Abstract
The search for biomarkers in systemic sclerosis (SSc) is driven by a goal to stratify patients, identify potential subgroups for treatment, and help assess response to therapy. Emerging evidence indicates that interleukin-6 (IL-6) and some family members are key biomarkers involved in SSc pathogenesis and therefore suitable targets for therapy. Recent studies evaluating IL-6 and its canonical Janus kinase/signal transducers and activators of transcription downstream pathways in modulating fibrotic response and immune cell function suggest a pivotal role for IL-6 in SSc pathogenesis. Although the significance and effect of local tissue expression of IL-6 and its family members are less well established, high levels of circulating IL-6 may identify subgroups of patients with early-stage disease, particularly those at risk for progressive lung fibrosis. In addition, higher disease activity may portend poor prognostic outcome in terms of survival and skin disease. Longitudinal assessment of serum levels of IL-6 and its signaling associates may prove valuable in monitoring response to treatment. As an IL-6–dependent surrogate marker, C-reactive protein may assist cohort enrichment if targeted treatment for IL-6 demonstrates efficacy, especially in subgroups with high IL-6 levels. Although IL-6 appears to be a key factor in the hierarchy of the complex network of disease-associated molecules, the systemic or autocrine/paracrine manner in which IL-6 asserts its profibrotic effects—particularly its interaction with other key pathogenic factors in SSc—is unknown. Ongoing clinical trials will help to delineate the mechanisms of IL-6 in SSc pathogenesis and inform on the role of these biomarkers.
Biomarkers and systemic sclerosis
There are substantial advantages to having measurable objective factors that reflect
the severity or activity of a disease and modification by treatment. This led to the
definition of
The need is great for biomarkers in systemic sclerosis (SSc) to help stratify patients, identify potential subgroups for treatment, and assess response to therapy. This is because SSc is a multisystem disease with variable clinical severity and outcome. Therefore, measures that can reflect this are of substantial potential value for the assessment and evaluation of patients. At present, objective measures of organ function (such as pulmonary function tests) and of renal function and clinical tools (such as modified Rodnan skin score) are available (2), but laboratory measures could be used to assess disease and determine the impact of treatment. It is also possible that such markers might be pathogenic mediators and targets for therapy. In this article, the extent to which this is true for IL-6 will be reviewed.
Studies of circulating CRP and IL-6 in SSc
Although SSc is a prototypic fibrotic disease with systemic vasculopathy, it has been recognized that markers of inflammation are important prognostic indicators. Thus, erythrocyte sedimentation rate (ESR) has been shown to correlate with poor survival and to be a predictor of poor outcome (3, 4). Analogously, it is expected that CRP also predicts outcome, and appreciation that CRP is generally a reflection of IL-6 levels adds further weight to this potential link. This association was shown more recently and robustly by a study from the Canadian Scleroderma Research Group (5). In this study of more than 1000 patients, CRP levels were elevated in approximately one-quarter of patients. This correlated, as expected, with ESR but was also more likely to be elevated in patients with early-stage disease or with higher skin score, worse lung function, and greater disease activity and severity. These findings are in line with the effects of IL-6 levels on SSc reported by Hasegawa et al (6), who showed in 1998 that IL-6 levels correlated with CRP and ESR and that levels were especially elevated in early diffuse SSc and were associated with lung fibrosis. This study built on earlier studies in 1992 (7) that reported increased serum levels of IL-2, IL-4, and IL-6 in SSc but lacked detailed investigation. Hasegawa et al (8) went on to show that there was enhanced production of IL-6 by peripheral blood mononuclear cells (PBMCs) in SSc, suggesting that these cells may be an important source of IL-6 in the circulation. Recent studies have further defined differential IL-6 expression by PBMCs, in particular B-cell subsets, in SSc (9). In contrast to later studies, Hasegawa et al (8) reported that there was an apparent difference in soluble IL-6 receptor (sIL-6R) in SSc but no difference for gp130. The link between CRP and IL-6 levels was shown more robustly in studies that used the better high-sensitivity CRP assays. In a comprehensive study of IL-6, CRP, and sIL-6R levels in a cohort of patients with SSc by Khan et al in 2012 (10), there was clear confirmation of the association between IL-6 and CRP and between IL-6 and elevated platelet counts (Fig. 1). This study (10) also demonstrated increased tissue expression of IL-6 in relevant sites that strengthened the possible pathogenic role of IL-6 and built on previous in vitro studies (11) that had suggested overproduction of IL-6 by SSc fibroblasts. Therefore, it is clear that acute phase markers, driven at least in part by IL-6, are important prognostic markers in SSc. The availability of these surrogates for IL-6 levels that are routinely clinically available has helped to enrich cohorts in trials and may ultimately be valuable in disease management if targeted treatment for IL-6 becomes available and is more effective in subgroups of patients with higher IL-6 levels.
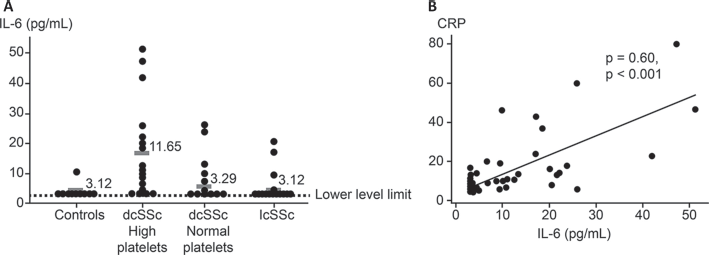
Serum IL-6 levels in patients with SSc and in healthy controls.
(
Circulating and tissue IL-6 and organ-based disease in SSc
Although the studies described are of interest in SSc, it is more relevant to outcomes if IL-6 and related serum proteins are reflective of organ-based complications. One of the first reports highlighted possible association between elevated IL-6 levels and lung fibrosis in a cross-sectional study of patients with SSc (12). Interestingly, those investigators also found elevated levels of other cytokines and explored their expression from activated PBMCs and T cells. Building on this was the study by Beirne et al (13) that used a novel multiplex approach to determine patterns of elevated cytokine and chemokine levels associated with lung fibrosis in patients with SSc. These patients were compared with other cohorts of patients, and it was notable that although no individual cytokine was predictive in this analysis, there was a trend for IL-6 and monocyte chemoattractant protein-1 (13). This trend was later explored in a larger validation cohort because a threshold level of IL-6 was associated with significant lung fibrosis, and the results of this landmark study defined the association between elevated IL-6 levels in initial samples and the development of lung fibrosis and disease progression (14). Patients with relatively preserved lung function, represented by forced vital capacity (FVC) more than 70% predicted, experienced higher mortality rates and greater risk for disease progression if they also had higher IL-6 levels. In contrast, in patients likely to have more advanced lung fibrosis, reflected by FVC less than 70% predicted, there was no predictive value of serum IL-6. These results are interesting because they suggest that IL-6 in serum may be more relevant in milder or earlier stage lung disease and also because FVC 70% has emerged as a predictive threshold for more severe disease that may require treatment with immunosuppressants (15). This observation is notable because emerging evidence from clinical trials suggests that IL-6R blockade may reduce the risk for significant worsening of lung function over 24 or 48 weeks of treatment. In line with this result is the observation by Liu et al (16) that CRP levels are associated with long-term progression and survival in SSc-associated lung fibrosis. In this study, not only were CRP levels higher in 266 patients with SSc compared with 97 controls (the GENISOS cohort), but CRP levels were associated with the absence of anticentromere antibodies and correlated with the concomitant severity of lung, skin, and joint involvement. Higher CRP levels were associated with shorter survival (p<.001) and predicted the long-term decline in FVC independent of potential confounders (age at baseline, sex, ethnicity, disease type, current smoking, body mass index, topoisomerase status, and treatment with immunosuppressive agents) in a multivariable model (p=0.006).
A broader analysis of the relationship between serum IL-6 and organ-based disease in SSc showed a link with measures of cardiorespiratory severity, including exercise capacity, pulmonary arterial estimated pressure, and lung fibrosis score (17). This is in line with earlier reports that IL-6 was associated with cardiac complications (18), albeit in a relatively small study of 31 patients with SSc and matched controls. Correlation with left ventricular function and N-terminal probrain natriuretic peptide was notable, though there was no correlation with echocardiographic measures of pulmonary or right ventricular function. These patients were not selected for pulmonary hypertension, so this may not be surprising. Similarly, the emerging interest in IL-6 in pulmonary arterial hypertension (PAH) is supported by early studies supportive of increased circulating levels (19, 20), and IL-6R blockade has been explored in a clinical trial for SSc-associated PAH (ClinicalTrials.gov, NCT02676947). The link between serum IL-6 and gastrointestinal (GI) disease is less clear because cases have been reported in which IL-6R antagonists may worsen GI disease (21). However, this connection seems unlikely based on much larger prospective trials that have not identified worsening of GI complications (22).
Taken together, the data do support a role for IL-6 in marking and possibly mediating organ-based complications in SSc. This is interesting given that hierarchical clustering and network analysis of dermal interstitial fluid identified a subset of patients with early-stage diffuse SSc with an innate inflammatory IL-6–associated profile (23).
IL-6 in the skin in scleroderma – morphea and related disorders
Studies of localized scleroderma (morphea) provide some of the best evidence that serum IL-6 levels may be reflective of local skin disease. Results are conflicting, however, with some studies reporting no evidence of elevated levels of IL-6 in morphea (24) and other, much earlier, studies reporting evidence of elevated IL-6 levels (25), suggesting that the stage and extent of morphea are most likely to be relevant. The most recent study (26) used modern multiplex analysis to examine 63 cytokines or growth factors in a cohort of patients with morphea and had positive results; therefore, it does seem possible that IL-6 is a marker of and a mediator in morphea. In that study, 63 cytokines, chemokines, and growth factors were measured in the sera of four patients with idiopathic morphea and of one patient with vitamin K1–induced morphea using a multiplex assay to identify the role of inflammatory/immunologic events in their pathogenesis. Full-thickness skin biopsy samples of affected skin were analysed by histopathology. Serum levels of IL-2, IL-4, IL-6, and interferon-β were markedly increased in the sera of patients with idiopathic morphea. However, the number of patients in this study is probably too small to draw strong conclusions.
IL-6 may be present and produced locally in SSc, as supported by analysis of dermal interstitial fluid and immunohistochemical evidence of local expression in affected skin, renal tissue, and pulmonary tissue (Fig. 2). In addition, some studies confirm a link with dermal interstitial fluid. Khan et al (10) showed that IL-6 levels may reflect long-term outcomes in skin and that elevated IL-6 levels could be a surrogate marker for clinical outcome for the subgroup of non-improver patients who have the worst skin trajectory, as identified in 2007 by Shand et al (27).
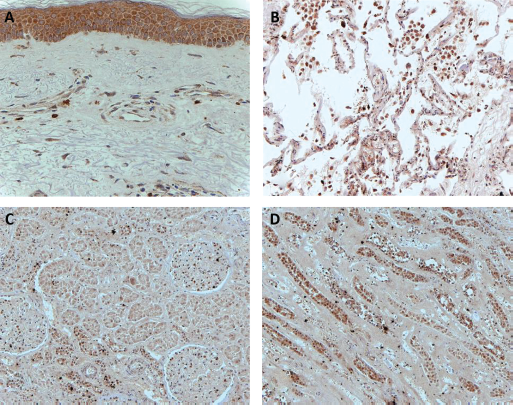
IL-6 expression in SSc skin and internal organs. IL-6 is detected in the
skin: in fibroblastic cells in the dermis, in the epidermis, and in
perivascular cells (
IL-6 changes in SSc interventional trials
Although the cross-sectional studies described have shed some light on the value of IL-6 and CRP in the assessment of SSc and localized scleroderma, there are many more informative and compelling data from longitudinal sampling in interventional trials. These trials have the value of sampling over time and may link the clinical changes in skin and internal organs with changes in soluble IL-6 and CRP levels. One of the most promising studies is the faSScinate clinical trial, which used IL-6 targeting for the treatment arm with a neutralizing antibody against IL-6R. Interestingly, although the cohort-enrichment strategies for this study recruited a group of patients with high baseline IL-6 levels, there was an initial increase in IL-6 levels that reduced with repeated dosing (22). Other studies show changes in IL-6 levels, such as with rituximab (RTX) treatment. Thus, in a series of 10 patients reported by Giuggioli et al (28), IL-6 levels were reduced after treatment in three of those patients with diffuse cutaneous SSc who had increased IL-6 at baseline. Consistent with those findings, Bosello et al (29) reported nine patients with SSc who had reduced levels of IL-6 in serum that were associated with reduction in skin score and severity index after treatment with RTX (29). A clinical trial of ultraviolet A1 phototherapy in 14 patients with morphea showed a reduction in mRNA for IL-6 and other inflammatory or profibrotic cytokines (30). Reports of other clinical trials that did not specifically target IL-6 also indicate changes associated with improved skin scores. This was not seen in a study of imatinib, which instead showed no evidence of benefit for this treatment and evidence of substantial toxicity (31). In a trial of hyperimmune caprine serum, skin scores did improve and IL-6 was among the serum cytokines whose levels decreased during therapy, thereby indicating it as a possible marker of skin change (32).
Links between IL-6 as a biomarker and its role in pathogenesis
The studies discussed here all point toward elevated IL-6 expression and activity in the skin or sera of patients with SSc, providing a plausible mechanistic link to pathogenesis given that IL-6 has been shown to be profibrotic and to modulate immune cell function in ways that are relevant to SSc pathogenesis. It is well established that the IL-6 trans-signaling pathway is pivotal in stromal cells, such as fibroblasts, which do not express membrane IL-6R, unlike some immune cells, such as macrophages and CD4 T cells, which are able to directly respond to IL-6. Activation of the key IL-6 downstream signaling Janus kinase/signal transducers and activators of transcription pathway in lesional tissue lends further support to the role of IL-6 in disease pathogenesis (10). Similar to profibrotic fibroblasts, these immune cells, which are important candidates in the development of SSc, can produce IL-6, raising the possibility that as well as being used as a marker or even a surrogate in SSc trials, IL-6 is a logical pathogenic mediator and a target for therapy. Although IL-6 appears to be a key factor in the hierarchy of a complex network of disease-associated molecules, the systemic or autocrine/paracrine way in which IL-6 asserts its effects, particularly the activation of dermal fibroblasts, is unknown, and the mechanism of the role of IL-6 should be better defined and will likely be informed by recently completed and ongoing clinical trials.
IL-6 in context; other members of the IL-6 family in SSc and their potential as markers and mediators
Among the 10 known members of the IL-6 family of cytokines, several have been reported to have altered expression levels in patients with SSc. Thus, in addition to the altered expression of IL-6 that is described in this article, some reports suggest that oncostatin M (OSM) is increased in SSc (Tab. I). Based on its diverse biological functions, including endothelial activation, cellular proliferation, migration and differentiation of fibroblasts, induction of acute phase responses, and wound healing, OSM may represent a candidate target for therapy. This is further supported by reports of increased expression of OSM in the circulation and lesional tissue of patients with SSc (33–34–35–36). A clinical trial of an anti-OSM therapeutic antibody is under way (ClinicalTrials.gov identifier, NCT02386436).
Cellular origin and relevant biology of IL-6 family members
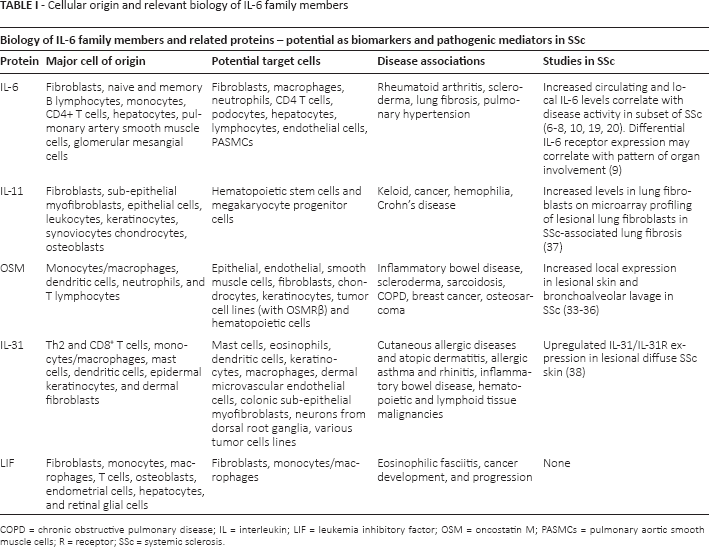
Although IL-11 shares many characteristics with IL-6, including its effect on megakaryocytopoiesis and induction of acute phase proteins, IL-11 has a complex effect on immunomodulatory activities by reducing proinflammatory cytokines but interacting with other cytokines and regulatory molecules in the induction and amplification of autoimmune responses. Although it is not normally detected in systemic circulation, IL-11 is locally expressed and is present in lesional areas in atopic dermatitis (39). Limited data support increased transcriptomic IL-11 expression in lung fibroblasts in SSc-associated lung fibrosis, and recent data suggest that the dermal T cells derived from the IL-11/11R axis may be critical in Th17 differentiation in SSc (37, 40).
IL-31 has relatively low homology to IL-6 and does not share gp130 as a common receptor and signal transducer subunit in its receptor complex. It is implicated mainly in the pathogenesis of allergic diseases and is an important mediator of pruritus in some diseases. It has been proposed as a possible candidate biomarker in some cases of diffuse SSc, of which pruritus is a complication. Its receptor is expressed on fibroblasts, but there is no evidence that fibroblasts express the cytokine. IL-31 levels are elevated in the interstitial fluid of patients with SSc and must therefore be derived from non-fibroblastic cells (38). Leukemia inhibitory factor is another member of the IL-6 family that shares its receptor with OSM. Levels are elevated in whole blood assays in patients with eosinophilic fasciitis, but studies of SSc have not yet been reported (41).
There is a paucity of data on other IL-6–related cytokines, including IL-27, neuropoietin, ciliary neurotrophic factor, and cardiotrophin 1, in SSc. IL-27 levels in sera, for example, were upregulated in patients with early-stage SSc with increased receptor expression in lesional fibroblasts (42), but this was not supported in a more recent study (24).
Conclusion
There is strong evidence to support the role of IL-6 in SSc, with emerging interest in its associated family members in early-stage diffuse scleroderma. Beyond an association with the induction of acute phase proteins and thrombopoiesis, correlation of levels of IL-6–related proteins with disease activity and survival outcome may help to stratify patient subgroups. The association between systemic or local IL-6 expression and pathogenic potential in specific organ manifestations, particularly skin and lung fibrosis, and in PAH may inform future therapeutic strategies in SSc. Greater understanding of the ligand-receptor interaction and downstream signaling pathways (summarized in Fig. 3) define the role of IL-6 and its family members in SSc and may help to elucidate potential targeted therapies.
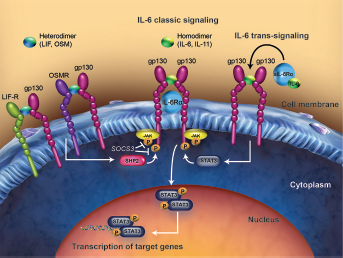
Distinct ligand-receptor signaling complexes for key IL-6 family members.
IL-6 and IL-11 can bind to their ligand-specific receptors (IL-6R and
IL-11R, respectively) to induce homodimerization of the common IL-6
receptor, gp130. However, in addition to membrane-bound (classic) IL-6R,
IL-6 may interact with the soluble form (trans-signaling) of IL-6R alpha to
facilitate signaling in cells that lack IL-6R. LIF and OSM bind to a
specific receptor, LIF-R, and recruit gp130 to form a heterodimer. Further,
OSM is capable of signaling via its own specific OSMR. OSMR is also part of
the heterodimeric complex for IL-31 signaling. These IL-6 members share
common downstream signaling transduction pathways with recruitment and
activation of JAKs, which phosphorylate the receptor cytoplasmic domain
providing docking sites for STAT3. STAT3 is then phosphorylated by JAK,
dimerizes, and trans-locates to the nucleus, where it regulates its target
genes, including
Footnotes
Acknowledgments
Medical writing and editorial assistance was provided by Liselle Bovell, PhD, and Sara Duggan, PhD, of ApotheCom (Yardley, PA, USA).
Conflict of interest: CPD reports receiving consulting fees from Genentech-Roche, Actelion, GlaxoSmithKline, Bayer, Sanofi-Aventis, Inventiva, and Boehringer Ingelheim; a research grant from Bayer; speaker fees from Actelion and Boehringer Ingelheim; and a research grant to his institution from CSL Behring.
VHO has nothing to disclose.
