Abstract
A case of an adult female with Fabry disease is described with discussion based on the following key questions:
What is the natural history of Fabry nephropathy? What are the indications to perform kidney biopsy in Fabry disease? How to perform the workout of the patient in recognition of systemic organ damage? Is the missed recognition of Fabry disease frequent in dialysis patients? When and which patients are eligible to start enzyme replacement therapy? Is enzyme replacement therapy effective in Fabry nephropathy? What is the outcome of the patient who underwent a kidney transplantation? Is the supportive therapy important in Fabry disease nephropathy? What are the future therapeutic perspectives?
Study case
SV was a 23-year-old female admitted into the nephrology department for a persistent non-nephrotic proteinuria around 1 g/day. Ten years before her father started hemodialysis for a chronic glomerulonephritis, which occurred during youth
On physical examination the girl was completely asymptomatic. The blood pressure and heart rate were 120/70 mmHg and 68 bpm, respectively. The laboratory tests showed a serum creatinine of 0.7 mg/dL and an estimated creatinine clearance of 92 mL/min. The proteinuria was 870 mg/day. All serum IgA, IgM, IgG, C3 and C4 and ANA, ENA, nDNA, ANCA were in the normal range. The kidney biopsy documented at light microscopy revealed numerous vacuolated cells in both glomerular and tubular cells. Immunofluorescent study was negative for IgA, IgM, IgG, C1q, C3 and C4. Electron microscopy showed numerous and extended osmiophilic and laminated ‘zebra’ bodies in podocytes, epithelial and endothelial cells of the glomerular tuft that lead to the diagnosis of Fabry disease. The subsequent workout demonstrated a normal leukocyte α-galactosidase A activity 19 nmoL/mg prot/h while the DNA analysis showed the missense mutation 1354 K. The skin examination was negative for angiokeratomas while the slit-lamp examinations revealed a typical cornea verticillata. Two weeks later she started enzyme replacement therapy (ERT) with a standard dose of agalsidase beta at 1 mg/kg every 2 weeks.
By the clinical reassessment of the father it was recognized that the kidney biopsy performed before the end-stage renal disease (ESRD) lacked of the EM examination. The leukocyte α-galactosidase A activity was absent and the DNA analysis showed the same missense mutation as the daughter.
A number of questions should be asked in such situations:
What is the natural history of Fabry nephropathy?
Fabry disease is a severe, progressive X-linked inborn error of metabolism due to the absent or deficient activity of lysosomal glycohydrolase α agalactosidase A that results in progressive accumulation of globotriaosylceramide (Gb3) into lysosomes. The storage occurs in a variety of cell types, and in particular in endothelial cells, cardiomyocytes, fibroblasts, nerve cells and renal cells (podocytes, glomerular endothelial, mesangial, tubular cells) (1). In classic forms of Fabry disease, signs and symptoms emerge in childhood and adolescence with the onset of typical episodes of neuropathic pain, gastrointestinal disturbances, angiokeratomas, proteinuria and hypohidrosis until the onset of signs and symptoms of severe kidney involvement, cardiomyopathy and stroke (Tab. I).
Typical signs and symptoms of Fabry disease according to age
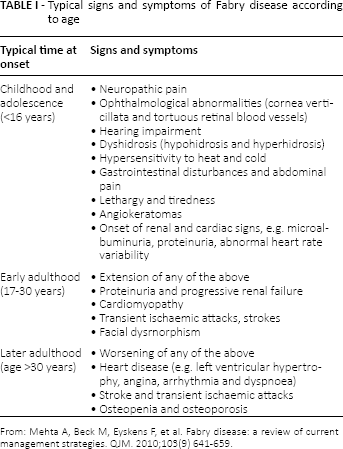
From: Mehta A, Beck M, Eyskens F, et al. Fabry disease: a review of current management strategies. QJM. 2010;103(9) 641–659.
The severe organ dysfunction is responsible for a substantial reduction in health-related quality of life and a premature mortality (1–5). In the cases that affect males but also a limited percentage of female patients, the leukocytes α-GAL A activity is nearly absent due to severe mutations such as deletion of the X-chromosome (1). In contrast, late-onset variants with residual α-Gal A activity (1%–5% of normal) have also been described. These patients present later in life a cardiac involvement without the presence of classic manifestations of angiokeratoma, acroparesthesias, hypohidrosis and the characteristic ophthalmologic findings. Among these “cardiac variants” the proteinuria is frequent, but usually do not develop renal failure (6).
The natural course of Fabry nephropathy begins in children or adolescent patients and include albuminuria and proteinuria developing as early as in the second decade of life. In both sexes, albuminuria may be an even more sensitive marker of renal involvement than proteinuria. Recent studies on affected pediatric patients have demonstrated that albuminuria may be present and precede overt proteinuria in a cohort of pediatric and adolescent patients with Fabry disease (7, 8).
In normal individuals, renal function declines with age at approximately −1 mL/min per 1.73 m2 per year during the sixth decade and more rapidly thereafter (9). The glomerular filtration rate (GFR) decline in males with Fabry nephropathy is comparable to that in patients with diabetic nephropathy. In the NIH series, progression from chronic kidney disease (CKD) to ESRD occurred over a mean of 4 ± 3 years while the decrease in creatinine clearance was −12 mL/min per year (10). As in other chronic proteinuric nephropathies, baseline proteinuria is an important prognostic indicator for progression of Fabry nephropathy (11). Ortiz et al (12) described the distribution of proteinuria and GFR in 585 male and 677 female patients from the Fabry Registry. Proteinuria was most frequently mild to moderate, between 300 and 3500 mg/24-h. Overt proteinuria was found in 62% of males and 33% of females. The prevalence of proteinuria and its degree increased with decreasing renal function in both sexes, except CKD stage 4/5. Nephrotic range proteinuria was observed only in 2%-3% of males or females with CKD stage 1 or 2. The prevalence of nephrotic range proteinuria increased in more advanced CKD stages; in other words, nephrotic range proteinuria may anticipate the decrease of GFR in males and perhaps also in females (12).
What are the indications to perform kidney biopsy in Fabry disease?
Since Fabry disease is a proteinuric disease, kidney biopsy represents the gold standard for the diagnosis of renal involvement and severity assessing. Indeed, renal biopsy is useful for confirming the diagnosis of Fabry nephropathy in case of the onset of a proteinuria in a patient with pain or angiokeratomas (13,14). In other cases, the indications to the renal biopsy require more attention. Simplifying this issue, a kidney biopsy should be performed: a) in male patients in case of an atypical presentation of the disease (i.e., macroscopic hematuria, nephrotic syndrome, absence of urine findings, ESRD), while in a typical classic patient it is not essential even if recommended for confirming the nephropathy (13, 14); b) in female patients (15,16); c) when there is the suspicion of a superimposed pathology such as IgA nephropathy (17–19), diabetes (20), Lupus (21), crescentic glomerulonephritis (22); d) to monitor the amount of renal storage and to monitor the efficacy of ERT in the clearance of renal cells storage (23, 24); e) in case of rapid progression of renal function (slope e-GFR >3 mL/min/1.73 m2/year) (13,14, 25).
However, in several countries the execution of renal biopsy is not frequent due to lack of expertise or to a different cultural approach to renal diseases. By contrast, recent studies on children and adolescent with Fabry disease have demonstrated that a repeated kidney biopsy every three or five years from the start of ERT can be of great utility for monitoring the efficacy of agalsidase therapy based on the entity of clearance of deposits in renal cells (6, 25). The same Norwegian group has demonstrated that the percutaneous renal biopsy is a low-risk procedure in all ages either for mild (hematoma and hematuria) or severe (blood transfusion, surgical or catheter intervention) complications (26, 27).
How to perform the workout of the patient in the recognition of systemic organ damage?
As the diagnosis of Fabry disease is confirmed, all affected patients should have a detailed medical and family history taken. Pain, gastrointestinal complications, presence and distribution of angiokeratomas, and all other signs and symptoms should be documented at baseline and at least annually. Monitoring and treating pain is especially important in children and may require additional follow-up. Annual evaluation should include a physical examination, routine hematology and chemistry tests, and urinalysis. Since Fabry disease is a systemic disease the diagnostic workout is based on a multitude of clinical and instrumental investigations able to recognize any sign or symptom of organ involvement to perform either in symptomatic or in asymptomatic patients (28).
In Table II are listed the main clinical and instrumental investigations to explore each organ damaged in Fabry disease and the follow-up suggested for the monitoring of affected patients.
Guidelines for baseline examination and follow-up of patients affected with Fabry disease
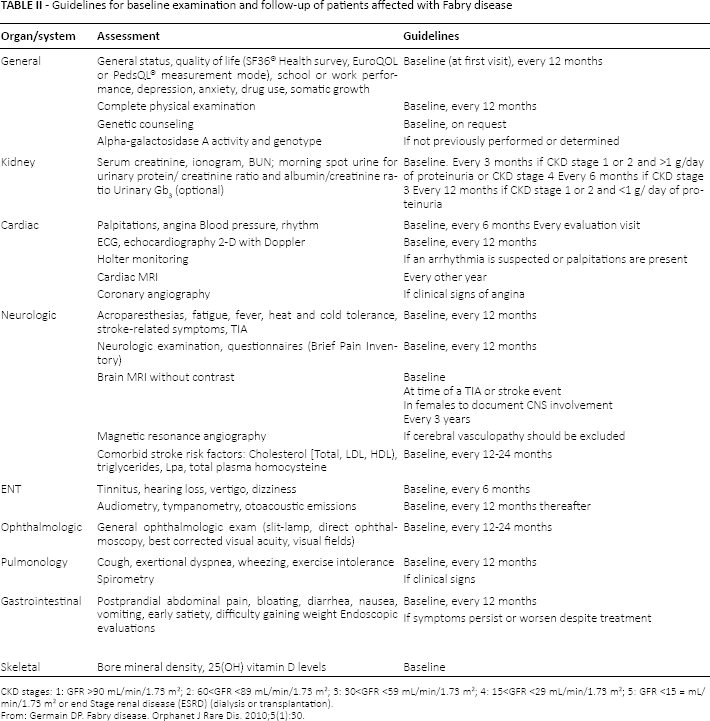
CKD stages: 1: GFR >90 mL/min/1.73 m2; 2: 60<GFR <89 mL/min/1.73 m2; 3: 30<GFR <59 mL/min/1.73 m2; 4: 15<GFR <29 mL/min/1.73 m2; 5: GFR <15 = mL/min/1.73 m2 or end Stage renal disease (ESRD) (dialysis or transplantation).
From: Germain DP. Fabry disease. Orphanet J Rare Dis. 2010;5(1):30.
Indeed, the recognition of a hidden abnormality linked to Fabry disease is very important whereas the decision of whether or not to start ERT is frequently based on the presence or the recognition of an asymptomatic abnormality. For Fabry nephropathy, the urine analysis together with urinary creatinine-to-albumin and creatinine-to-protein ratio and creatinine clearance or GFR tests, represent the main blood and urine investigations to explore the kidney function. They should be repeated every 3, 6 or 12 months in base on the baseline CKD stage.
Heterozygote females were considered simply carriers of the disease until some years ago. However, evolving knowledge about the natural course of disease indicates that female patients with Fabry disease may suffer from the same signs and symptoms as classic males but with a wide variability ranging from an asymptomatic disease course occasionally observed to the “classic” severe phenotype observed in males. This phenotypic heterogeneity is due to lyonization (29), a process whereby one copy of the X-chromosome is randomly inactivated in all cells of the female leading to a ‘mosaicism’ of normal and mutant cells in varying proportions. Such variability in symptom severity is characteristic of X-linked heterozygotes and should be kept in mind when assessing and diagnosing potential patients (15). Consequently, heterozygote females should not be re-evaluated every 3 to 5 years as suggested some years ago. Now the follow-up time for monitoring female patients is stricter with increasing frequency with age and clinical phenotype. Therefore, after a complete baseline examination, symptomatic female carriers should be followed with the same periodicity as male patients while asymptomatic females may be followed annually.
Is the missed recognition of Fabry disease frequent in dialysis patients?
The overall prevalence of renal replacement therapy (RRT) in the European population is about 1000 per million population (pmp) (30). Up-to-now it can be estimate that about 30% of these patients starts RRT without a clear diagnosis of renal disease. Hence, ESRD is not a rare condition. Some of the underlying conditions that lead to ESRD are considered to be rare and recent reports estimate that about 12.4% of all the RRT patients would be suffering from a rare disease (31). Fabry disease represents one of the most frequent rare diseases that lead to ESRD. In the past, large registries have reported a prevalence of Fabry disease in ESRD patient populations of up to 0.017%-0.020% (32,33). Spada and Pagliardini performed the first screening in a RRT population from northern Italy observing a prevalence of Fabry disease almost 10 times as high (34). After that, several studies based on screenings in patients with chronic renal failure, on dialysis or with transplant confirmed a prevalence of Fabry disease ranging from 0.20% to 0.35% (35–37). The reasons for the underestimation of Fabry disease in RRT population can be attributed to the lack of renal biopsy in several ESRD patients, a wrong interpretation of kidney biopsy, missed implementation with ultrastructural analysis of the biopsy, the co-existence of associated nephropathies or a misinterpretation of pain in children. The main reason why misdiagnosis is frequent is that one does not think about it in the presence of some sign or symptom indicative of Fabry disease.
As a consequence, the diagnosis is late, occurring during the 3–4th decade of life delaying subsequently the beginning of ERT This delay has allowed the disease to progress since the storage of substrate proceeds in several tissues (kidney and heart in particular) which leads to an irreversible failure of renal and cardiac function.
When and which patients are eligible to start ERT?
Since Fabry disease is an inherited disease that classically onsets in childhood, ERT should be started in children at the first onset of pain, abdominal symptoms or albuminuria, but also in asymptomatic children with a severe mutation or a severe familiar phenotype. Unfortunately, the diagnosis is often late leading to a delayed start of ERT. All adult patients with proteinuria or impaired renal function who are diagnosed for Fabry disease, should begin ERT as soon as possible By contrast, data from Fabry registry document show that there is a gap of about 3.5 years in males and 7 years in female between the age of diagnosis and the age of start of ERT (38). This gap confirms that Fabry disease is overlooked: in untreated symptomatic patients ERT is not introduced as soon the diagnosis is made and clinical monitoring and follow-up is often lacking in treated and untreated patients.
Beginning ERT in patients with advanced renal impairment, such as patients with proteinuria >1 g/day, with eGFR <60 mL/min/1.73 m2, or already on dialysis or with transplant, is controversial. Recent European Renal Best Practice (ERBP) through systematic literature reviews and a consensus meeting with a panel of experts, have released some recommendations about Fabry nephropathy. One of the statements presented in this study suggests not starting ERT in patients with proteinuria (protein-to-creatinine ratio >1 g/g [>0.1 = g/moL] creatinine) or eGFR <60 mL/min/1.73 m2, except for non-renal indications. In a patient on hemodialysis and when ERT is indicated, they recommend administering the ERT during a hemodialysis session. In the same study, kidney transplantation is considered as a valuable option in patients who are eligible for this intervention. However, after renal transplantation they do not suggest ERT for renal indications, but it can be continued for non-renal indications (39). Even if not supported by randomized studies but only by clinical experience, criticisms of these recommendations have arisen and they come from the demonstration in cohort studies that ERT is able to slow down cardiomyopathy and in both dialysis and transplant patients (40, 41). Moreover, is quite likely that patients with advanced renal damage and on RRT do not have non-renal indications (such as pain or abdominal disturbances or cardiomyopathy) to start or continue ERT.
Finally, in late onset variant adult patients, ERT should be started when the diagnosis has been made, while in affected familiar members agalsidase therapy should be initiated at the onset of signs or symptoms of the disease.
Is ERT effective in Fabry nephropathy?
Until now, ERT represents the main therapeutic choice in the treatment of Fabry disease. Two enzyme preparations are available, agalsidase alfa (Replagal®, Shire Human Genetic), given at 0.2 mg/kg every other week, approved in the European Union in 2001, and agalsidase beta (Fabrazyme®, Genzyme Sanofi), given at 1.0 mg/kg every other week, approved in the USA and in and EU (42, 43). Since the introduction of agalsidase, results on the efficacy of ERT in Fabry patients are especially based on cohort studies or registries data analysis, while very few are randomized controlled studies performed up to now.
In one of the larger studies aimed to investigate the effectiveness of ERT with agalsidase alfa in patients enrolled in the Fabry Outcome Survey (FOS), renal function was assessed in 208 patients (134 males and 74 females) followed for a mean period of 7.4 years. The eGFR changed from 92.9 ± 55.4 at the baseline to 76.4 ± 48.7 mL/min per 1.73 m2 at the end of follow-up (p = NS). Patients with 24-h protein excretion >1 g/24-h had poorer renal function at baseline and follow-up compared with patients with protein excretion of 500–1000 mg/24-h or with proteinuria <500 mg/24-h (44). Similar results have been observed recently by Beck et al analyzing data from FOS (45). Over a median follow-up of ˜5 years the estimated annualized eGFR change with treatment was −1.68 mL/min/1.73 m2/y in treated males with baseline eGFR ≥60 mL/min/1.73 m2 compared with −3 mL/min/1.73 m2/y in the published untreated cohort (46). Among male patients with poor renal function at baseline (eGFR <60 mL/min/1.73 m2) the eGFR change was −2.86 ml/min/1.73 m2 compared with −6.8 in the published untreated cohort (46). However, there are patients who demonstrated a continued progressive decline in renal function despite administration of agalsidase alfa (47).
The effects of intensified ERT with agalsidase alfa in patients with advanced Fabry disease who did not respond to biweekly ERT have been investigated by Schiffmann et al with off-label weekly administration of agalsidase alfa. The doubling of dose decelerated the slope of decline of GFR and ESRD was significantly delayed in nine out often patients (48, 49). These data support the hypothesis that the amount of dose infused to the patients could have a role in the outcome of treated patients with Fabry disease. These result were not confirmed in a more recent randomized, double-blind, placebo-controlled, crossover study where three different doses of agalsidase alfa (0.2 mg/kg every other week, 0.1 mg/kg/week, 0.2 mg/kg/week) were administered for a short period of four weeks in 18 patients without recognizing a significant difference in terms of self-assessed health state, or for pain scores (50). But the role of dose infused has been recently stressed by Warnock and Mauer (51) after the publication of a study where the effects on renal inclusion and albuminuria were investigated in a cohort of pediatric and adolescent patients (25). In this study they evaluated the effect of 5 years of treatment with agalsidase alfa or agalsidase beta in 12 consecutive patients aged 7–33 years (median age, 16.5 years) monitoring the efficacy of ERT with renal biopsies and albuminuria at baseline and after 5 years of ERT. After a median period of 65 months, biopsy findings from all patients showed total clearance of inclusion in endothelial, mesangial and tubular cells. The patients who received the highest dose of agalsidase exhibited substantial clearance of podocytes inclusion and albuminuria normalization (25). Also the analysis of registries data during the shortage period confirms that patients who withstand a 50% dose reduction or a switch from beta to alfa formulation showed an increase in proteinuria over 1 g/day while in patients who continued with a standard dose of agalsidase beta proteinuria reduced after a 12-month follow-up period (52).
Concerning the efficacy of agalsidase beta, in a multicenter, randomized, placebo-controlled, double-blind, phase 3 clinical trial of 58 patients with classic Fabry disease and in the subsequent open-label 54-month extension study, Fabrazyme® given at a standard dose of 1 mg/kg/2 weeks was demonstrated to stabilize renal function in patients with mild-to-moderate renal involvement. In this study, baseline proteinuria (>1 g/24–h), or a glomerulosclerosis at renal biopsy >50% at treatment baseline were identified as important factors that limited renal response to therapy (53). More recently, Germain et al investigated the long-term 10-year follow-up period outcomes of 52 of 58 patients from the phase 3 clinical trial of agalsidase beta (1 mg/kg/2 weeks). Patients with a urine protein to creatinine ratio (UPCR) ≤0.5 g/g and with <50% sclerotic glomeruli were classified as low renal involvement (LRI); patients with UPCR >0.5 g/g or with ≥50% sclerotic glomeruli at baseline were classified as high renal involvement (HRI). The LRI group (mean age 25 years at baseline) experienced some loss of eGFR (–1.89 mL/min/1.73 m2/year) while patients with HRI, who began treatment at a mean age of 38 years showed the greatest decline in eGFR slope (–6.82 mL/min/1.73 m2/year). In summary, over the 10-year follow-up, renal disease progression seems to correlate to the severity of the disease before treatment. Eighty-one per cent of adult patients with classic Fabry disease receiving agalsidase beta treatment for a median of 10 years remained free of severe clinical events and 94% of patients were alive (54).
In summary, these studies suggest that ERT is at best only effective in CKD Stage 1 or 2, before the deterioration of renal function or onset of overt proteinuria, as it does not reduce proteinuria per se.
What is the outcome of the patient who underwent a kidney transplantation?
Early analysis of transplant outcome in untreated patients documented as either graft and patient survival rate were similar in transplant patients with or without Fabry disease (55, 56). However, these reports are based on United States Renal Data System Registry database or Organ Procurement Transplant Network and do not analyze the effect of ERT in treated patients, the outcome of graft function and the outcome of cardiomyopathy. Up to now no randomized controlled studies are available on the effect of ERT in patients with renal transplant but only cohort or registries studies.
The largest nationwide collaborative open-label study was conducted in Italy where 16 transplant patients on agalsidase therapy were observed for a long-term follow-up period. After 48 months of ERT, no change in renal function was observed and serum creatinine increased from 1.78 mg/dL at baseline to 1.92 mg/dL (p = NS) after 4 years of ERT with a rate of decline in renal function from baseline of −1.92 ml/min per year. No significant change in cardiomyopathy was documented at study conclusion (41). Cybulla et al (57) explored the effects of agalsidase alfa in transplant patients with Fabry disease based on FOS data analysis. After 2 years of ERT in 20 allograft patients there was a slight but not significant increase in serum creatinine (1.4 mg/dL at baseline vs. 1.6 mg/dL) and a decrease in eGFR (59.2 mL/min/1.73 m2 at baseline vs. 51.1 mL/min/1.73 m2 at 2 years) (57). Therefore, agalsidase alfa and beta seem to be safe, well tolerated and effective in renal transplant recipients.
Is the supportive therapy important in Fabry disease nephropathy?
Since it is generally accepted that in most forms of proteinuric kidney disease, overt proteinuria, per se, is a specific risk of progression of kidney disease (58), it is mandatory to restore it also in Fabry disease. Several studies have demonstrated that both agalsidase formulations cannot restore proteinuria alone (44,45,59–61). In a recent study, Warnock et al (62) assessed the efficacy of a combined therapy with a standard dose of agalsidase beta and antiproteinuric therapy with ACE inhibitors or angiotensin II receptor blockers (ARBs) in a cohort of 24 patients followed over 21 months (Fig. 1). The goal was the maintenance of UPCR <0.5 g/g or a 50% reduction in baseline UPCR. Eighteen out of 24 patients reached the UPCR goal with eGFR slopes that were significantly better than six patients who did not achieve the UPCR goal. Kidney function was not preserved in the six patients who did not reach the UPCR treatment goal. But also in 12/18 patients achieving the UPCR goal, kidney function still progressed with an eGFR slope <–2 mL/min/1.73 m2/year. These were the patients who started ERT later (37.9 years vs. 45.1 years) and who had a shorter duration of treatment (2.6 years vs. 5.1 years). The eGFR slope was significantly worse in patients who did not achieve the goal for controlling proteinuria using ACE inhibitor and/or ARB urine protein excretion and in patients who started later with a short duration of therapy, which is confirmed by the present findings.
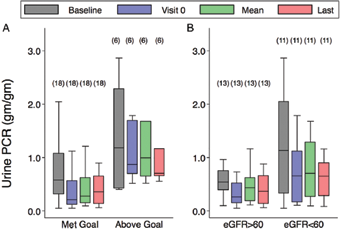
Stratified at urine protein to creatinine ratio (UPCR) throughout the treatment. (UPCR) at different study levels. (
These results underline the importance of the age at which ERT is initiated and support the recommendation that agalsidase beta therapy should be started before there is significant renal damage expressed by the presence of proteinuria.
What are the future therapeutic perspectives?
One of the most studied alternative and promising therapy in Fabry disease is the chaperon therapy. Pharmacological chaperone therapy is a rather new approach consisting of targeting incorrectly folded proteins by small molecules, thus facilitating the correct folding of the protein and inducing a recovery of its functionality. Migalastat HCL is a pharmacological chaperone for the treatment of α-galactosidase A (a-Gal A) deficiency responsible for Fabry disease. It is an analog of the terminal galactose of GL-3 that selectively and reversibly binds and stabilizes some mutant forms of α-Gal A (63). The mechanism of action of migalastat HCL is to bind and stabilize mutant α-Gal A initially in the ER, preventing misfolding and premature degradation and facilitating cellular trafficking to lysosomes where the breakdown of the GL-3 substrate can proceed. Migalastat HCL is currently being evaluated in Phase 3 studies as a single agent for the treatment of Fabry disease in patients with GLA mutations that are conformable to chaperone therapy. A missense mutation in the GLA gene and residual α-Gal A activity of at least 3% of normal was the demonstration of an increase in α-Gal A activity in the presence of migalastat HCL in patient-cultured lymphocytes. The initial criteria for enhancement required a relative increase in α-Gal A activity of at least 20% in the presence of migalastat HCL In contrast to agalsidase therapy, migalastat is orally available and has broad tissue distribution.
As such, migalastat HCL is currently in clinical development to evaluate its safety and efficacy either as a monotherapy or associated with agalsidase for Fabry disease. Germain et al demonstrated that when administered alone at an oral dose of 150 mg on an every-other-day regimen, migalastat HCL was well tolerated, increased α-Gal A activity in patients with responsive GLA mutation and resulted in GL-3 substrate reduction. Phase 3 studies of migalastat HCl for Fabry disease are ongoing (64). More recently, in an open-label phase 2 study in 23 male patients Warnock et al (65) analysed the effects of co-administration of two dose levels of migalastat HCL (150 mg and 450 mg) together with three different doses of agalsidase (agalsidase alfa 0.2 mg/kg, agalsidase beta 1.0 mg/kg and agalsidase beta 0.5 mg/kg). The rationale for this study was to provide drug-drug interaction information after co-administration of migalastat HCl with agalsidase. The combination of migalastat HCL and agalsidase increased the α-GAL Cmax. These data indicate that migalastat HCl co-administration can increase the exposure of active agalsidase in the circulation. Moreover, co-administration of migalastat HCl at doses of 150 mg and 450 mg with agalsidase alfa or beta was generally well tolerated (65).
Footnotes
Financial support: No grants or funding have been received related to this study.
Conflict of interest: None of the authors has financial interest related to this study to disclose.
