Abstract
Cardiorenal syndromes (CRS) involve disorders of the heart or kidney whereby one organ dysfunction leads to the dysfunction of another. Five types of CRS are defined. While the first 4 types describe acute/chronic cardiorenal or renocardiac syndromes, type-5 CRS refers to secondary CRS or cardiorenal involvement in systemic conditions and describes the concomitant presence of renal and cardiovascular dysfunction. Type-5 CRS is a recently defined clinical syndrome and complete epidemiological data on this entity are still lacking. In the following review, epidemiological, pathophysiological, clinical, and therapeutic approaches to type-5 CRS will be discussed according to more recent findings.
Introduction
The cardiorenal syndromes (CRS) have been systematically defined recently as disorders of the heart or kidney whereby 1 organ dysfunction leads to the dysfunction of another. Five types of CRS are defined (1). The first 4 types describe acute or chronic cardiorenal or renocardiac syndromes. Type-5 CRS refers to secondary CRS or cardiorenal involvement in systemic conditions. It is a clinical and pathophysiological entity to describe the concomitant presence of renal and cardiovascular dysfunction. Type-5 CRS can be acute or chronic (Tab. I) and it does not strictly satisfy the definition of CRS. However, it encompasses many conditions where combined heart and kidney dysfunction is observed. As this entity has only recently been described, there is limited information about the epidemiology, clinical course, and treatment of this condition. All vital organs of the body share biological information, which is termed “organ crosstalk.” The normal physiological functions of the body depend on this normal network. One organ dysfunction can result in the dysfunction of another. The interaction between the heart and the kidney is fairly common. Heart and kidney dysfunction can be observed in many hospitalized patients, especially in the intensive care unit. Over the last decade, many intensivists, cardiologists, and nephrologists have shown keen interest in the pathophysiology of this organ crosstalk between the heart and kidney. Many terms for this organ crosstalk have been suggested, such as cardiorenal anemia syndrome, cardio-renal syndrome, reno-cardiac syndrome. Ronco et al (1) have proposed the definition and subdivision of CRS into 5 subtypes. Irrespective of the first insult (heart failure causing kidney injury or renal failure causing heart disease), CRS portends increased mortality and morbidity. Type-5 CRS is a recently defined clinical syndrome and complete epidemiological data on this entity are still incomplete.
Conditions causing acute and chronic type-5 cardiorenal syndrome

Pathogenesis of CRS-5
CRS-5 and Sepsis
Inflammation and microvasculature alterations form the basis of the pathogenesis for involvement of both the kidneys and the cardiovascular system during sepsis, leading to cell ultrastructural alterations and organ dysfunction (2, 3). The cardiovascular system is frequently involved in sepsis and is always affected by septic shock (Fig. 1). Cardiovascular dysfunction in sepsis is associated with a significantly increased mortality rate of 70% to 90% compared with 20% in patients without cardiovascular impairment (4). Myocardial dysfunction in sepsis has been the focus of intense research. Many mediators and pathways (Fig. 1) have been implicated in the pathogenesis of septic myocardial depression; however, the precise etiopathogenesis is unclear (5). Calvin and colleagues (4) were the first to demonstrate myocardial dysfunction in adequately volume-resuscitated septic patients with a decreased ejection fraction and an increased end-diastolic volume index. Echocardiographic studies have demonstrated impaired left ventricular systolic and diastolic function in septic patients (6, 7). Many other studies have confirmed decreased contractility and impaired myocardial compliance in sepsis (8-9-10-11).
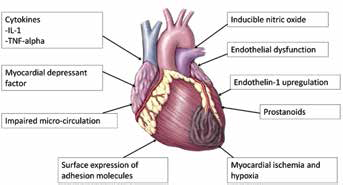
Pathogenesis of cardiac dysfunction in sepsis.
Septic cardiac dysfunction is multifactorial. Like septic acute kidney injury (AKI), ischemia and inflammatory mediators are the chief culprits. Global myocardial ischemia was postulated initially as a the main mechanism of cardiac dysfunction, but later septic patients were shown to have high coronary blood flow and diminished coronary artery–coronary sinus oxygen difference (12). Further experiments suggested a possibility of myocardial hypoxia due to alterations in coronary blood flow and myocardial metabolism as a possible mechanism of cardiac dysfunction (13). In patients with underlying coronary artery disease, myocardial ischemia is aggravated (14).
Inflammatory mediators also play a key role in the pathogenesis of cardiac dysfunction. The tumor necrosis factor (TNF) and interleukin-1 (IL-1) are the principal culprits (15, 16). Elevated levels of prostanoids, such as thromboxane and prostacyclin, which may alter coronary autoregulation and endothelial function, have also been demonstrated in septic patients (17). One of these cytokines may also act as a myocardial depressant factor.
Nitric oxide (NO) has important biological role in cardiovascular system. Higher dose of NO has been demonstrated to induce myocardial dysfunction by depressing energy generation (18). Sepsis leads to the expression of inducible nitric oxide synthase (iNOS) in the myocardium, which, in turn, importantly lead to myocardial dysfunction (19, 20).
AKI is a common complication of patients with sepsis and carries a poor prognosis. It occurs in 20% of critically ill patients and in 51% of patients with septic shock and those with positive blood cultures (21). The mortality rate of sepsis-induced AKI is high at approximately 70%, whereas the mortality of AKI alone is 40%-45% (22, 23). Although the presence of multiple organ dysfunction and other co-morbidities contributes to high mortality, AKI independently increases morbidity and mortality (24). Sepsis is characterized by a generalized inflammatory response and by the activation of coagulation and fibrinolytic system, resulting in endothelial injury (25, 26). Current opinion suggests that the pathogenesis of septic AKI relies on hemodynamic factors and inflammatory mediators (Fig. 2). AKI in sepsis was earlier considered to be secondary to renal ischemia due to septic shock. Experimental studies of septic AKI have reported conflicting results (27). On one hand, some studies showed that global renal blood flow (RBF) declines after the induction of sepsis or endotoxemia, leading to acute tubular necrosis, a reduction in glomerular filtration, and severe AKI (28, 29). On the other hand, Ravikant and Lucas (30) demonstrated renal vasodilation with increased RBF. A meta-analysis of 160 experimental sepsis studies found preserved or increased RBF in about 30% those studies (31). Changes in intrarenal hemodynamics also play a role in the pathogenesis of septic AKI. The RBF may be preferentially redistributed to the cortex, causing a relative hypoxia of the medulla (32).
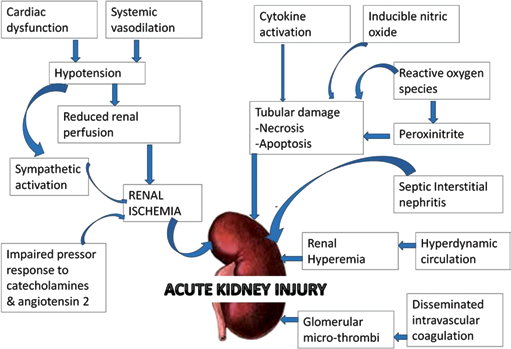
Pathogenesis of acute kidney injury in sepsis.
Nonhemodynamic kidney injury is mediated by various inflammatory mediators, such as cytokines, arachidonate metabolites, and vasoactive and thrombogenic agents. These various mediators are involved in the pathogenesis of organ dysfunction in sepsis (33). Among the variety of mediators, TNF seems to have the predominant role in septic AKI (34). Apoptosis seems to be an important pathway of cell dysfunction in sepsis rather than necrosis. All in all, there is a recent paradigm shift in understanding about the pathogenesis of septic AKI from ischemia and vasoconstriction to hyperemia and vasodilation, and from acute tubular necrosis to acute tubular apoptosis.
Sepsis also affects central structures or pathways, including the autonomic nervous system (ANS), the renin-angiotensin-aldosterone system (RAAS) and the hypothalamus-pituitary gland-adrenal gland axis (HPA), impacting cardiac and/or renal function. Sepsis causes ANS dysfunction (35), as pointed out by a change in heart rate variability (HRV) associated with the release of inflammatory mediators (e.g., IL-6, IL-10, and C-reactive protein [CRP]). Data with respect to kidney-related changes in ANS during sepsis are limited to animal studies. Here, sepsis-induced changes in renal sympathetic nerve activity did not seem to affect renal blood flow (36).
Sepsis activates the RAAS, reflecting the body's attempt to restore and maintain a sufficient blood pressure. Recent limited clinical data suggest that a blockade of the RAAS might be beneficial, as RAAS activation has also been implicated in endothelial dysfunction (37). Experimental studies also suggest deleterious effects of RAAS activation on renal function during sepsis (38). The administration of ACE inhibitors improves creatinine clearance and urine output during experimental bacteremia; the application of the selective angiotensin II type 1 receptor antagonist improves renal blood flow and oxygenation during experimental endotoxemia (38) (Fig. 2).
Finally, sepsis causes complex alterations of HPA and glucocorticoid signaling, leading to severe adrenal insufficiency in some patients. As a consequence, an increased production of pro-inflammatory cytokines, free radicals, and prostaglandins, as well as the inhibition of chemotaxis and the expression of adhesion molecules occurs. The administration of moderate-dose glucocorticoids for 7 days can exert positive effects, reducing the need for vasopressors and intensive care unit (ICU) assistance (39).
CRS-5 and Amyloidosis
The systemic amyloidoses are an uncommon group of disorders characterized by the extracellular deposition of amyloid in 1 or more organs. Cardiac and renal deposition leading to restrictive cardiomyopathy and proteinuric renal disease is a common feature of amyloidosis. Importantly, the presence and severity of CRS drives the prognosis of systemic amyloidosis.
Among many types of amyloidoses, AL (primary) and AA (secondary) amyloidosis are the most frequently encountered types in clinical practice. AL amyloidosis, in which amyloid is derived from monoclonal light chains, is associated with clinical cardiac involvement in about 50% of all cases (40). Subclinical cardiac involvement at autopsy or on endomyocardial biopsy may be detected in almost all patients. Renal involvement occurs in 30%-40% of all AL cases (41). In contrast, the AA type is characterized by predominant renal involvement in 60%-100% of all cases (42-43-44-45). Cardiac involvement is less frequent and varies from 0% to 39.5% (42-43-44-45).
In amyloidosis, the heart demonstrates thickening of all 4 chambers, with biatrial dilation, mild dilation of the right ventricle with a normal or small left ventricular cavity. Myocardial cells are separated by amyloid deposits with the infiltration of intramyocardial vessels. Occasionally epicardial coronary vessels are also involved leading to myocardial ischemia (46). The conduction system is frequently involved. The predominant manifestation of amyloid heart disease is congestive heart failure. In patients with small vessel involvement and minimal or no myocardial infiltration, the presenting complaint may be angina. In addition, atrial arrhythmias are frequently seen (40).
Renal amyloid is characterized by deposits in the glomerular basement membrane, the subendothelial area, and the extracellular mesangial system. Occasionally, tubular deposits are seen. The majority of patients with renal amyloidosis present with proteinuria, which can vary from minimal asymptomatic proteinuria to nephrotic syndrome. Hematuria is present in about one-third of patients. Chronic renal insufficiency with little proteinuria can also be seen in patients with extensive vascular deposits (47). In patients with tubular deposits, tubular dysfunction can be seen.
CRS-5 and Systemic Lupus Erythematosus
Heart is very commonly involved in systemic lupus erythematosus (SLE). Any cardiac structure, including the pericardium, myocardium, endocardium, conduction tissue, and even coronary arteries, are involved in SLE.
The spectrum of cardiac complications in SLE is shown in Figure 3. Pericarditis is the most frequent cardiac manifestation of SLE, and pericardial involvement is seen in 11%-54% of patients on echocardiographic studies (48). Pericarditis is also included in the American Rheumatolgy Association/American College of Reumathology (ARA/ACR) classification criteria of SLE (49). Direct immunofluorescence shows the granular deposition of immunoglobulin and C3. It indicates the role of immune complexes in the pathogenesis. Acute or chronic inflammatory changes are seen in the pericardium. Acute pericarditis can be fibrinous or serofibrinous, and chronic pericarditis can be fibrous or fibrofibrinous. Pericarditis generally manifests at the start of the disease or during relapses, and rarely leads to cardiac tamponade, constrictive pericarditis or purulent pericarditis.
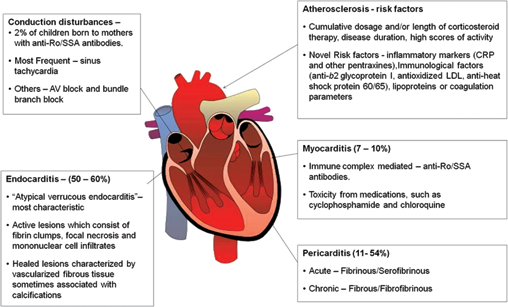
Pathogenesis and manifestation of cardiac dysfunction in systemic lupus erythematosus.
Myocardial involvement was seen in 40% of SLE cases in postmortem examinations (50) and in 20% of cases on echocardiography (51). However, myocardial involvement is seen in only 7%-10% of patients (52). Immune complex and complement deposition is seen on direct immunofluorescence, whereas an association with anti-Ro/SSA antibodies is also proposed (53). A patient may present with acute illness or have a chronic course with the development of cardiomyopathy, but left ventricular failure is rarely seen (54). Myocardial dysfunction in SLE may also be due to renal failure and hypertension, coronary artery disease (CAD), vulvular affection, or the toxic effects of medications used for treatment of SLE.
Libman–Sacks endocarditis (also known as atypical verrucous endocarditis) is the most typical presentation of endocardial involvement in SLE. These vulvular abnormalities are detected in 40%-50% of cases with transthoracic echocardiography and 50%-60% with transesophageal echocardiography. Anti-phospholipid antibodies bind to endothelial cells and activate them. This leads to platelet aggregation and thrombus formation (55). Immune-complex and complement deposition also have been reported to have an association with vulvular involvement.
Libman–Sacks endocarditis is clinically silent in the majority of patients and rarely leads to the development of a cardiac murmur. Verrucae develop near the edge of the valve and even if they become large they do not deform the closing line of the valves (56). Endocardial involvement may lead to vulvular insufficiencies, most commonly of the mitral or aortic valves. Although complications are rare, embolic events do occur, and stroke and peripheral embolism have been reported in 13% of cases. Infectious endocarditis has been reported in 7% of cases, and the risk of endocarditis is increased by dental treatments. Antibiotic therapy should be considered for patients with vulvular abnormalities, as SLE patients may receive immunosuppressant therapy for their primary disease.
While patients with SLE live longer, due to improved therapies and preventive measures, death and disability from cardiovascular events are increasing. SLE patients are 4-8 times more likely to suffer from CAD than non-SLE patients; this is seen in 6%-10% of SLE patients (57), and women are 50 times more at risk of CAD (58). Atherosclerosis, hypertension, arteritis, a thrombotic event, an embolism due to endocarditis or vasospasm are the risk factors for the development of CAD (59).
Hypertension, sedentary lifestyle, hyperlipidemia and hyperhomocysteinemia may lead to atherosclerosis in SLE patients (60). Steroid therapy in these patients increases the lipoprotein and homocysteine levels (61). Inflammation plays an important role in the development of atherosclerotic plaque. Atherosclerotic lesions begin with the recruitment of inflammatory cells, such as monocytes and T cells to the endothelial wall. Recently, CRP and pentraxines have been considered to be inflammatory markers in patients with SLE (62). Autoantibodies and immune complexes also play a major role for atherosclerosis. Circulating antibodies to oxidized low-density lipoprotein (anti-OxLDL) have been described, though their relationship to the development and progression of atherosclerosis is unclear. Svenungsson et al (63) have demonstrated that autoantibodies to OxLDL are more common in SLE patients who have a history of cardiovascular disease than in SLE controls or normal subjects.
Sinus tachycardia is the most frequent rhythm disturbance observed in SLE patients. Atrioventricular block and bundle branch block are seen in children of mothers with anti-Ro/SSA antibodies, but rarely in adults (64). These patients are mostly asymptomatic or may have fatigue and palpitations. Syncope is seen in very rare cases (65). Sinus tachycardia in SLE patients may be due to pericarditis, myocarditis, or chloroquine use (49).
Renal involvement remains a major cause of morbidity in patients with SLE. Abnormalities of immune regulation lead to auto-antibody production in SLE. Antibodies directed against nuclear antigens and specifically against DNA (anti-dsDNA) are considered diagnostic of SLE. Among these, anti-Sm antibodies have a significant association with lupus nephritis. The initiating event may be the local binding of nuclear or other antigens to glomerular sites followed by in situ immune complex deposition. Immune complexes made up of DNA-anti-DNA along with some other aggregates (nucleosomes, ribosomes, chromatin, C1q, laminin, Sm, La [SS-B], Ro [SS-A], and ubiquitin) cause glomerular injury. Previously, T cells were considered only as a helping factor for B cells to produce auto-antibodies, but recent studies support the significant role of T cells for the progression of renal disease in SLE. Additionally, deposition of the immune complex leads to the release of chemokines, such as MCP 1 and RANTES in glomeruli. These chemokines cause a proliferation of mesangium, which results in acute glomerular nephritis characterized by mesangial expansion and cellular infiltration. With the progress of disease, acute glomerulonephritis turns into chronic glomerulonephritis characterized by glomerulosclerosis, interstitial fibrosis, and tubular atrophy. Recent studies have been done on toll-like receptors (TLR), and TLR expression on renal cells causes the activation of end-organ response and renal injury.
Females are more commonly affected by SLE, but clinical manifestations are similar in both genders whether adults or children. SLE is a multisystem disease, and any organ system can be involved in SLE. Kidneys are affected from the start of SLE, or at any stage, and follow a protracted course of remissions and exacerbations. Clinical renal involvement correlates well with a degree of glomerular involvement (66). The clinical features of renal involvement may be correlated with histologic findings seen on renal biopsy and were classified by the International Society of Nephrology/Renal Pathology Society (ISN/RPS) in 2003 (Tab. II) (67).
International Society of Nephrology/Renal Pathology Society (ISN/RPS) 2003 classification of lupus nephritis
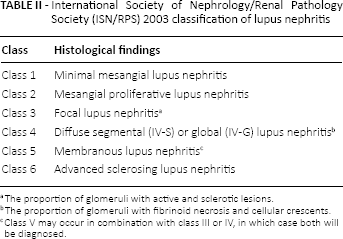
The proportion of glomeruli with active and sclerotic lesions.
The proportion of glomeruli with fibrinoid necrosis and cellular crescents.
Class V may occur in combination with class III or IV, in which case both will be diagnosed.
Patients of class I SLE, having only mesangial involvement, often have no or at the most, mild evidence of clinical renal disease. Patients of class II have proteinuria of less than 1 g/day. But these patients have high anti-DNA antibody titer and low serum complement. Hypertension is infrequently seen and serum creatinine, glomerular filtration rate (GFR) remains in the normal range.
In class III patients, proteinuria is often more than 1 g/day and many patients present with nephrotic range proteinuria. Most of the patients suffer from hypertension and have elevated creatinine at the presentation. Serologic tests usually indicate active lupus disease at this stage.
Patients of diffuse lupus nephritis (class IV) present with extensive clinical features. Almost all patients have proteinuria and half of these patients fall in the nephritic range. Hypertension is very common and renal dysfunction is typical. These patients have very high titers of anti-DNA antibody and low complement levels.
Patients with membranous lupus nephritis (class V) usually present with proteinuria, edema, and other typical nephrotic syndrome features. Out of these, 40% of patients will have less than 3 g/day proteinuria and up to 60% of patients will have elevated anti-DNA antibody titers and low serum complement levels. Usually these patients present with hypertension and renal dysfunction. Patients of this class are likely to develop thrombotic complications as seen in idiopathic membranous nephropathy.
Patients end up in class VI after long periods of disease's flares alternating with periods of inactivity. Patients will have inactive sclerotic and fibrotic lesions. Almost all patients have hypertension and renal dysfunction. But anti-DNA antibody titers and serum complement levels may be normalized by the time patients reaches this stage (67).
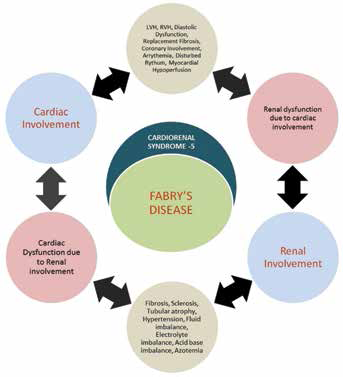
CRS-5 and Fabry Disease
Fabry disease is responsible for CRS-5 with insidious onset where the kidney and cardiac dysfunction may develop slowly until a “point of decompensation.” It can also be chronic, acute, or acute-on-chronic CRS-5. Mechanisms in acute and chronic CRS-5 are different; the nature, severity, and duration of organ dysfunction are also influenced by the management interventions. In most cases of CRS-5 there is usually a precipitating event that brings the condition to attention; for example, Fabry crises, precipitated by fever, exercise, fatigue, stress, and rapid changes in temperature (68, 69).
Being a systemic disease, Fabry disease starts with a specific effect(s) involving the kidney and/or the heart, contributing to the bilateral organ crosstalk for the development of CRS-5.
Pathology of Renal Involvement
The natural course of Fabry nephropathy in children or adolescent patients is still largely not understood. Like most aspects of the disease, renal pathology increases in severity with age. In classically affected Fabry patients, renal lesions result from Gb3 deposition in the glomerular endothelial, mesangial, intersticial cells, and in podocytes, which are terminally-differentiated epithelial cells that accumulate numerous myelin-like inclusions in their lysosomes. Podocyte foot process effacement has been described and it represents the histological counterpart of proteinuria. Glycosphingolipid storage also occurs in the epithelium of the loop of Henle and the distal tubules, and in the endothelial and smooth muscle cells of the renal arterioles (69, 70). Histologic, potentially irreversible changes to glomeruli, interstitial tubules, and vascular structures before the first appearance of signs can be observed in renal biopsy specimens from children (71) (Fig. 4). The glomerular podocytes are swollen and finely vacuolated in a light microscopy examination, such as the epithelial cells of distal tubules (Fig. 5); lamellated lipid inclusions (zebra bodies) in podocytes’ cytoplasm also can be seen on electron microscopy.
Fabry disease pathophysiology.
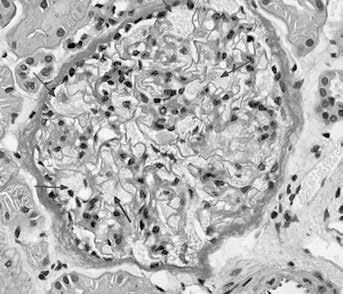
Light microscopy. The glomerular podocytes are swollen and finely vacuolated (arrows) in a patient with Fabry nephropathy disease.
Clinical Renal Involvement
Signs indicative of early, insidiously progressing renal damage include microalbuminuria and proteinuria developing as early as the second decade of life, which, like in diabetic nephropathy, are believed to directly contribute to the progression of Fabry nephropathy. With advancing age, proteinuria worsens (72). Isosthenuria accompanied by alterations in tubular reabsorption, secretion and excretion develop. Initially, glomerular compensation (hyperfiltration) may mask impairment of renal function but, once a critical number of nephrons have been damaged, renal function will progressively decline. The gradual deterioration of renal function and the development of azotemia usually occur in the third to fifth decades of life (73). At this stage, fibrosis, sclerosis, and tubular atrophy dominate the disease activity portending end-stage renal disease that generally occurs in males in the fourth to fifth decade of life. The nephrological aspects of Fabry disease are major contributors to the morbidity and mortality associated with the disorder. Progression to end-stage renal failure is the primary cause of death in male patients with untreated Fabry disease, and death most often results from uremia, unless chronic hemodialysis or renal transplantation is undertaken (Fig. 4).
Pathology of Cardiac Involvement
Storage of globotriaosylceramide (Gb3) is found in various cells of the heart, including cardiomyocytes, conduction system cells, valvular fibroblasts, endothelial cells within all types of vessels, and vascular smooth muscle cells (74). However, Gb3 storage by itself is unable to explain the observed level of cardiac manifestations. An autopsy of an individual with Fabry disease who had an extremely hypertrophied heart revealed a relatively limited contribution (1%-2%) of the stored material to the enormous increase in cardiac mass. It appears that storage induces progressive lysosomal and cellular malfunctioning that, in turn, activates common signalling pathways. Energy depletion was recently proposed as the common denominator in multiple metabolic and even sarcomeric hypertrophic cardiomyopathies (75) (Fig. 6). Energy depletion may also occur in Fabry disease, as suggested by the impairment in energy handling seen in skin fibroblasts. This might be further supported by the observation of a decreased ratio of adenosyl triphosphate (ATP) to inorganic orthophosphate, as has been shown by magnetic resonance imaging studies in patients with sarcomeric hypertrophic cardiomyopathies (76) (Fig. 4).
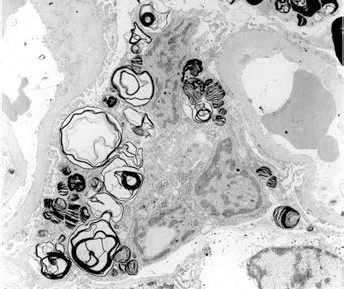
Electron microscopy. Lamellated lipid inclusions (zebra bodies) in a podocyte cytoplasma.
Clinical Cardiac Involvement
Cardiac symptoms including left ventricular hypertrophy, arrhythmia, angina, and dyspnea are reported in approximately 40%-60% of patients with Fabry disease (77). Arrhythmias and impaired heart rate variability arise from the involvement of the sinus node, the conduction system, and an imbalance between sympathetic and parasympathetic tone. Diastolic dysfunction and concentric left ventricular hypertrophy, which is typically nonobstructive, are important features, with men generally more severely affected than women. Myocardial ischemia and infarction may result from compromised function of the coronary vascular bed (78). With age, progressive myocardial fibrosis develops with both interstitial and replacement fibrosis (79). Replacement fibrosis almost always starts in the posterior-lateral wall and in the mid-myocardium. In end-stage patients, transmural replacement fibrosis gradually reduces cardiac function to the stage of congestive heart failure (80). Malignant arrhythmias are responsible for a number of cardiac deaths in patients affected with Fabry disease (80). The cardiomyopathy of Fabry disease is characterized by reduced myocardial contraction and relaxation tissue Doppler velocities are sometimes detectable even before the development of left ventricular hypertrophy. Right ventricular hypertrophy with normal chamber size and preserved systolic but impaired diastolic function represents the typical right ventricular structural change in Fabry disease. The myocardial perfusion reserve was found to be significantly reduced in patients affected with Fabry disease (81). Patients with Fabry disease have abnormal coronary microvascular function. Fabry disease is associated with an increased risk of developing aortic root dilatation in male patients (82). Aortic root dilation was detected in 24% of 71 hemizygous male patients, and was statistically associated with the presence of a dolicho-ectatic basilar artery (p = 0.008) (Germain D.P.,) (82).
Diagnosis of CRS-5
For a diagnostic approach to sepsis (a prototype of type-5 CRS), the initial emphasis needs to be on the setting of the severe sepsis and septic shock, then on the heart and kidney assessment, and finally a risk evaluation to start the appropriate treatment.
Systemic inflammation, like sepsis, has to be suspected when the body temperature is less than 36°C (96.8°F) or greater than 38°C (100.4°F); the heart rate is greater than 90 beats/minute; and tachypnea is already present (more than 20 breaths/minute). The white blood cell count can be less than 4 × 100 cells/L or greater than 12 × 100 cells/L.
A recent review pointed out some characteristic biomarkers whose elevation is typical during the septic process: lipopolysaccharide binding protein, pro-calcitonin, CRP, and pro-inflammatory cytokines (IL-6, TGF-β) (83).
The assessment of cardiac function in type-5 CRS is quite similar to other clinical situations in which myocardial dysfunction is present. Natriuretic peptides and troponins level assays provide information about cardiac chambers (especially left cardiac chambers) and myocardial cell damage. Leukocytosis and CRP are not specific for myocardial injury diagnosis and imaging devices are preferred by clinicians.
Sepsis cardiomyopathy presents a complex clinical picture and its pathophysiology is not well understood at all. In the early stages of the septic process, there is low output myocardial involvement. After starting fluid therapy, the clinical picture shifts to typical distributive shock characterized by increased cardiac output and systemic vasodilatation (84). An echocardiographic assay can confirm high output cardiomyopathy with abnormalities in the left ventricular regional contractility together with the dilation of the left heart chambers (85).
The diagnosis of kidney involvement in sepsis related to type-5 CRS overlaps to other forms of AKI with acute changes in serum creatinine levels, according to RIFLE, AKIN, and KDIGO criteria (86).
At the present time, several other biomarkers are proposed, such as cystatin C (the only new biomarker approved in the USA), KIM-1, NGAL, and NAG; however, RIFLE, KDIGO, and AKIN criteria still recommend serum creatinine levels and urine output for the diagnosis and monitoring of AKI in type-5 CRS.
Management of CRS-5
Once the diagnosis of type-5 CRS is made, every organ and tissue involved must be investigated to pay attention to risk prediction and to protect it from further and irreversible alterations in organ function.
Preliminary data (not published at the present time) seem to indicate that biomarkers of cell cycle regulation may be able to predict which patients will develop severe AKI in few days.
Regarding cardiac risk, patients who survive to septic shock are shown to have lower ejection fractions and higher left ventricular end-diastolic volumes, which suggests a protective role of myocardial depression (87).
The treatment of type-5 CRS is mainly based on underlying disease management and kidney and heart complications.
Maintaining hemodynamic stability and guaranteeing tissue perfusion are key points in the prevention of type-5 CRS in the hyperacute phase of sepsis, together with fluid control and correct antibiotic treatment. Fluid therapy must be carefully managed to avoid fluid overload and other iatrogenic complications (88).
Since inflammation and immune disorders play an important role in the pathogenesis of sepsis, the removal of cytokines and immunomodulation are 2 approaches based on extracorporeal techniques utilizing convection, high-volume hemofiltration, and high-permeability membranes (89). The best results were obtained with high-permeability membranes and absorption (89).
A therapeutic alternative is provided by hit cellular elements accountable for apoptosis and neutrophil activation and removed by polymyxin filters or a citrate anticoagulant-based selective cytopheretic device (90, 91).
To manage heart complications, especially in the hyperacute stage, a multipronged approach is required to maintain filling pressures with fluid therapy, together with vasopressors, vasodilators, and inotropes. The vasopressors should be carefully employed because of depressive effects on cardiac output (increased afterload) especially with concomitant hypovolemia. Vasodilators increase cardiac output, especially in ischemic patients, while phosphodiesterase inhibitors have inotropic and vasodilatory effects, but they provide less increase of myocardial oxygen requirements.
Vasopressin increases arterial pressure, but it has negative effects on cardiac output. More recently levosimendan has been proven to provide benefits in decompensated heart failure to increase ejection fraction and diuresis. Levosimendan efficacy is still to be proven in the prevention of type-5 CRS (92).
Renal support includes the removal of any nephrotoxic drug and media, the maintenance of adequate perfusion pressure and, if indicated, early intervention with dialysis therapy (92, 93).
There is no role for dopamine for improving renal hemodynamics (94) and there are limited studies with fenoldopam (95). Norepinephrine decreases renal perfusion in normal conditions, but increases systemic blood pressure in septic patients (89), while vasopressin increases diuresis and the GFR in septic patients (96).
Diuretics have a limited role in managing heart and kidney involvement in septic patients (97). Renal replacement therapy with continuous renal replacement therapy should be promptly started (94). Early ultrafiltration seems to improve renal outcomes in septic shock patients. However, all of these data need to be confirmed in further clinical trials.
Footnotes
Financial support: No grants or funding have been received for this study.
Conflicts of interest: None of the authors has financial interest related to this study to disclose.
