Abstract
Aims and background
Left ventricular hypertrophy (LVH) is a common finding in hemodialysis patients. The aim of the present study was to investigate if the diameter of the distal radiocephalic fistula could influence left ventricular variables in stable hemodialysis patients.
Methods
Nineteen patients were investigated. Measurements of the diameter of the arteriovenous (AV) fistula were performed in 4 different locations. The patients were investigated using M-mode recordings and measurements in the 2D image. Doppler ultrasound was also performed. Transonic measurements were performed after ultrasound investigation.
Results
Fistula mean and maximal diameter correlated with left ventricular characteristics. Fistula flow correlated neither with the left ventricular characteristics nor with fistula diameters.
Conclusions
The maximal diameter of the distal AV fistula seems to be a sensitive marker of LVH in stable hemodialysis patients.
Introduction
Left ventricular hypertrophy (LVH) is a common finding in hemodialysis (HD) patients. Several causes are considered to impair left ventricular volume and mass characteristics. Such causes can be anemia, systemic hypertension, atherosclerosis, sodium and water retention, and the uremic state itself.
Left ventricular hypertrophy is also a well-known risk factor for cardiovascular morbidity and mortality in patients with end stage renal disease (1–4). In addition, the creation of a vascular arteriovenous (AV) anastomosis results in an increase in workload on the heart that leads to increase of a preexisting LVH. This further increases cardiac output by the mechanism of increasing stroke volume, heart rate, and ejection fraction (EF), and decreasing systemic vascular resistance (5–8). The latter is classified as high output cardiac failure, owing to large brachiocephalic AV fistulas. A reversal of this state, of hemodynamic derangements, has been achieved after corrective surgery (9–11).
Investigations of fistula flow with ultrasound velocity dilution (Transonic Systems, Inc., Ithaca, NY) have shown a correlation between fistula flow and cardiac output in hemodialysis patients (12). In general, the evaluation of fistula flow is influenced by the flow velocity and the absence of laminar flow in most cases (13). Besides using fistula flow, ultrasound measurement of the venous diameter increased the predictive value of fistula adequacy from 33% to 95% having both a flow volume of 500 mL/min or greater and a minimum venous diameter of 0.4 cm or greater (13), indicating the benefit adding fistula diameter as a variable. In a previous study, we found differences between the AV fistula diameter and underlying disease (14). However, to our knowledge, no investigation has been performed using these measurements to investigate the possible correlation with cardiac characteristics.
The aim of the present study was to investigate whether the diameter of the AV fistula has an impact on left ventricular variables in stable hemodialysis patients.
Methods
Nineteen stable HD patients, without cardiac problems or valvular defects upon echocardiographic investigation, were identified at 3 dialysis centers in southern Sweden. All echocardiographic investigations were performed by one clinical physiology assistant. Inclusion criterion was presence of well-functioning native primary distal radiocephalic AV fistula used for HD. The fistulas had no known flow problems or stenoses. All AV fistulas had been created as side-to-side anastomoses. All patients had used their AV fistula for HD for a minimum of 1 month. All dialyses were performed using the double needle and buttonhole puncture technique described by Ball (15). No patient had a history of fistula corrective surgery, thrombotic episodes, or fistula stenosis. Two of the patients had a history of a minor myocardial infarction without congestive heart failure.
Body surface area (BSA) was defined as BSA = 1 + (kg body weight + cm height – 160)/100. Blood pressure was measured manually in a sitting position and as systolic blood pressure (SBP), diastolic blood pressure (DBP), and mean arterial pressure (MAP).
Patient weight used in the study was the one recorded after the dialysis sessions. Demographic data for study subjects are presented in Table I.
Demographic data

ACE = angiotensin-converting enzyme; BSA = body surface area.
The study was approved by the ethics committee of the University of Linköping, and all subjects gave written and verbal informed consent.
Ultrasound investigation of the AV fistula
All measurements were performed at Högland Hospital, Eksjö with the patient in a sitting position with the forearm on an investigation table using an Acuson Sequoia C256 (Acuson, Mountain View, CA, USA) at the Department of Radiology.
The examinations were all carried out by one skilled radiologist (U.H.) to exclude intraobserver variability. All investigations took place directly after a regular HD treatment session. The investigation technique was adapted to the study in terms of extra use of gel to avoid compression of the vessel. The entire AV fistula was investigated from the proximal forearm to the wrist to the thumb. The AV fistula was recognized by palpation and visualization of the feeding artery and the receiving vein and anastomosis area. The anastomosis itself was investigated longitudinally and transversally in order to identify the maximum diameter position. Thereafter, the arterial side of the AV fistula was investigated for at least 15 cm in the proximal flow direction up to and including the connection to the artery. Then, the intermediate and distal parts of the AV fistula were investigated in order to identify the maximum dilated area.
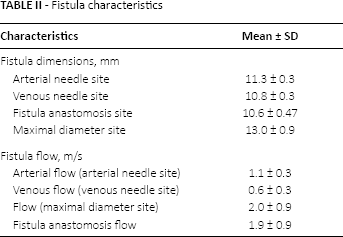
The diameter measurement in the needle insertion site took place after visual identification of the buttonhole sites. The diameter of the AV fistula was measured as maximal and at a mean of 3 locations in the arterial part (proximal of the arterial site of the access needle insertion site of the AV fistula), the venous part of the fistula (distal part of the fistula where the venous needle is placed), and the part between the needle sites. In every measurement site the procedure was repeated at least 3 times. The mean value was then determined as the value for the location. The probe was positioned at an angle of 90° in relation to the vessel longitudinal axis. Data of fistula characteristics are presented in Table II.
Fistula characteristics
Echocardiography
The investigations were performed by one experienced investigator (G.L.). All investigations took place within 2 hours after a regular HD session in order to reduce the influence of interdialytic fluid retention on the measurement results (16).
M-mode and 2D echocardiography
Both M-mode recordings and measurements were guided by the 2D image. M-mode measurements were made according to the recommendations of the American Society of Echocardiography (17). Each echocardiographic value represents the average of high-quality records of 3 cardiac cycles. Left ventricular hypertrophy was defined as left ventricular mass (LVM) index (LVMI) >134 g/m2 in men and >110 g/m2 in women and the left ventricular mass using the cube formula (18). The EF was calculated according to the method of Teichholz et al (19). The cardiac variables given from the investigations were as follows: left ventricular diameter in diastole (LVD), left ventricular diameter in systole (LVS), left ventricular end diastolic volume (LVEDV), left ventricular end systolic volume (LVESV), left ventricular systolic volume (LVSV), LVM, LVMI, and EF.
Doppler measurement
Blood flow velocity in the left ventricular outflow tract was estimated using pulsed wave Doppler from an apical 4–chamber view (sample size of 5 mm). Mitral flow was recorded between the mitral leaflets in the 4-chamber view. Continuous wave Doppler signals were recorded from the apical windows by a 4.0 MHz scanning probe. All measurements were performed with the patient in a left lateral position. Echocardiography was performed using an Acuson Sequoia C256. Data of Echocardiography characteristics are presented in Table III.
Echocardiography characteristics
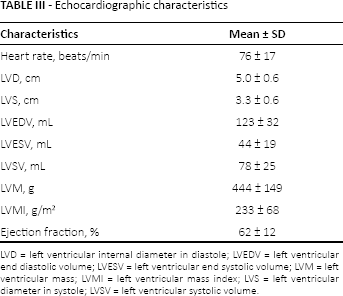
LVD = left ventricular internal diameter in diastole; LVEDV = left ventricular end diastolic volume; LVESV = left ventricular end systolic volume; LVM = left ventricular mass; LVMI = left ventricular mass index; LVS = left ventricular diameter in systole; LVSV = left ventricular systolic volume.
Transonic measurements
Measurements were performed within a week after ultrasound investigation of the fistula and echocardiography according to instructions by Transonic Systems Inc. (Ithaca, NY, USA). Data of Transonic measurements are presented in Table IV.
Transonic measurement

Statistics
All values in the study are expressed as mean (SD) or median (range) unless otherwise stated.
Two-tailed p values less than 0.05 were considered to be statistically significant. Mann-Whitney U test was used for nonparametric group comparisons. Bivariate correlation analyses were performed using the Spearman (ρ) test. Multiple regression was used for correlation calculations.
Multivariate analyses were performed using cardiac variables as dependent factors and the following as independent factors in the model: age, sex, body mass index (BMI), height, weight, BSA, use of β-blockers, AV fistula arterial and venous diameter, and AV fistula flow. SPSS software version 19.0 was used.
Results
The mean value of the AV fistula diameters correlated with LVD (ρ = 0.48, p = 0.037), LVS (ρ = 0.61, p = 0.006), LVEDV (ρ = 0.47, p = 0.041), LVESV (ρ = 0.59, p = 0.008), LVM (ρ = 0.46, p = 0.049), arterial diameter (ρ = 0.81, p< 0.001), venous diameter (ρ = 0.80, p< 0.001), maximal diameter (ρ = 0.97, p< 0.001), and diameter at the fistula anastomosis (FI) (ρ = 0.86, p< 0.001).
The diameter of the arterial part of the AV fistula correlated with LVS (ρ = 0.63, p = 0.004), LVESV (p = 0.60, p = 0.007), and inversely with the EF (ρ = −0.58, p = 0.01), to the maximum diameter (ρ = 0.82, p< 0.001), and also to FI (ρ = 0.56, p = 0.013). These data were similar to the vein diameter except for lack of correlation with the EF.
The maximum diameters of the AV fistulas correlated with LVD (ρ = 0.48, p = 0.037), LVS (ρ = 0.54, p = 0.017), LVEDV (ρ = 0.48, p = 0.04), LVESV (ρ = 0.53, p = 0.002), and FI (ρ = 0.80, p< 0.001). Mean fistula diameters did not correlate with mean fistula flow.
The ejection fraction (EF) correlated with dialysis time (ρ = 0.47, p = 0.041) and inversely with LVS (ρ = −0.59, p = 0.008), LVESV (ρ = −0.60, p = 0.007), LVSV (ρ = −0.51, p = 0.027), arterial AV fistula diameter (ρ = −0.58, p = 0.01), and fistula flow (ρ = 0.46, p = 0.049).
The blood flow in the AV fistula did not correlate with any of the variables included in the study.
Considering the age span of the patients, age as a variable only correlated, inversely, with mean fistula flow (ρ = −0.51, p = 0.027). Older patients had a lower flow and vice versa.
Blood hemoglobin level did not correlate with any of the variables.
The BMI of the patient correlated inversely with blood pressure (MAP: ρ = −0.68, p = 0.001; SBP: ρ = −0.65, p = 0.003; DBP: ρ = −0.65, p = 0.002).
The height of the patient did not correlate with the blood pressure. The height correlated with the cardiac variables LVD (ρ = 0.73, p< 0.001), LVS (ρ = 0.53, p = 0.021), LVEDV (ρ = 0.69, p = 0.001), LVESV (ρ = 0.48, p = 0.037), LVSV (ρ = 0.60, p = 0.007), and LVM (ρ = 0.69, p = 0.001).
Blood pressures (SBP, DBP, MAP) did not correlate with any of the cardiac variables. The heart rate was not correlated with cardiac variables but with mean flow (m/s) (p< 0.05), and inversely with weight (p< 0.05).
The dialysis time, measured as h/wk, correlated with EF (ρ = 0.47, p = 0.041), patient height (ρ = 0.52, p = 0.024), and BSA (ρ = 0.52, p = 0.024).
Regarding cardiac variables investigated, patients treated with β-blockers compared with those who were not treated with β-blockers had lower EF (p = 0.02) and larger LVDS (p = 0.019) and LVESV (p = 0.018). There was no difference in cardiac variables regarding patients taking or not taking α-blockers, Ca channel blockers, or diuretics (too few to analyze for angiotensin-converting enzyme inhibitors [2 patients]).
Multiple regression analyses, using variables described in the statistics, exhibited significance for the cardiac variables (1) LVS, with a model containing vein diameter, female sex, and lower blood flow (r 2 = 0.79, p< 0.014); (2) LVD, with a model containing patient height, vein diameter, and lower blood flow (r 2 = 0.78, p< 0.014); (3) LVEDV, with a model containing height of patient, vein diameter, and lower blood flow (r 2 = 0.75, p< 0.041); LVESV, with height of patient and vein diameter (r 2 = 0.64, p< 0.017); LVMI, with height of patient (r 2 = 0.25, p = 0.029); and EF, with arterial diameter (r 2 = 0.32, p = 0.012).
Discussion
The present study showed that the mean diameter and maximal diameter of the AV fistula, in patients with optimal HD conditions, was correlated with left ventricular variables. Unexpectedly, within the conditions of the study, blood flow in distal radiocephalic fistula did not correlate to left ventricular characteristics. In general, one would expect to see an impact of fistula flow on cardiac output and left ventricular dimensions. In fact, this would probably apply to a population with more heterogeneous flow characteristics, as found by others (5, 7, 9, 12, 20). Another explanation could be the difficulty of achieving adequate measurements of the flow, since flow measurements can only be made in locations with laminar flow and free from disturbance. A disturbance is not necessarily due to a stenotic formation; for example, there might be the presence of sclerotic plaque, inducing greater variance (13).
The present study indicates that when using the diameter of the AV fistula as a variable, instead of fistula flow, data correlated significantly with left ventricular characteristics of the heart in HD patients. Since we did not possess data on the patients' echocardiographic characteristics in the period before the creation of the AV fistula (before start of HD), we could not estimate the extent of change in the left ventricular variables of the heart during the HD. There is a strong correlation between the AV fistula diameter and cardiac variables. This indicates that a wider diameter is coupled with the development of a larger LVM, wider LVD in diastole and in systole, as well as LVEDV and LVESV. This also indicates a wider and stiffer heart. The presence of less elastic tissue is known to occur when glucose degradation products are deposited in tissue. A marker for such correlation and loss of elasticity is the presence of high values of skin autofluorescence (21) that has been found in dialysis patients (22). Thus a loss of elasticity could result in wider AV fistula diameters but also in a stiffer and less patent heart. In addition, it is known that patients in HD may have small silent cardiac ischemic events that make the left ventricle even more susceptible to the additional influence of the AV fistula (23). Notable was that, within the frame of the present study, age did not correlate to the cardiac variables but only inversely to the AV fistula flow. This could indicate that elderly patients have a lower fistula flow due to the presence of more narrow vessels and not due to impaired cardiac variables.
The BMI did not correlate with cardiac variables. However, height and BSA were correlated with these variables. This seems reasonable, since a larger patient would be expected to have a larger heart and greater cardiac capacity. This may also fit with the finding that LVD was increased. The latter would not indicate a negative marker.
Fistula maturation varies in different patients, and the adaptive cardiac mechanisms may vary over time. However, a steady state is expected to be present after approximately 6 weeks, once fistula maturation is reached (16). Locally, wall shear stress develops in the radial artery after surgical creation of an AV fistula. It is the tractive force that acts on the endothelium and influences endothelial cell function. The changes in blood flow induce vascular remodeling and this phenomenon seems to be correlated to the shear forces imposed on the vessel wall by motion of blood (24). Shear stress-induced microinjuries of the endothelium in hemodynamically compromised regions together with local coagulation activation associated with microinflammation of the plaque are currently thought to cause plaque rupture. This event is the reason for local clot formation and ultimate organ infarction (25). In addition, vascular smooth muscle and endothelial function are altered in renal failure (26). An increased recirculation by a larger AV fistula may result in similar endothelial changes also in other organs at stress such as the heart, indicated by the data in the present study.
The present study does not rule out that β-blockers, used here as antihypertensives, may negatively influence cardiac function; those patients had a lower EF and larger LVS and LVESV.
The finding that more extensive dialysis was correlated with EF is probably related to the fact that a patient with larger body mass would have a greater EF and also receive more hours of dialysis/week due to the presence of more waste products.
Conclusions
This study of carefully selected stable HD patients exhibited that a maximal AV fistula diameter seems to be a more sensitive marker of left ventricular impairment than the methods used for blood flow measurement.
Footnotes
Acknowledgment
The authors thank Dr. C. Frisenette Fich, Ryhov Hospital, Jönköping; Dr. Pavel Oruda, Värnamo Hospital; and head nurse Lena Carlsson-Bjering, Högland Hospital, Eksjö, for arranging the measurements at the various dialysis units; and clinical physiology assistant Göran Lindholm for echocardiographic investigations.
Financial support: None.
Conflict of interest: None.
