Abstract
Systemic sclerosis (SSc) is a connective tissue disease of unknown etiology, manifesting in patients as tissue fibrosis, endothelial dysfunction, and inflammation. The disease is characterized by autoantibodies, a hallmark of autoimmunity. Various cytokines and growth factors are elevated in the systemic circulation and fibrotic lesions of patients with SSc. In particular, several studies over the past 2 decades have shown that interleukin-6 (IL-6) appears to be involved in the pathogenesis of SSc. Based on the association between aberrant IL-6 production and tissue fibrosis in patients with SSc, the anti-IL-6 receptor antibody, tocilizumab, is being investigated in clinical trials. This article reviews the biological features of IL-6 and the IL-6 receptor; the role of IL-6 in the pathogenesis of SSc; and the potential for IL-6 inhibition to be used in the treatment of patients with SSc.
Keywords
Introduction
Systemic sclerosis (SSc) is a complex disease resulting from impaired biological cell function, affecting endothelial cells, fibroblasts, lymphocytes, monocytes, and bone marrow cells (1–2–3–4). Vasculopathy and tissue fibrosis occur as a result of these impairments and are considered to be the main clinical features of patients with SSc. These patients have dysregulation of genetic and epigenetic function (5–6–7), innate immunity (8), and response to infections, resulting in aberrant immune activation and accelerated tissue damage leading to tissue fibrosis, vasculopathy, and autoimmunity. In the past 2 decades, many cytokines (9–10–11–12–13–14–15–16–17–18), chemokines (19–20–21), growth factors (22–23–24–25–26), and transcription factors (27, 28) have been studied for their potential involvement in the pathogenesis of SSc. For example, in vitro and in vivo experiments using scleroderma murine models have revealed that transforming growth factor-beta (TGF-β), platelet-derived growth factor (PDGF), and connective tissue growth factor may play crucial roles in collagen production by tissue fibroblasts in SSc (22–23–24–25–26). In addition to these growth factors, cytokines – including interleukin-1 (IL-1), IL-2, IL-4, IL-6, IL-8, IL-13, IL-33, and IL-35 – were found to be potent regulators of tissue fibrosis and endothelial damage (9–10–11–12–13–14–15–16–17–18). The findings of these experimental studies provided the rationale for developing novel targeted therapies in SSc, which have been evaluated in several clinical trials in the past decade; however, a disease-modifying therapy has yet to be approved for this indication. In a recent randomized, double-blind, placebo-controlled, phase 2 trial, tocilizumab, an anti-IL-6 receptor-alpha (IL-6Rα) antibody, was investigated as a treatment for skin fibrosis in patients with SSc and was shown to reduce skin thickening and improve lung function (29).
This article reviews the biological and physiological features of IL-6 and related peptides and its role in the pathogenesis of SSc.
Biological activity of IL-6
Acute phase response
IL-6 is a pleiotropic and pro-inflammatory cytokine that is produced by both lymphoid and nonlymphoid cells (30) and is associated with a wide range of biological functions. Interferon-beta 2 (IFN-β2), B-cell stimulatory factor type 2, and hepatocyte-stimulating factor, were each originally identified in the 1980s as distinct proteins (31–32–33). Molecular cloning of these molecules revealed that they were in fact identical, resulting in this 26-kDa peptide being referred to universally as IL-6 since late 1988 (34).
The biosynthesis of acute phase proteins, including C-reactive protein, fibrinogen, α1-acid glycoprotein, amyloid protein, and globulin by hepatocytes is regulated by IL-6 and, to a lesser extent, by IL-1 and tumor necrosis factor (TNF) (35–36–37). Further demonstrating the key role of IL-6 in the acute phase response, IL-6-deficient mice exhibited a severely defective inflammatory acute phase response following tissue damage or infection (38). This evidence indicated that the production of IL-6 was absolutely required to regulate an acute phase response as a proinflammatory cytokine.
Immune response
B cells play a central role in antibody (immunoglobulin) production under the
control of various cytokines produced by T cells and macrophages. IL-6 was
identified as a helper factor in monocyte supernatants, mediating
phytohemagglutinin-induced T-cell activation and proliferation (39). Furthermore, IL-6 was
shown to play a role in the late phase of
Hematopoiesis
IL-6 can stimulate hematopoietic stem cells in vitro and can synergize with macrophage colony-stimulating factor (M-CSF) under stimulation by macrophage colony forming units, affecting both the number and the size of macrophage colonies (47). IL-6 has also been found to act synergistically with granulocyte-macrophage colony stimulating factor (GM-CSF) to act on myeloid hematopoietic progenitor cells (48).
IL-6 is involved in the maturation of megakaryocytes and promotes the production of platelets, suggesting that it functions as a thrombopoietic factor (49). The administration of IL-6 was shown to increase the number of platelets in both mice and monkeys. Human megakaryocytes have been observed to express the IL-6 receptor and to produce IL-6, suggesting that IL-6 may regulate the terminal maturation of megakaryocytes in an autocrine manner (50).
Other responses
The potential involvement of IL-6 in proliferative glomerulonephritis suggests that this cytokine may induce the proliferation of kidney mesangial cells (51). Indeed, IL-6 transgenic mice exhibit mesangial proliferative glomerulonephritis, which was prevented by the anti-IL-6R antibody (51). IL-6 may also directly influence osteoclast development and may play a role in postmenopausal osteoporosis. In an in vivo experiment, mice that underwent ovariectomy exhibited an enhancement of osteoclast development; this phenomenon was prevented by the administration of an anti-IL-6 antibody (52).
The IL-6 receptor
The IL-6 receptors consist of 2 subtypes – an 80-kDa IL-6-binding protein (IL-6R chain) and a 130-kDa signal transducer (gp130) – each of which has the 4 conserved cysteine residues and a tryptophan-serine-X-tryptophan-serine motif that characterizes members of the cytokine receptor family (53–54–55). It has been established that IL-6 is unable to directly bind to gp130 and that IL-6 binding to IL-6R is required to generate the high-affinity complex of IL-6/IL-6R/gp130 (56). IL-6R is also expressed as a soluble form, and the complex of IL-6 and soluble IL-6R can generate IL-6-mediated signal transduction through gp130 (57). A schematic representation of complex formation between IL-6 and the transmembrane or soluble form of IL-6R is shown in Figure 1.
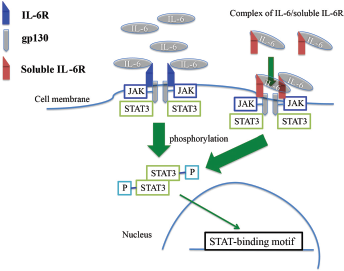
IL-6 receptors and signal transduction. IL-6 exerts various biological effects by binding to IL-6R on the cell surface and the IL-6/IL-6R complex, and then acts on gp130. Alternatively, the complex of IL-6/soluble IL-6R stimulates gp130 directly. These pathways are involved in the phosphorylation of JAK and STAT3, and the subsequent transcription of many genes, including procollagen. gp130 = glycoprotein 130; IL-6 = interleukin-6; IL-6R = interleukin-6 receptor; JAK = Janus activated kinase; STAT3 = signal transducer and activator of transcription 3.
Signal transduction of IL-6
The binding of IL-6 to IL-6R and the subsequent association with gp130 induces the formation of a hexamer with a 2:2:2 stoichiometry (58). Although IL-6R lacks a kinase domain, gp130 has several tyrosine kinases, Janus kinase 1 (JAK1) and JAK2. Binding of IL-6 and IL-6R induces homodimerization of gp130 and phosphorylation of JAKs (59). In addition, signal transducer and activator of transcription (STAT) plays a central role in this cytokine signal transduction pathway, mainly through STAT3 (60).
The signal transduction of IL-6 is negatively regulated by 2 families of inhibitors: the suppressor of cytokine signaling (SOCS) and the protein inhibitors of activated STAT (PIAS). The SOCS family is characterized by a central SH2 domain flanked by an N-terminal region containing a conserved motif, known as a SOCS box (61). Unlike the SOCS family, PIAS constitutes a family of constitutively expressed negative regulators of STAT-mediated gene activation (62). Five members of PIAS have been identified with a shared homology and the presence of several highly conserved domains (62).
Elevation of IL-6 in SSc
Elevated levels of serum IL-6 in patients with SSc have been reported in several studies (10–11–12–13, 15, 63–64–65). Significantly higher levels of soluble IL-6R and anti-IL-6 autoantibodies have also been reported in patients with SSc compared with healthy controls (66, 67). The complex of IL-6-soluble IL-6R, and IL-6-anti-IL-6 autoantibody can bind to gp130 and induce signal transduction in SSc (68, 69). Significant correlations have been observed between serum levels of IL-6 and the severity of skin thickening (70), disease progression in interstitial lung disease (63), and cardiac involvement (64, 65); these complications of SSc are characterized by tissue fibrosis. In addition to elevated circulating IL-6 levels, cultured fibroblasts from SSc patients were found to express strikingly high levels of IL-6 at both the transcriptional and the protein levels (71–72–73–74). Inhibition of IL-6 signal transduction using an anti-IL-6 antibody attenuated collagen production in cultured fibroblasts of SSc patients (14). Additionally, elevated secretion of IL-6 from peripheral monocytes was observed in patients with SSc-associated interstitial lung disease (75). Increased numbers of peripheral B cells and higher IL-6 levels were observed in SSc patients compared with healthy subjects, along with increased in vitro fibroblast proliferation and collagen production when normal and scleroderma fibroblasts were exposed to B-cell supernatants from these patients (76). B cell activating factor (BAFF) is a cytokine that is primarily produced by monocytes, macrophages, and myofibroblasts and is implicated in the regulation of B cell survival, activation, proliferation, and maturation (77). Serum levels of BAFF were elevated in patients with SSc and correlated with the extent of skin fibrosis in SSc (78). Moreover, it was reported that BAFF mRNA levels in monocytes in patients with SSc, especially in IFN type I signature positive monocytes (79). BAFF receptor (BAFFR) was also overexpressed in B cells in of patients with SSc (80). Those observations suggest that the signal transduction through BAFF/BAFFR may contribute to the activation of B cells in SSc. The activation of B cells is one of the key pathways to induce excessive IL-6 production in SSc.
In summary, high concentrations of IL-6 are found in the sera of SSc patients (10–11–12–13, 15, 63–64–65) and have been shown to be highly correlated with the severity of tissue fibrosis. In vitro experiments further support these observations, demonstrating an increased secretion of IL-6 by cultured skin fibroblasts, monocytes, and B lymphocytes from patients with SSc (71–72–73–74–75–76). In particular, the overproduction of IL-6 can lead to excessive collagen production by fibroblasts, which contributes to tissue fibrosis in SSc (11, 13, 14) (Fig. 2).
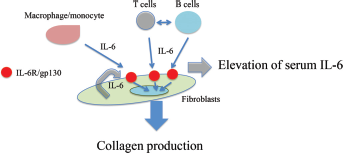
Excessive collagen production by IL-6 in SSc. IL-6 is produced by T cells, B cells, macrophages/monocytes, and fibroblasts. In SSc, the high levels of IL-6-stimulated fibroblasts results in excessive collagen production, and the formation of skin and lung lesions. IL-6 = interleukin-6; IL-6R = interleukin-6 receptor; SSc = systemic sclerosis.
Precise mechanisms of upregulation of IL-6
As previously noted, the sources of IL-6 in the systemic circulation of patients with SSc may be fibroblasts, monocytes, and B cells. In fibroblasts, intracellular IL-1α (pro-IL-1α) was shown to be constitutively expressed, and the autocrine effect of pro-IL-1α induced the production of IL-6, which was inhibited by use of an antisense oligodeoxynucleotide complementary to IL-1α mRNA (14). Furthermore, IL-6 production was found to be enhanced by PDGF in cultured fibroblasts from SSc patients and could be inhibited by an IL-1 receptor antagonist (81). These observations indicate that PDGF-induced IL-6 overexpression is mediated through the action of IL-1. Another study reported that the increased expression of CD40 on the surfaces of skin fibroblasts, coupled with its interaction with CD154, induced IL-6 production in patients with SSc (82). Recently, an autotaxin-lysophosphatidic acid pathway induced IL-6 production in SSc fibroblasts, suggesting that the autotaxin/lysophosphatidic acid/IL-6 axis is fundamental to the development and progression of fibrosis in SSc (83).
Effects of IL-6 on fibrosis
IL-6 is a potent inducer of collagen production in fibroblasts, through direct and indirect mechanisms. It is well known that IL-6 induces the expression of procollagen mRNA through the phosphorylation of JAK, extracellular signal-regulated kinase, and mitogen-activated protein kinase (84–85–86). IL-6 also enhances TGF-β, which is a strong inducer of collagen production (87). In addition to enhanced collagen production, the establishment of tissue fibrosis occurs because of a lack of balanced degradation of the extracellular matrix, which may be strictly controlled by matrix metalloproteinases and tissue inhibitors of metalloproteinases (TIMPs) (3). IL-6 significantly increases TIMP-1 synthesis through the phosphorylation of STAT3, resulting in collagen accumulation (88). Furthermore, IL-6 increases the population of myofibroblasts differentiated from normal fibroblasts (89), and these myofibroblasts play a crucial role in tissue fibrosis.
Therapeutic effects of inhibiting IL-6 action in SSc
The involvement of IL-6 in SSc-associated tissue fibrosis makes it a rational therapeutic target (90–91–92). Other autoimmune diseases – such as rheumatoid arthritis, Sjögren's syndrome (93), vasculitis (94), and graft-versus-host disease (95) – can be treated by blocking the action of IL-6 (96, 97). Monoclonal anti-IL-6 antibodies have previously been developed to inhibit the signal transduction of IL-6, but this strategy failed in the initial clinical trials. Another avenue that was explored involved targeting the receptor instead of the cytokine itself. Tocilizumab, a humanized monoclonal antibody against IL-6Rα, was generated and tested in clinical trials for the treatment of rheumatoid arthritis, demonstrating efficacy and safety similar to that of TNF-α antagonists (98, 99). Currently, tocilizumab is approved for the treatment of patients with rheumatoid arthritis, polyarticular or systemic juvenile idiopathic arthritis, and, most recently, giant cell arteritis (100). Case reports of SSc patients treated with tocilizumab 8 mg/kg every 4 weeks reported skin softening (101), thinning of collagen fiber bundles in the dermis (101), and improvements in joint mobility (102). These were the first reports to demonstrate preliminary evidence of efficacy for tocilizumab in patients with SSc. A phase 2 randomized controlled trial evaluating subcutaneous tocilizumab for the treatment of SSc was subsequently conducted (29), reporting reductions in skin thickening. These promising findings supported the continued development of tocilizumab for the treatment of SSc, which is being investigated in a phase 3 trial.
Similar to the direct suppression of IL-6 and IL-6R, inhibition of IL-6/IL-6R/gp130 signal transduction has the potential to be a novel strategy for the treatment of SSc. A recent investigation revealed that the inhibition of JAK-2 prevented bleomycin-induced fibrosis in mice and reduced skin fibrosis in TSK-1 mice (84). In addition, the pathway of JAK-STAT3 promotes collagen synthesis, proliferation, and migration of fibroblasts in fibrotic diseases, including SSc and keloid. Aberrant activation of this pathway also has been reported in the skin of SSc patients, particularly in fibroblasts.
Rituximab (anti-CD20 antibody) is a B-cell depletion therapy that may be useful in the treatment of patients with SSc. A multicenter, open-label, comparative study of rituximab versus conventional treatment reported that rituximab had a beneficial effect on lung function and skin thickening in SSc (103). In addition, B-cell depletion in patients with SSc resulted in the decrease of serum IL-6 (104). Therefore, the beneficial effects of rituximab on tissue fibrosis may be explained by the inhibition of IL-6 production from B cells.
Conclusion
IL-6 was initially reported as a B-cell stimulating factor that plays a crucial role in B-cell differentiation and immunoglobulin production. However, further research has made significant advances in the characterization of the biological features of IL-6, the IL-6 signaling pathway, IL-6 synthesis, and the cells targeted by IL-6. Among them, fibroblasts are both a source of IL-6 synthesis and a target for IL-6 through its specific receptor. In addition to TGF-β, IL-6 is a strong regulator of tissue fibrosis in patients with SSc. Although several therapies inhibiting TGF-β have not affected tissue fibrosis in SSc patients in clinical trials, IL-6-blocking therapy using tocilizumab may have potential for the treatment for skin and lung fibrosis in patients with SSc.
Footnotes
Acknowledgments
Medical writing and editorial assistance was provided by Maxwell Chang of ApotheCom.
Conflict of interest: The author has no financial interest related to this study to disclose.
