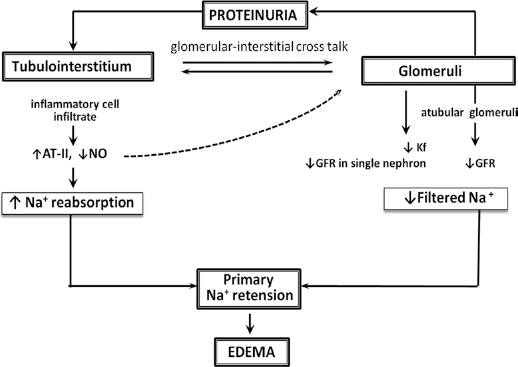
The role of tubulointerstitial inflammation in the pathogenesis of nephrotic edema. Adapted from Rodriguez-Iturbe et al (30). Kf = ultrafiltration coefficient, GFR = glomerular filtration rate, AT-II = angiotensin II, NO = nitric oxide.
Vascular permeability
The asymmetric expansion of the interstitial volume in NS is explained by changes in intrinsic properties of the endothelial capillary barriers, including its increased hydraulic conductivity and permeability to proteins, rather than to an imbalance of the Starling forces (2, 32–34). Increase of capillary permeability could be related to the release of numerous vascular permeability factors and other cytokines, from immune cells, including histamine, endotoxins, anaphylatoxins, catecholamines, vascular endothelial growth factor, interleukin-1, interleukin-2, and tumor necrosis factor-alpha.
What are the mechanisms of edema in nephritic syndrome?
Acute glomerulonephritis is the prototypical form of nephritic edema (35). Overexpansion of the vascular compartment is responsible for the hemodynamic characteristics of patients with nephritic syndrome (increased effective arterial blood volume, plasma volume, blood pressure, and cardiac output) (36). Hypervolemia and edema in such patients result from primary salt retention and GFR reduction due to glomerulopathy. The glomerular damage leads to urinary protein loss with following increase of tubular albumin reabsorption and urine proteases concentration, which activates renal salt and water retention. The glomerular lesion results in the reduction of Kf, which is driven by the effective capillary wall hydraulic permeability and total capillary surface area per glomerulus (37). At the onset of the disease the GFR remains normal for some period of time. The initially preserved GFR is maintained by the equilibrium between the increased transcapillary hydraulic pressure, which is the driving force for filtration, and the reduced Kf. The progressive GFR deterioration observed in more advanced stages of this disorder is the consequence of the further reduction of Kf. The significant fall of Kf leading to subsequent reduction of GFR results in the decrease of sodium filtration with sodium and water retention.
How can the volume status be assessed in patients with nephrotic syndrome?
Evaluation of the volume status is crucially important for edema management. Table III summarizes the typical clinical and laboratory features for distinguishing nephrotic patients with underfill or overfill physiology. Clinical characteristics of hypovolemia include low blood pressure, postural hypotension, tachycardia, signs of peripheral vasospasm (pallor, cool extremities), abdominal pain secondary to decreased intestinal perfusion. Diarrhea and vomiting, provoking dehydration and hypovolemia, demand attention from the doctor. By contrast, in hypervolemic NS, high blood pressure without postural hypotension and other signs of volume depletion are found.
Evaluation of the predominant volemic status in patients with nephrotic syndrome

Not available for routine practice and quick diagnosis.
Influenced by therapy with diuretic.
GFR = glomerular filtration rate; FENat = fractional excretion of sodium.
Urinary sodium and potassium concentration, fractional excretion of sodium (FENa+) and index of RAAS axis activation may be useful tests for evaluation of renal sodium handling and volume status in nephrotic patients.
FENa+ is the ratio of sodium excreted to that filtered by the renal tubules.
FENa+(%) = (urinary Na+ × serum creatinine/serum Na+ × urinary creatinine) × 100.
A urinary sodium concentration below 20 mEq/L and FENa+ below 1% indicate sodium retaining conditions. The blockade of sodium reabsorbtion by diuretic increases urinary sodium concentration and fractional sodium excretion to a greater or lesser extent (38). Loop diuretics can increase FENa to 30%, thiazides to 5%-10% and potassium sparing diuretics to 2%-3% (39).
Vande Walle and coworkers revealed significant positive correlations between the ratio of urinary concentrations of K+ to the sum of K+ and Na+ [K+(mEq/L)/K+ (mEq/L) + Na+(mEq/L)], reflecting of Na+/K+ exchange in the distal nephron, and increased serum concentrations of renin, aldosterone and ADH, which are observed in hypovolemic children with minimal lesion nephrotic syndrome (8–10). The ratio has been offered as an index of activation of RAAS axis. A ratio K+/K++Na+ more than 60% suggests activation of the axis and renal potassium wasting (8–10).
Patients with NS and hypovolemia typically show low FENa+ (often below 0.2%) and a high urinary K+/K++Na+ index (greater than 60%) (8, 39–43). Management of edema in such patients demands correction of the intravascular volume with infusion of albumin ore non-protein colloids before initiation of diuretic therapy. Active usage of diuretics without adequate volume correction poses a risk to hypovolemic shock. In hypovolemic patients with relapse of steroid responsive NS treatment with corticosteroids leads to rapid decrease of proteinuria, steroid-induced diuresis within 7–10 days. It may be better to avoid giving furosemide to these patients if possible and wait for the diuretic effects of remission due to corticosteroid treatment.
Patients with edema and clinical and/or laboratory features of hypervolemia (FENa+>1% and urinary K+/K++Na+ index <60%) can safely be treated with diuretics (8, 39–43). Intravascular infusion of albumin and colloids in such patients put them at risk of volume overload and lung edema.
It should be noted that the prior therapy with diuretic may limit the practical utility of these urinary tests for distinguishing patients with underfill or overfill physiology. Therefore, close monitoring of both clinical and laboratory features (Tab. III), are required for evaluation of predominant mechanism of nephrotic edema formation and therapeutic decision.
What are therapeutic strategies for edema management?
Treatment of renal edema is directed to the limitation of further sodium retention, and to the enhancement of the sodium and water excretion, sequestered in the interstitial compartment. Diuretics are fundamental for edema treatment in NS; this type of medication ensures diuresis by inhibiting of sodium reabsorption in the different segments of the renal tubular system (2, 39).
The efficacy of diuretics is determined by site of their action in the nephron. Major proportion of filtered Na+ is reabsorbed in the proximal tubule (55%–60%) and in the loop of Henle (25%–30%); only 5%–7% of filtered sodium is reabsorbed in the distal segments of the renal tubular system. However, in NS the efficacy of diuretics acting in proximal segments is hampered by compensatory increase of sodium and water by distal reabsorption in the ascending limb of Henle's loop. Therefore, loop diuretics, which can increase FENa+ up to 30%, remain the first choice for edema treatment in patients with NS. Nevertheless, the treatment of edematous patients should be individualized according to their clinical manifestations.
The general principles of edema management are as follows:
Sodium intake restriction to 100 mEq Na+/day should be recommended to all patients with edema (1 mEq Na+ = 23 mg Na+ = 58.3 mg of NaCl)
Strict limiting sodium intake (to 1 mEq/kg/day) is necessary only in subjects with refractory edema
Diuretics are not required for minimal periorbital puffiness or feet edema. Most of such patients demonstrate reduction of edema with moderate restriction of sodium intake
Mild edema in patients with steroid responsive NS resolve under treatment with corticosteroids, which usually leads to steroid-induced diuresis within 7–10 days
Patients with moderate-to-severe edema, beyond sodium restriction, also need treatment with one or more diuretic agents
Evaluation of volume status is necessary for edema management
Albumin infusions with diuretic co-administration should be recommended to subjects with hypovolemia or refractory severe edema.
In the absence of emergency situations, the body-weight reduction under the diuretic treatment should be targeted to 1% of body weight.
Edema is considered refractory if therapy with maximum doses of diuretics results in the daily weight loss less than 1% of body weight.
The algorithm of edema management in patients with NS is shown in Figure 3.
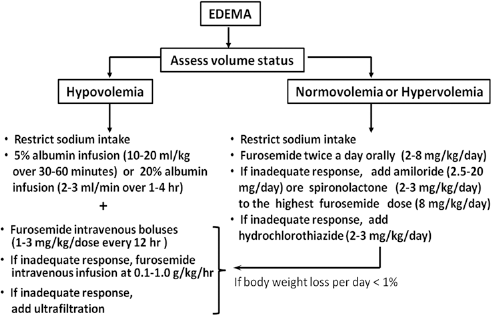
The algorithm for edema management in patients with nephrotic syndrome.
What are the factors affecting response to diuretics?
The efficacy of loop diuretics is restricted by the functional resistance of nephrotic patients to the natriuretic effect of these drugs (2, 39, 44, 45).
Several mechanisms may contribute to diuretic resistance in patients with NS, including:
Decreased gastro-intestinal drug absorption. (Bowel wall edema impairs absorption of oral medications)
Decreased diuretic entry into the tubular lumen. (Loop diuretics are highly bound to proteins, and significant hypoalbuminemia results in the reduced free drug secretion into the tubules)
Increased distal sodium reabsorption, so called rebound effect or braking phenomenon. (With long-term use of loop diuretics sodium, that originates from Henle's loop to the distal segments, causes hypertrophy of distal nephron cells with concomitant increase of the sodium reabsorption)
Use of non-steroidal anti-inflammatory drugs (NSAIDs) (Through blocking the synthesis of prostaglandins, NSAIDs can cause vasoconstriction, reduction of renal perfusion and sodium retention. In addition, NSAIDs can competitively inhibit the secretion of diuretic into proximal tubule, preventing their effects).
The binding of furosemide to albumin in the tubular fluid has been demonstrated in the experiment, but has not been confirmed in the human study.
How can we overcome the diuretics resistance?
Effective doses of loop diuretics
The diuretic effect relies on an adequate amount of the drug reaching its site of action in the renal tubules. Primarily, in patients with NS and edema it is necessary to increase the dose of loop diuretics to maximally effective oral dose (furosemide 480 mg/day, torasemide 50 mg/day, bumetanide 6 mg/day). Higher doses of oral diuretics do not exert any additional effect, while the frequency of side effects is greatly increased. Diuretics with a high bioavailability are preferred (50% for furosemide, 80% for torasemide, 90% for bumetanide).
Second, to overcome the sodium reabsorption in the distal tubules after the termination of the loop diuretic's action, they can be prescribed more than once per day. The daily dose may be divided into two parts, provided that each dose reaches an effective concentration.
Combined diuretic therapy
In patients with NS adding thiazides (hydrochlorothiazide 25–100 mg/day or metolazone 100 mg/day) on top of furosemide markedly increases the natriuresis (46).
First off all, thiazide diuretics have a longer half-life and prevent or reduce the sodium retention after termination of the loop diuretic's action, prolonging its natriuretic effect.
Second, administration of the thiazide may interfere with the so-called rebound effect, following loop diuretic treatment. Thiazide diuretics block the nephron sites, prone to sodium-induced hypertrophy, accounting for the synergistic response of the combination of thiazide and loop diuretics. In the combined administration, thiazide diuretics have to be prescribed 1 hour before the loop diuretics in order to achieve full blockade of the distal tubules.
Nephrotic patients also demonstrate intensive sodium reabsorption in the connecting and cortical collecting tubules. Thus, co-administration of furosemide and amiloride in patients with NS increases urinary sodium excretion and promotes edema resolution (47). Amiloride inhibits the above-mentioned Na+/H+exchanger in the proximal tubules and ENa+C in the distal tubules. Moreover, amiloride has recently been shown to inhibit the synthesis of the urokinase receptor uPAR. This molecule converts plasminogen to plasmin at the cell surface of distal tubular cells, which subsequently up-regulates ENa+C activity, resulting in salt and water retention and edema formation (47, 48).
Spironolactone, the inhibitor of mineralocorticoid receptors in the collecting tubules, is used to counteract aldosterone-mediated sodium reabsorption. The presence of hyperreninemic hyperaldosteronism in patients with NS and hypovolemia is an indication to spironolactone's administration. Adding spironolactone (2–3 mg/kg/day) to loop diuretics in patients with NS and hypovolemia increases urinary sodium excretion, reduces refractory edema, and prevents hypokalemia.
Intravenous administration of diuretics
Intravenous administration of loop diuretics is preferred in patients with refractory edema, where high doses of oral diuretics are ineffective. The maximal natriuretic response occurs by intravenous furosemide boluses 1–2 mg/kg, or the equivalent doses of bumetanide and torasemide, which should be administered slowly over 10–15 minutes (not exceeding the rate 4 mg/min). However, bolus diuretic therapy may be associated with a higher incidence of diuretic resistance due to prolonged intervals of sub-therapeutic drug concentrations in the kidney.
In patients poorly responding to bolus doses of the loop diuretics, a continuous intravenous infusion can be recommended. After an initial bolus of 1–2 mg/kg, furosemide, continuous infusion should be started with a dose of 0.1 mg/kg/h, which may be increased up to 1 mg/kg/h. Continuous infusions of furosemide result in a more constant delivery of diuretic to the tubules and more substantial natriuresis. Moreover, infusions of diuretics are associated with lower peak plasma concentrations and a lower incidence of side effects.
Albumin infusions
Treatment of edema in severe resistant NS and hypovolemia requires high doses of loop diuretics combined with albumin infusions. Volume repletion in this type of nephrotic patients plays a pivotal role in the management of edema. In order to achieve a temporary increase of intravascular volume and to facilitate binding of the diuretic to albumin, an intravenous infusion of 5% albumin (10–20 mL/kg over 30–60 minutes, infusion rate 5 mL/min) or 20% albumin (1 g/kg over 1–4 h) in combination with maximum doses of furosemide (120 mg) may be performed. The 20% albumin solution increases the vascular volume to 3–4 mL per 1 mL of infused solution. The rate of 20% albumin infusion should not exceed 2–3 mL/min. Usually albumin is administered as an undiluted solution, but it may be diluted with isotonic saline or 5% dextrose to increase the infused volume. Furosemide may be administered as a bolus in the midst or after the end of albumin infusion. To achieve a maximum effect, both medications may be mixed before intravenous administration. Since the effect of albumin infusion is transient the infusion should be repeated for achieving a sustained reduction in edema. On the other hand, infusion of hyperoncotic albumin in nephrotic patients without decrease of intravascular volume may trigger the development of pulmonary edema and congestive heart failure. Therefore, albumin infusions should be restricted to subjects with hypovolemia or refractory severe edema.
Alternative methods of edema management
Excess of fluid in patients with refractory edema and oliguria can be removed by prolonged veno-venous ultrafiltration with highly permeable membranes, or by hemofiltration. These methods are more effective and safe compared to a further increase of the doses of diuretics. If these methods are not available, it is also possible to use intermittent hemodialysis with isolated ultrafiltration.
Head-out water immersion (3–4 h/day) is a quite physiological method for management of patients with massive edema. There is perfect distribution of gravity on the surface surrounding the body in water immersion. Increased venous return due to hydrostatic pressure results in release of ANP, increasing natriuresis and diuresis, with a reduction of edema. However, this procedure is not widely applicable in current renal practice.
Conclusion
Renal edema is associated with renal sodium retention as a result of different pathogenetic mechanisms, including “underfill” and “overfill” pathways.
In a great number of patients with NS, the pathophysiology of edema is not related to hypoalbuminemia, hypovolemia and secondary RAAS activation, but is associated with a primary renal defect in sodium excretion. Proteinuria itself up-regulates the tubular sodium reabsorption through stimulation of NHE3 expression in the apical membrane of the proximal tubules and thick ascending limb cells, induction of Na, K-ATPase synthesis and activation of apical amiloride-sensitive ENa + C in the cortical collecting ducts.
Proteinuria-induced interstitial inflammation in persistent NS is also involved in edema formation. Both a decrease in sodium filtration and increase in sodium reabsorption occur due to production of vasoconstrictive substances in the interstitium, driven by the inflammatory cell infiltrate. Another mechanism of sodium and water retention in NS is associated with markedly blunted diuretic and natriuretic response of medullar collecting ducts to ANP. Finally, increased capillary hydraulic conductivity and hyperpermeability to proteins play an important role in the pathophysiology of nephrotic edema.
In patients with nephritic syndrome, overexpansion of the vascular compartment and edema formation result from primary salt retention and reduction in Kf and GFR.
The understanding of the mechanisms involved in edema formation is important for efficient treatment strategy. Treatment with diuretics for minimal edema is not required, and salt restriction should be recommended to such patients. Mild edema in patients with steroid-responsive NS resolves promptly upon the initiation of corticosteroid therapy. Evaluation of volemic status is necessary for rational diuretic usage. In the majority of patients, edema can be managed adequately with oral administration of an effective dose of loop diuretics. The treatment of patients with refractory edema is more difficult and comprises a strong salt restriction, treatment with intravenous furosemide, additional use of thiazide, amiloride, spironolactone and intravenous albumin. Albumin infusions should be restricted to patients with hypovolemia or refractory severe edema. Infusion of hyperoncotic albumin in nephrotic patients without decrease of intravascular volume may trigger the development of pulmonary edema and congestive heart failure.
Further clarification of sites and cellular mechanisms of primary sodium retention in patients with renal edema should be continued to create new therapeutic options.
Key messages
– Assessment of circulatory volume is the key to determining management approaches to reduce edema and avoid common complications in NS - acute kidney insufficiency in patients with “underfill”, or hypertensive and pulmonary complications in patients with “overfill”.
– Albumin infusions with diuretics co-administration should be recommended to patients with hypovolemia or refractory severe edema.
– Mild edema in patients with steroid-responsive NS resolves promptly following therapy with corticosteroids.
– Edema in patients with NS and hypervolemia can safely be treated with diuretics.
Footnotes
Financial support: No grants or funding have been received for this study.
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
