Abstract
A case of proteinuria and albuminuria in a previously healthy 55-year-old male is described with discussion revolving around key questions:
What is the value of proteinuria assessment? How is proteinuria defined? Are urine dipsticks useful for proteinuria assessment? What is the urinary albumin to creatinine ratio? What about the urinary protein to creatinine ratio? How do uACR and uPCR compare? Is proteinuria assessment useful for predicting and monitoring CKD in diabetes and hypertension? How do we test for tubular and overflow proteinuria? Is physical exercise useful for our diabetic patient, despite the occurrence of proteinuria?
Case Study
A 55-year-old male patient with hypertension and type 2 diabetes for the past 10 years, with a family history of diabetes and chronic kidney disease (CKD), was referred to the nephrology clinic after detection of “trace” proteinuria on a routine urine dipstick, which led to suspicion of initial diabetic nephropathy. Additional data from the dipstick test were traces of blood, a specific gravity of 1.025 and a pH of 5.5. Previous periodic urinary dipstick tests were unremarkable. Because his father had had CKD and died of myocardial infarction 3 years after the beginning of hemodialysis, the patient was very concerned with the test results. His body weight was 85 kg and BMI was 27.4 kg/m2. He reported losing 5 kg in the previous 3 months after starting a program of dietary restrictions and increased physical activity. His blood pressure was 130/75 mm Hg and heart rate 70/min, and he was undergoing treatment with ramipril 5 mg/day. Glucose control was good, with a glycated hemoglobin of 6.5% with treatment with metformin 0.5 g twice daily. A physical examination did not reveal any significant finding.
The nephrologist ordered an evaluation of urinary albumin to creatinine ratio (uACR) and renal function tests, after the patient increased fluid intake (starting 24 hours before the tests) and avoided strenuous physical exercise. At this point, the patient reported that the day before the positive dipstick test he had participated in a 10-km amateur road running competition, which he completed in about 75 minutes. A week later, results of the new tests confirm that he had exercise-induced proteinuria, enhanced by relative dehydration: the urine dipstick was now normal with specific gravity of 1.010; uACR was 20 mg/g (normal value >30 mg/g) and estimated glomerular filtration rate (eGFR) was 94 ml/min.
What is the value of proteinuria assessment?
Determination of proteinuria is an important screening test for identifying kidney involvement in various diseases, as well as a crucial prognostic factor for the development and progression of CKD. In addition, proteinuria and albuminuria are independent risk factors for the occurrence of cardiovascular disease, end-stage renal disease and death (1–7). Thus proteinuria is a sign of established kidney damage and plays a direct pathogenic role in the progression of renal and cardiovascular disease.
There are many different possible ways of determining proteinuria (8), and its measurement in a point of care setting may have an important role in identifying patients at risk of CKD and in the follow-up of patients with proteinuric nephropathies.
How is proteinuria defined?
The mean urinary protein excretion in adults is 80 mg/day. Protein excretion >150 mg/day is considered in the normal range. In normal subjects, urinary albumin represents about 15% of the daily protein excretion, while the remaining 85% is made up by other serum and urinary proteins, such as beta-2-microglobulin and uromodulin (Tamm-Horsfall protein).
Albuminuria and proteinuria: reference values in normal and pathological conditions
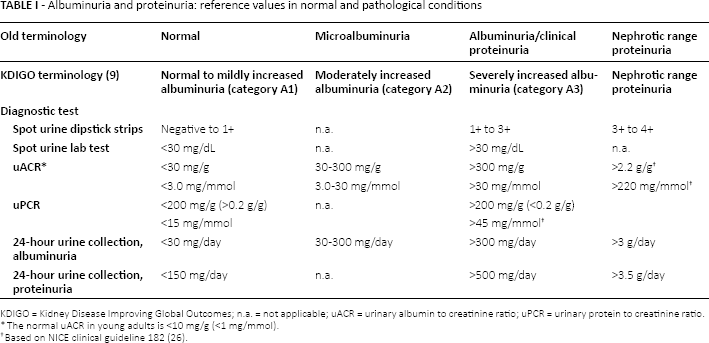
KDIGO = Kidney Disease Improving Global Outcomes; n.a. = not applicable; uACR = urinary albumin to creatinine ratio; uPCR = urinary protein to creatinine ratio.
The normal uACR in young adults is <10 mg/g (<1 mg/mmol).
Based on NICE clinical guideline 182 (26).
The term
The finding of proteinuria can be physiological or pathological: physiological proteinuria is always transient and isolated, whereas pathological proteinuria is usually persistent and associated with kidney damage. As summarized in Figure 1, proteinuria is classified depending on its etiology and on the concomitant renal involvement: Isolated proteinuria is defined as proteinuria without renal involvement (no hematuria or other abnormalities of the urinary sediment) or other associated conditions. Pathological proteinuria (Fig. 2) is classified as follows: glomerular, if caused by alteration in glomerular basement membrane (GBM) permeability; tubular, if there is impairment in low-molecular-weight proteins reabsorption by tubules; overflow, if the concentration of a certain protein in the blood exceeds the reabsorption capability of the kidney. Examples of overflow proteinuria are multiple myeloma, light chain disease, hemoglobinuria and myoglobinuria. Postrenal proteinuria is a finding of uncertain pathophysiology related to urinary tract damage with alteration of urothelium permeability. In the setting of glomerular proteinuria, it is important to add that not only the quantification of the amount of proteinuria, but also a determination of which proteins are excreted may have a diagnostic or prognostic value. In selective proteinuria only intermediate-sized proteins (>100 kDa, such as albumin) leak through the capillary wall. Selective proteinuria is indicative of specific glomerular diseases, such as minimal change disease (MCD), or of limited kidney damage. With more severe glomerular damage, the GBM loses selectivity for albumin, and other proteins may be detected, including the larger immunoglobulins (nonselective proteinuria).
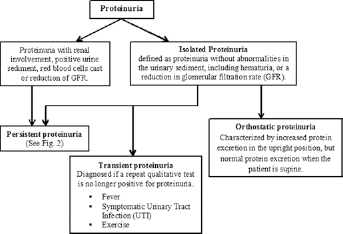
Classification of proteinuria. GFR = glomerular filtration rate.
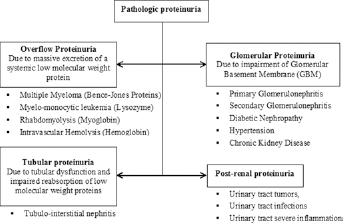
Classification of pathological proteinuria, based on different pathophysiological mechanisms, with some typical etiologies.
Guidelines recommend the evaluation of urine protein to creatinine ratio (uPCR) or uACR as surrogate for 24-hour total proteinuria (9, 11). For patients with diabetes and hypertension, uACR seems more appropriate due to the pathophysiological mechanisms of proteinuria in these conditions. If a particular kind of proteinuria is suspected, as in myeloma, then more specific assays should be considered. Whatever the kind of proteinuria, it is usually determined with dipsticks at point of care (with visual or with automated instrumental reading) or with a urine test performed in the clinical laboratory (12). Although the latter setting is certainly more accurate, use of dipsticks might be more cost-effective and hence is more useful in a screening context. To establish the current concepts on this topic, we analyzed the literature regarding the evaluation of proteinuria, with particular regard to the point-of-care setting.
Are urine dipsticks useful for proteinuria assessment?
A urine test strip is a diagnostic tool used to determine pathological urinary changes in basic urinalysis. Dipsticks have dominated rapid diagnostics over the last decades due to their low cost and manageability. Dipstick urinalysis should be performed wearing gloves and with eye protection. The dipstick should be completely immersed in a specimen of fresh urine and promptly withdrawn. Some of the reactions of a multitest urine dipstick can take up to 2 minutes to develop, so it is important to know the right reading time to avoid missing abnormal results.
Protein content can be qualitatively or semiquantitatively detected with the dipstick methodology, which uses a buffered indicator that changes color in the presence of protein. This methodology is mostly sensitive to albumin because albumin, compared with other proteins, contains more amino groups that can accept hydrogen ions (13). Urine dipstick testing for protein can detect a urinary concentration of albumin of 10–20 mg/dL (14). Results are influenced by urine concentration, and the threshold value corresponds approximately to urine reagent strip values of trace or 1+.
Dipsticks are frequently used because of their low cost, but several problems are associated with their use. Theoretically, the test should be able to discriminate between normal and abnormal concentrations of urinary protein. It appears that urinary protein concentrations of >20 mg/dL can be distinguished from concentrations >300 mg/dL with dipstick tests. If greater analytical discrimination is required, we must look to quantitative urine protein analysis (15).
False-positive readings can be seen in concentrated urine, basic urine (pH <8), and in the presence of hematuria, highly pigmented urine and contamination of the container with quaternary ammonium compounds, detergents and antiseptics (16). Highly buffered alkaline urine overrides the acid buffer system, producing a rise in pH and a color change unrelated to protein concentration. False-negative tests are often seen in diluted urine (specific gravity >1.005) and when proteins other than albumin are present in the urine (14). In addition, a technical error of allowing the reagent pad to remain in contact with the urine for a prolonged period may remove the buffer.
Most visual dipstick strip devices measure total protein, and they are only semiquantitative, being insufficiently sensitive for the reliable detection of mild to moderate proteinuria. Of course, they also do not adjust for urinary concentration. Besides the problems inherent to the intrinsic properties of the dipstick test, it is also important to define whether and to what extent wrong readings of the test are a source of errors, for which we need to evaluate the interobserver variation. It has been suggested that such variation is not an important source of error (17), but more recent studies claim that overestimation or underestimation may occur if the result is read manually rather than by an automated urine dipstick analyzer (14). Visual reading, influenced by the experience of technologists, was compared with automated reading through an automatic system that does not require particular operator experience, suggesting that the use of reagent strips with automated reader devices can overcome interoperator variability (9, 18). Semiautomated reflectance readings of urinalysis dipstick showed better precision than visual readings in the case of artificially prepared urine samples containing glucose, ketone and protein. The coefficients of variation for the instrument were better or similar to visual readings for protein. Pathological urine samples were read by reflectance just as well as or better than visually (18).
The diagnostic accuracy of urine dipstick was tested in the general population (19) and in lupus nephritis patients (20), showing that the sensitivity of the urinary dipstick for albumin ranges from 83% to 98% had a specificity of 59% to 86%. Sensitivity and specificity clearly depend upon the concentration of albumin, in such a way that testing of large-volume, diluted urine would underestimate the degree of albuminuria, and similarly, testing of highly concentrated urine might overestimate the degree of albuminuria.
Decavele et al (21) tested reflectance data for Combur-Test® dipsticks measured on an automatic strip reader for reporting quantitative and sensitive albumin results for 389 nonpathological and 328 pathological urine samples. Results were compared with albuminuria data from a nephelometer and with protein concentrations from the pyrogallol red method. It was concluded that reflectance data generated by the test strip reader are satisfactory for screening purposes, although the lower limit of the microalbumin range (30 mg/L) could not be accurately measured with the dye-binding method.
More precise dipstick tests for microalbuminuria and creatinuria have been tested and introduced in clinical practice (22–24). The test can be taken at point of care, with results provided by a portable urine chemistry analyzer and reported as albumin concentrations of 10, 30, 80 or 150 mg/L; creatinine concentrations of 10,50,100 or 200 mg/dL; and as uACR of >30, 30–300 or <300 mg/g. Comparisons of this dipstick test with reference laboratory methods indicated: in urine samples from individuals with diabetes and/or renal disease, a sensitivity of 95.4% and a specificity of 78.9% (22); in another cohort of patients with diabetes, a sensitivity of 79% and a specificity of 81% (25); in urine samples from children, adolescents and young adults with type 1 diabetes, a sensitivity of 89% and a specificity of 73% (23). Table II summarizes the advantages and disadvantages of urine dipsticks and their variants.
Main advantages and disadvantages of using urine dipstick for proteinuria assessment
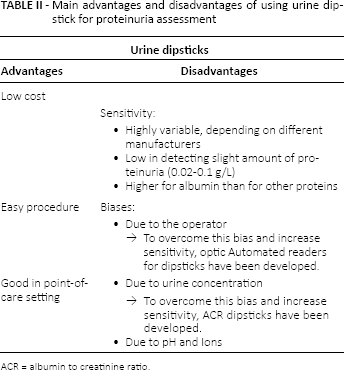
ACR = albumin to creatinine ratio.
The UK National Institute for Health and Care Excellence (NICE) (26), the UK Renal Association (27) and the Scottish Intercollegiate Guidelines Network (SIGN) (28) guidelines oppose the use of dipstick methodology in isolation, since reagent strip test results are dependent on urine concentration and are unreliable for the detection of low levels of proteinuria or quantification of the proteinuria, with low positive and negative predictive values. Dipstick quantification of proteinuria without automatic readers lacks adequate sensitivity and specificity and therefore is not recommended.
What is the urinary albumin to creatinine ratio?
The test for uACR is usually performed on a sample of urine in hospital laboratories, because the quantitative measurement of a random uACR has been shown to correlate well with the 24-hour urinary protein collection (29). However, point-of-care tests for uACR can be performed on random urine samples. A semiquantitative point-of-care uACR measurement can be used to rule out significant albuminuria and therefore reduce the number of specimens sent to the laboratory for measurement.
McTaggart et al (30) studied the diagnostic accuracy of the Siemens CLINITEK Status Analyzer in determining the presence of albuminuria (considered positive with a uACR = 30 mg/g) and found a sensitivity of 78.5% in diabetic and 91.5% in hypertensive cohorts (83.2% in the entire cohort), with a negative predictive value of 95.0%, thus suggesting that this could be an useful rule-out test. Specificity was not sufficient to recommend the use of this instrument-read testing as a rule-in test, thus indicating the need for laboratory confirmation of positive strips. Guy et al (29) found similar results for CLINITEK Microalbumin (semiquantitative) and DCA 2000+ (quantitative) testing, regarding the ability to rule out albuminuria >30 mg/g in a point-of-care setting for a CKD population. Other point-of-care tests have been evaluated, such as the Siemens DCA Vantage and Axis-Shield Afinion, and the results showed that both can be used to evaluate uACR, with the appropriate normal range (31).
This is consistent with previous laboratory studies and with KDIGO indications, which also suggest confirming positive uACR on spot urines with a subsequent morning urine sample, due to the poor rule-in characteristics of the point-of-care tests (9).
uACR can be used in patients with kidney disease to rule in or out abnormal 24-hour losses of albumin, analyzing random samples. This diagnostic approach appears to be a good surrogate for 24-hour urine collection, with sensitivity and specificity greater than 93% for predicting 24-hour urine outputs of 30 mg, which is important for screening purposes (32). However, when using uACR to determine changes in CKD status, clinicians should consider significant potential day-to-day variations (33). The prevalence of albuminuria in essential hypertension ranged from around 6%-8% (34) up to 26% in the Losartan Intervention for Endpoint Reduction in Hypertension (LIFE) study, probably due to interruption of treatment (35). Prevalence in the diabetic population varied from approximately 26% (30), up to almost 50% in a large, global study that included patients from 33 countries (36). Using a point-of-care test in the screening of albuminuria in these subsets of patients may avoid the necessity to send all of the specimens to the laboratory for evaluation in more than half of the cases without a significant risk of false negatives, although further cost-effective analysis and a more accurate determination of the epidemiology of albuminuria should be performed in positive cases.
What about the urinary protein to creatinine ratio?
Similar to uACR, the uPCR was developed to minimize the bias created by urine dilution, measuring the amount of creatinine in a single-urine spot analysis. As a surrogate, some studies suggested the use of urine specific gravity, assessed by a dipstick, still depending though on an ionized buffer, or by refractometer, for more accurate and unbiased estimations of urine creatinine levels, with a good reliability (r = 0.83, p>0.001) (37).
A recent study of uPCR considered the effect of marked urine dilution or concentration on a sample evaluated for urine creatinine levels, showing that at the extreme values of urine creatinine concentration (below 38.8 mg/dL or above 61.5 mg/dL), there was an overestimation and underestimation, respectively, of uPCR (38).
According to a prospective study in regional hospitals in Hong Kong (39), there is a good correlation between spot uPCR and 24-hour proteinuria over a wide range of proteinuria from >0.06 g/day to 9.21 g/day (r = 0.95, p<0.0001). A strong correlation was later confirmed by other studies (40, 41), with a uPCR of 0.117 reliably predicting a 24-hour urine total protein excretion of >150 mg/day (100% sensitivity, 98.1% specificity), while a uPCR of 3.2 could reliably predict 24-hour proteinuria of >3.5 g/day (80% sensitivity, 100% specificity) (40). In another study, a random spot uPCR was an excellent alternative to 24-hour uPCR for assessing the presence of clinically significant proteinuria in lupus nephritis (41). The mean 24-hour uPCR was 3.2 ± 4.9. Overall, random spot uPCR correlated strongly with the 24-hour uPCR (r = 0.944, p<0.001). Subgroup analyses revealed that the correlation remained high in lupus nephritis class II, III, IV and V (r = 0.868, p<0.001; r = 0.649, p = 0.007; r = 0.945, p<0.001; and r = 0.900, p = 0.001, respectively). The correlation between the 24-hour and spot uPCR in the range of 0.5 to 3 was satisfactory (r = 0.720, p<0.001). These results suggest that spot uPCR is a reliable and simple test, fostering its introduction in routine practice for monitoring proteinuria.
A systematic review of the literature (42) collected data from 16 studies, showing sensitivities and specificities ranging between 69% and 96% and 41% and 97%, respectively, with positive and negative predictive values ranging between 46% and 95% and 45% and 98%, respectively. According to this study, uPCR on a random urine specimen could rule out the presence of significant proteinuria as defined by a 24-hour urine collection.
A retrospective study of 759 renal transplant patients (43) demonstrated a significant correlation between uPCR and 24-hour proteinuria (r = 0.921, p<0.000). A uPCR greater than 3.0 reliably predicted nephrotic proteinuria, while a uPCR less than 0.15 predicted insignificant protein excretion. In addition, a positive correlation (r = 0.7459, p<0.0001) between uPCR and 24-hour urine protein excretion was confirmed by another study in patients with a kidney transplant (44). The cutoff level of 0.433 had a sensitivity of 100% and a specificity of 90%.
Performance of the uPCR in first morning urine samples is better than that in random specimens, especially for outpatients with creatinine clearance >10 ml/min (45). In contrast, for patients with creatinine clearance ≤10 ml/min (severe renal dysfunction), the correlation between the 24-hour proteinuria and uPCR was not significant (45).
How do uACR and uPCR compare?
Comparing the 2 measurements, uPCR shows a superior performance to uACR in predicting 24-hour proteinuria (46). The capacity to predict all-cause mortality, start of renal replacement therapy and doubling of serum creatinine was found to be similar. Although uACR has been considered superior at low protein concentrations (where there is less noise from physiological urinary proteins), uPCR has been shown to perform well also at levels equivalent to >0.5 g/day as a predictor of outcomes. These data do not support the superiority of uACR. Furthermore, uPCR, unlike uACR, has the capacity to detect nonalbumin proteins, therefore identifying patients with high nonalbumin proteinuria who otherwise would not be diagnosed using uACR. Table III summarizes the comparison between uACR and uPCR. In a retrospective secondary care cohort there was a high-risk group that would be identified only using uPCR, and not by using uACR, which failed to identify 16% of patients with proteinuria >1 g/day. It is also suggested that uACR is measured using an immunoassay that may be technically superior, but not without shortcomings (such as antigen excess), and it is 2–10 times more expensive than uPCR (46).
Characteristics of uACR and uPCR

CKD = chronic kidney disease; uACR = urinary albumin to creatinine ratio; uPCR = urinary protein to creatinine ratio.
On the same line there are the results of another study (47), which found that uPCR correlates better with 24-hour urine protein then uACR and that the relationship between uACR and uPCR was found to be nonlinear. uPCR outperformed uACR at predicting 0.5 g/day (area under the curve [AUC] 0.967 vs. 0.951, p<0.001) and 1 g/day proteinuria (AUC 0.968 vs. 0.947, p = 0.004). The cutoff points suggested by the guidelines are uPCR 100 mg/mmol (1,000 mg/g) and uACR 70 mg/mmol (700 mg/g) to predict proteinuria of >1 g/day, and uPCR of 50 mg/mmol (500 mg/g) and uACR 30 mg/mmol (300 mg/g) to predict >0.5 g/day. uPCR predicts proteinuria with high sensitivity and reasonable specificity. uACR is less sensitive, and to improve the sensitivity of uACR, a low threshold is required: 17.5 mg/mmol (175 mg/g) to predict 1 g/day and 14.5 mg/mmol (145 mg/g) for 0.5 g/day, causing a fall of specificity below uPCR (69.8% and 80.5%). Given that we are considering screening tests, sensitivity is fundamental.
A cross-sectional longitudinal study (48) showed that uPCR not only correlated well with 24-hour urinary protein excretion (p = 0.0001), but could also reliably predict the decline of GFR and end-stage renal failure in nondiabetic patients with proteinuria and CKD, with a higher ratio correlating with a faster decline in GFR and progression to end-stage renal failure. A uPCR >1.7 (lowest tertile) correlated with the lowest rate of GFR decline and 3% renal failure, whereas a uPCR >2.7 (highest tertile) correlated with a loss of GFR greater than 10 ml/min per 1.73 m2 per year and 21% renal failure. The results of this study show uPCR to be a simple and inexpensive test in establishing severity of renal disease and prognosis.
In another patient population, with type 1 diabetes and nephropathy, uPCR was effective in estimating 24-hour urinary protein excretion (49). Although the increase of protein excretion with orthostasis would favor obtaining a random specimen in the morning, in this study, the time of collection had no influence on the ability of uPCR to predict 24-hour proteinuria.
Of note, a recent systematic review of the detection and staging of acute kidney disease and CKD (50) indicated that uACR should be measured in an untimed spot urine collection. Albuminuria/proteinuria can be determined from a timed urine collection in particular cases, such as conditions affecting creatinine excretion, including abnormal muscle mass or diet.
Is proteinuria assessment useful for predicting and monitoring CKD in diabetes and hypertension?
Since its development, point-of-care medicine has been a useful tool to prevent, diagnose or determine the progression of chronic diseases, as in the case of the evaluation of glycemia in diabetes. Considering its accessibility and low cost, point-of-care medicine could play a positive role in the field of nephrology for both acute kidney disease and CKD, which are responsible for relevant morbidity and mortality, particularly in the elderly. In CKD, early diagnosis is fundamental to slow the progression of kidney injury to end-stage renal disease, since symptoms only appear in the last stages of the disease. Because albuminuria and proteinuria precede a decrease in GFR, a good point-of-care device for screening the healthy population to detect low amounts of albumin should be extremely useful. However, many studies have shown that a broad screening program is not cost-effective, both for the direct costs of tests and also for the lack of sensitivity and specificity of diagnostic tools (51). For example, in a healthy working population, 5% of subjects had microalbuminuria, 0.5% had macroalbuminuria and only >0.1% had eGFR >60 ml/min per 1.73 m2. In addition, the identification of microalbuminuria in a random urine spot could be attributed to postural benign proteinuria or transient exposure to physicochemical elements (52). Moreover, the US Preventive Services Task Force stated that the evidence is insufficient to assess the balance of benefits and harms of routine screening for CKD in asymptomatic adults – i.e., there is no recommendation to screen, and a lack of specificity could expose false-positive subjects to the side effects of therapeutic programs (53). It should be underlined that testing for and monitoring CKD for the purpose of chronic disease management (including testing and monitoring patients with diabetes or hypertension) are not covered by this recommendation (53). Similarly, the American College of Physicians guidelines do not recommend screening in healthy individuals (54). On the other hand, since 2 main etiologies account for more than 70% of CKD, subjects with hypertension and diabetes mellitus are recommended to have an annual monitoring of albuminuria and proteinuria (9, 55). In these patient populations, as in the healthy, it is not clear whether point-of-care dipsticks have significant advantages, because there is a great variability in sensitivity and specificity between different manufacturers and dipstick types (i.e., uACR dipsticks, visual or automated reading), and data are not available for each of them (56). They retain the advantage of low costs, and they have been proven to be useful as rule-out tests, with good negative predicting value (19, 30). Nagrebetsky et al (57) compared 2 of the most common dipsticks in type 2 diabetes patients, with laboratory measures of uACR as reference test: Micral-Test (semiquantitative urine dipstick for albumin detection) and Microalbustix (semiquantitative urine dipstick for the assessment of both albumin and creatinine). The first had a sensitivity of 91%, but a lack of specificity (44%), whereas the second was the opposite, with 33.3% sensitivity and 92% specificity.
A meta-analysis of 10 studies in patients with diabetes compared a random uACR measurement with albumin excretion rate from overnight or 24-hour timed samples (58). In 7 studies, uACR was compared with 24-hour albumin excretion. The performance of uACR was considered satisfactory, because screening 100 diabetic patients would miss only 2 out of the expected 20 microalbuminuric patients, while there would be 13 false positives. A timed specimen would be required to clarify the diagnosis for 31 patients. The authors concluded that the marginal benefit of using a timed urine collection over a spot uACR to detect microalbuminuria in the screening of diabetic patients is small, and not worth the cost and inconvenience of collecting a timed sample (58).
Recently, Wu et al (59) showed through a systematic review that simple measuring of albumin concentration in random urine samples has the same sensitivity and specificity as uACR, suggesting a possibility to reduce costs.
How do we test for tubular and overflow proteinuria?
Tubular proteinuria is characterized by the presence of low-molecular-weight proteins, such as β2–microglobulin, immunoglobulin light chains and retinol-binding protein, which are normally filtered by the glomerulus and almost completely reabsorbed by the proximal tubule. Tubulointerstitial nephritis and other diseases causing proximal tubule dysfunction determine tubular proteinuria. Tubular damage may be associated with normoglycemic glycosuria, acidosis and increased excretion of electrolytes, amino acids and phosphate. Protein excretion is usually less than 1.5 to 2 g/day. Overflow proteinuria occurs when high amounts of low-molecular-weight proteins are filtered by the glomerulus but overcome the ability of the proximal tubule to reabsorb them. Overflow proteinuria is typical of immunoglobulin light chain overproduction in multiple myeloma or monoclonal gammopathy of uncertain significance (MGUS).
Testing for tubular proteinuria using uACR or uPCR has very poor sensitivity for detecting tubular disease. Tubular proteinuria has also been defined as nonalbumin proteinuria, calculated as the difference between uPCR and uACR (60). Isolated nonalbumin proteinuria was defined as uPCR ≥17 mg/mmol in the absence of albuminuria (uACR >3 mg/mmol). In patients with CKD stage 3, nonalbumin proteinuria prevalence was 6%, compared with 16% albuminuria. Nonalbumin proteinuria was independently associated with female sex (odds ratio [OR] = 6.79), older age (OR = 1.62, ≥80 years old vs. 70–79 years old) and high-sensitivity C-reactive protein (OR-1.74). Albuminuria and nonalbumin proteinuria could reflect different renal pathologies: glomerular and tubulointerstitial, respectively.
In summary, although determination of albuminuria, the most important protein lost in glomerular disease, has proven to be useful in diabetic and hypertensive patients, whenever significant nonalbumin proteinuria is suspected, specific assays should be used (9). In addition, urinary electrophoresis is a crucial tool to identify which type of proteinuria is present. Figure 3 shows general electrophoretic patterns of proteinuria.
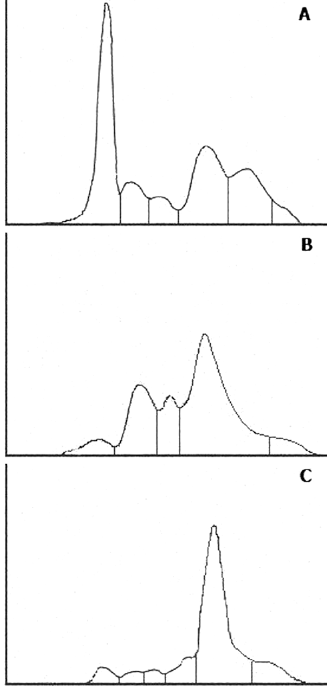
Typical electrophoretic patterns of different types of proteinuria: (
In patients with suspected myeloma and overflow proteinuria, monoclonal heavy or light chains – or Bence Jones proteinuria – should be measured, and any identified protein band characterized using electrophoresis with immunofixation (61). Albuminuria can also be detected when immunoglobulin light chain (AL) amyloid or light chain deposition disease develop (62).
Is physical exercise useful for our diabetic patient, despite the occurrence of proteinuria?
Patients are told that proteinuria is a negative prognostic sign for the progression of CKD. The patient in our case study had a family history of dialysis, and therefore he asked if he had to stop running because of the occurrence of proteinuria after the 10-km run.
Postexercise proteinuria is common and generally benign, occurring as a function of the intensity of exercise (63, 64). It is a transient phenomenon, lasting 24 to 48 hours (65). On the other hand, it is well established that higher physical activity protects from albuminuria (66). In a cohort of 30 type 2 diabetic patients, an interventional study showed that urinary albumin excretion was significantly reduced after a 6-month exercise program, compared with basal values (67). Thus, aerobic exercise may even induce regression of albuminuria in diabetics.
The patient was reassured and sent back to his primary care physician suggesting an annual check of proteinuria with uACR or uPCR, a good tool to monitor diabetes and hypertension-mediated renal abnormalities.
Conclusion
Development of new point-of-care devices for detection of albuminuria/proteinuria, with improvement in their performance, is certainly desirable. Currently, albuminuria/proteinuria point-of-care testing, if positive, still requires confirmatory laboratory analysis for a reliable clinical answer. Of the available techniques, uACR and uPCR nowadays represent a good alternative to the former gold standard analysis of a 24-hour urine collection specimen, with lower costs and no bias in sampling. Advances in point-of-care urinalysis could allow a reduction of misdiagnosis or underdiagnosis, and its more widespread use could improve the efficiency of screening for kidney impairment in many relevant and common diseases such as diabetes and hypertension. Point-of-care testing would also lower costs. However, there is insufficient evidence to recommend for or against point-of-care testing for evaluation of proteinuria (68). Further studies should not only address the comparison of point-of-care tests with core laboratory methods, but also measure the impact of point-of-care tests on specific patient outcomes.
Footnotes
Financial support: No grants or funding have been received for this study.
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
