Abstract
Management of complicated vascular access. A step-by-step description of a case of hyperkalemia and vascular access failure in a patient receiving maintenance hemodialysis treated for numerous previous stenosis and thrombosis of the native vascular access. The discussion revolves around the following key questions:
Basilic vein thrombosis and re-stenosis, what do you think it should be done after this complication? Venous back bleeding was satisfactory; do you think thrombectomy was effective? We thought that central vein stenosis stenting was needed, wasn't it? What do you think about basilic stenosis? The patient had undergone multiple recent basilic angioplasties. Consequently, a surgical intervention could have been definitive. Various surgical options were possible. But first of all, we had to solve a dilemma: The patient had received multiple previous central vascular catheters; our aim was to avoid a new central vascular access placement. At the same time, we required a vascular access for prompt cannulation. Do you think that was a real problem? Do you agree with us, or in the present circumstance, would you have preferred a central venous device? Finally, we decided in favor of creation of a prosthetic vascular access. We chose a graftspecifically designed for early cannulation. Do you agree with us? Do you think early cannulation could cause a worse vascular access outcome? Patient underwent last hemodialysis (HD) 24 hours before surgery. Consequently we trusted that the next HD could be delayed for at least 24 hours. Unfortunately, intraoperative arterial blood gas analysis documented hyperkalemia and acidosis. Our nephrologist suggested HD. In your opinion what does “early” mean?.
Introduction
Autogenous vascular access (aVA) is recommended as the first choice for the delivery of hemodialysis (HD) (1–3). The number of patients suffering end-stage kidney failure is increasing, and the majority of them have a late diagnosis, with HD initiation via central venous catheter (CVC) (1). The introduction of a graft specifically designed for early cannulation has made prosthetic vascular access (pVA) a safe alternative to CVC, also in emergencies (4, 5). In this report, we present a case of immediate cannulation of pVA.
Case report
A 63-year-old man presented with a history of hypertension, dyslipidemia, overweight (BMI 29 kg/m2), myocardial infarction treated with percutaneous transluminal coronary angioplasty, segmentectomy for liver injury (grade 5 according to the American Association for the Surgery of Trauma liver injury grading system) and chronic glomerulonephritis.
Seven years previously the patient was admitted to the emergency department for end-stage kidney failure. He was unknown to the nephrologist service at that time and received a temporary CVC in his right internal jugular vein. Urgent HD was carried out through it. Soon after, a radiocephalic fistula (Brescia-Cimino) was placed in his left forearm, but cephalic vein maturation did not occur. Therefore, a tunneled CVC (Tesio Caths®; MedCOMP, Harleysville, PA, USA) was placed in his right internal jugular vein, and the patient was submitted to the proximalization of arteriovenous fistula inflow with a Gracz fistula. Adequate cephalic vein maturation was documented 3 months later, and after 2 months, the CVC was removed. Unfortunately, 4 years later, diffuse cephalic vein stenosis was observed and treated 3 times with percutaneous transluminal angioplasty (PTA). Subsequent to cephalic vein thrombosis, a temporary CVC was placed in his internal right jugular vein. The patient was submitted to basilic vein transposition and humeral artery-to-basilic vein fistula. Three weeks later, the basilic vein was cannulated, and the CVC was removed soon afterwards. Two years later, an ultrasonography examination documented a critical stenosis between the basilic and axillary vein. Consequently, the patient underwent basilic and axillary vein PTA, but the procedure was demonstrated not to be effective. The patient was referred to our center after basilic vein thrombosis occurred 6 months later during HD.
What do you think it should be done after this complication?
The patient underwent preoperative surgical evaluation with physical examination and echo-color Doppler evaluation before surgical exploration of the fistula. The patient received short-term antibiotic prophylaxis with vancomycin (1 g) at induction and regional blockade anesthesia. A previous anastomosis between the humeral artery and basilic vein was exposed through a 4-cm longitudinal skin incision at the level of the cutaneous projection of the fistula. Intravenous heparinization at the low standard dose of 1,250 IU was administered just before a bulldog clamp was placed. Thrombectomy of the basilic vein was performed with a Fogarty balloon catheter.
Venous back bleeding was satisfactory; do you think thrombectomy was effective?
The operating theater was equipped with a mobile C-arm. Completion angiography documented the presence of diffuse basilic stenosis and a subocclusive subclavian vein stenosis.
We thought that central vein stenosis stenting was needed, wasn't it?
Subclavian vein stenting was performed (Viabahn® 7 × 10 cm; W.L. Gore, Flagstaff, AZ, USA).
What do you think about basilic stenosis? The patient had undergone multiple recent basilic angioplasties. Consequently, a surgical intervention could have been definitive. Various surgical options were possible. But first of all, we had to solve a dilemma: The patient had received multiple previous central vascular catheters; our aim was to avoid a new central vascular access placement. At the same time, we required a vascular access for prompt cannulation. Do you think that was a real problem? Do you agree with us, or in the present circumstance, would you have preferred a central venous device?
In our opinion, CVCs should not be considered as a bridge crossing the fistula maturation whirlpool. Reports in the literature largely recognize that central vein cannulation is associated with a higher risk of infection, central vein thrombosis and death (5). However, we deny any “witch hunt” against use of CVC. Nevertheless they should be limited to selected patients with severe cardiopathy and reduced ejection fraction, pediatric patients weighing less than 20 kg and very elderly patients in poor clinical condition with short life expectancy or who affected by neoplasms (6).
Finally, we decided in favor of creation of a prosthetic vascular access. We chose a graft specifically designed for early cannulation. Do you agree with us? Do you think early cannulation could cause a worse vascular access outcome?
As noted in a recent review, there is no evidence that early cannulation affects polytetrafluoroethylene (PTFE) graft survival and patency, especially with new-generation prostheses (7). In our experience with early cannulation grafts, we have not documented any relationship between the time of first cannulation and incidence of thrombosis. Instead, we have noticed in our everyday experience a higher incidence of hematomas associated with earlier cannulation, and therefore we suggest an ultrasound-guided cannulation to avoid this complication (8).
Patient underwent last HD 24 hours before surgery. Consequently we trusted that the next HD could be delayed for at least 24 hours. Unfortunately, intraoperative arterial blood gas analysis documented hyperkalemia and acidosis. Our nephrologist suggested HD. What does “early” mean?
After the introduction in the early 1990s of specifically designed grafts, the concept of early cannulation has changed. In fact, while at the beginning, early cannulation was permitted by technical expediency, new prostheses permit a safer and more effective cannulation (6). Data collected in Table I refer to 329 patients treated between 2011 and 2015 with 3 different grafts specially designed for early cannulation (4, 5, 8,9–13). Median time of first cannulation was 48 hours (interquartile range [IQR] 37–57 hours). Median time of the earliest cannulation was 24 hours (IQR 21–24 hours). Particularly noteworthy is that only 2 articles have reported a first cannulation performed earlier than the median time of 24 hours (4,10). The Acuseal instructions for use do not report a minimum time required for first cannulation.
Five-year literature review on early cannulation of pVA
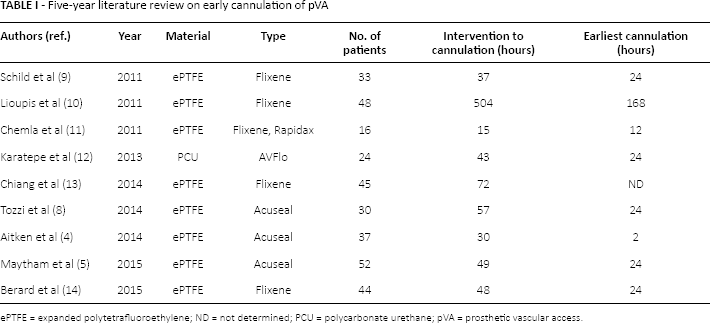
ePTFE = expanded polytetrafluoroethylene; ND = not determined; PCU = polycarbonate urethane; pVA = prosthetic vascular access.
Intervention and postoperative management
The patient's fistula was ligated. The axillary vein was exposed through a 5-cm longitudinal skin incision at the level of the cutaneous projection of the targeted vessel. The graft (Acuseal® 6 mm; W.L. Gore, Flagstaff, AZ, USA) was positioned in the subcutaneous tissue, describing an arm loop pattern using a specific 6-mm tunneler. The pVA was constructed between the humeral artery and axillary vein. The vein was gently dilated with a saline solution, and the anastomosis carried out with 2 expanded PTFE (ePTFE) 7/0 running sutures (Gore-Tex®; W.L Gore, Flagstaff, AZ, USA). Time of the intervention was 80 minutes. Blood loss was 60 mL Intraoperative quality control was performed at the end of the intervention with echo-color Doppler echocardiography. A short-stretch bandage was placed to reduce immediate postoperative edema. Postoperatively, the patient was immediately moved to the Nephrology Department, where HD was properly conducted according to the Acuseal instructions for use, for 3 hours at a flow rate of 200 mL/min with two 15-gauge needles. HD was conducted 46 minutes after pVA implantation (Fig. 1). The patient was discharged the day after intervention. Hospitalization time was 32 hours. Major local or systemic morbidity was not observed. After discharge, patient started regular HD treatment 3 times a week. The patient also started dual antiplatelet therapy using clopidogrel bisulfate (Plavix®; Sanofi-Aventis SpA, Milan, Italy) 75 mg once daily, plus acetylsalicylic acid (Cardioaspirin®; Bayer SpA, Milan, Italy) 100 mg once daily, with the addition of fish oil capsules 4000 mg for 3 months after surgery and then Clopidogrel 75 mg once daily indefinitely. Follow-up was performed through monthly clinical evaluation and echo-color Doppler echocardiography evaluation of the pVA. Follow-up time was 217 days. Local complications such as access thrombosis, infection, hematomas or fluid collection have never been observed.
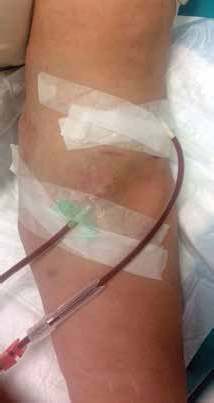
Prosthetic vascular access immediately cannulated after surgical creation. Accurate cannulation was mandatory due to presence of edema and hematoma.
Conclusion
Results from a single-center randomized controlled trial on early cannulation grafts versus tunnelled CVCs are expected (15). Nevertheless, what can be deduced from our case is that careful cannulation of pVAs immediately after their implantation is feasible and safe. This may be considered a suitable alternative to central vein catheterization whenever HD is urgently required.
Footnotes
Financial support: No grants or funding have been received for this study.
Conflict of interest: The authors declare that there is no conflict of interest regarding the publication of this paper.
