Abstract
Background
Long noncoding RNAs (lncRNAs) are emerging as key molecules in human cancer. In the present study, we explored the role of the lncRNA PANDAR in colorectal cancer (CRC).
Methods
The relative expression level of lncRNA PANDAR in CRC tissues and cell lines was determined by quantitative real-time polymerase chain reaction (qRT-PCR). The associations between PANDAR expression and clinicopathological features of CRC patients were further analyzed. Kaplan-Meier survival analysis was performed to evaluate the value of PANDAR in the prognosis of CRC patients. Furthermore, the biological function of PANDAR on CRC cell growth, apoptosis and mobility was investigated through MTT, flow cytometry, transwell migration and invasion assays in vitro.
Results
The expression level of PANDAR was higher in CRC tissues and cells compared with adjacent nontumor tissues and normal colonic cell line NCM460. PANDAR expression was significantly correlated with local invasion, lymph node metastasis and TNM stage. Kaplan-Meier analysis showed that patients with high PANDAR expression had poorer overall survival than patients with low PANDAR expression. Multivariate Cox regression analysis indicated that PANDAR might be an independent prognostic factor for CRC patients. Furthermore, PANDAR knockdown significantly inhibited cell proliferation, cycle progression, migration and invasion of CRC in vitro.
Conclusions
Our results suggest that high expression of PANDAR was involved in CRC progression and could act as an independent biomarker for prognosis of CRC patients.
Introduction
Colorectal cancer (CRC) is the third most commonly diagnosed cancer and the fourth most common cause of cancer deaths worldwide, with over 1.2 million new cases each year (1). It is one of the most common malignant tumors in China. During the past few decades, the incidence of CRC has been increasing (2). Despite improvements in surgical and adjuvant chemotherapies, the 5-year overall survival rate of CRC patients has not changed dramatically (3). One of the major factors for the poor outcome is metastasis (4). Thus, there is an urgent demand for research into novel molecular biomarkers that can serve as diagnostic and prognostic markers for CRC.
The human genome sequencing project has found that 70% of the genome is transcribed, but only up to 2% of the human genome serves as blueprints for proteins (5). Long noncoding RNAs (lncRNAs) are defined as endogenous cellular RNAs more than 200 nucleotides in length that lack an open reading frame of significant length (6). In recent years, numbers of lncRNAs have been shown to be involved in carcinogenesis and cancer progression. For example, Zhang et al showed that up-regulation of the lncRNA MALAT1 correlated with tumor progression and poor prognosis in clear cell renal cell carcinoma (7). Yang et al reported that overexpression of the lncRNA HOTAIR predicted tumor recurrence in hepatocellular carcinoma patients following liver transplantation (8). Cao et al found that decreased expression of the lncRNA GAS5 could predict a poor prognosis in cervical cancer patients (9). However, the expression level and clinical significance of the lncRNA PANDAR in CRC remains unclear. Thus, the aim of this study was to explore the biological function and underlying mechanism of PANDAR in CRC.
Materials and Methods
Patients and Tissue Samples
Surgical specimens of cancer tissue and adjacent nontumor tissues were obtained from 102 patients with primary CRC who underwent surgery without pre-operative treatment at The First Affiliated Hospital of Zhengzhou University, from 2008 to 2010. All tissue samples were immediately frozen in liquid nitrogen and stored at −80°C until the extraction of RNA. The diagnoses were confirmed by pathological findings. All tumors were classified according to the seventh edition of the International Union for International Cancer Control (UICC) TNM staging system for CRC. Written informed consent was obtained from each patient, and research protocols were approved by the ethics committee of The First Affiliated Hospital of Zhengzhou University.
Cell culture
The human CRC cell lines HCT116, HT29, SW480 and DLD-1 and a normal colonic cell line NCM460 were obtained from the American Type Culture Collection (ATCC). All of the cell lines were maintained in RPMI 1640 medium (Gibco) supplemented with 100 U/mL penicillin sodium, 10% fetal bovine serum (FBS; Gibco) and 100 mg/mL streptomycin sulfate in humidified atmosphere with 5% CO2 at 37°C.
Cell transfection
Cells were transfected with specific siRNA against PANDARs (si-PANDARs) with Lipofectamine 2000 (Invitrogen) according to the manufacturer's instructions. The target sequences for si-PANDAR were si-PANDAR-1: 5′-GCAATCTACAACCTGTCTT-3′, si-PANDAR-2: 5′-GGGCAUGUUUUCACAGAGGUU-3′. Briefly, cells were seeded into 6-well plates and cultured for additional 24 hours. Afterwards, the medium was refreshed with serum-free RPMI 1640 medium, and then siRNA together with transfection reagent was added into each well. Six hours later, the medium was replaced with normal RPMI 1640 supplemented with 10% FBS. Cells were harvested for subsequent analysis 48 hours after the transfection. siRNAs were synthesized by RiboBio (China).
Quantitative real-time PCR
Total RNA was extracted from the tissue samples or cells with Trizol reagent (Takara), and first-strand cDNA was synthesized from 2 μg of total RNA using random primers and the M-MLV Reverse Transcriptase (Invitrogen). The following primers were used to detect the expression of PANDAR and glyceraldehyde-3-phosphate dehydrogenase (GAPDH; internal control): PANDAR (sense): 5′-CAATGCCTTGCTTCACAGTC-3′; PANDAR (antisense): 5′-TGGGGTTCTTAGAAGTGGTGA-3′; GAPDH (sense): 5′-AGAAGGCTGGGGCTCATTTG-3′; GAPDH (antisense): 5′-AGGGGCCATCCACAGTCTTC-3′. RNA expression was measured by quantitative real-time polymerase chain reaction (qRT-PCR) using the SYBR-Green method (Takara) according to the manufacturer's instructions. The results were normalized to the expression of GAPDH.
MTT assay
The effect of PANDAR on the proliferation of cells was measured by using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Briefly, cells were seeded in 96-well plates and incubated under normal conditions for 4 hours. MTT was then added to the corresponding well at 24, 48, 72 and 96 hours after transfection. Finally, the cells were dyed with dimethyl sulfoxide (DMSO). The absorbance was measured with a microplate reader (Bio-Rad) at 490 nm.
Cell cycle analysis
Flow cytometry was performed to determine the effect of PANDAR on cell cycle. Cells transfected with si-PANDAR and si-NC were harvested 48 hours after transfection. Cells for cell cycle analysis were stained with propidium oxide using the Cycle Test Plus DNA Reagent Kit (BD Biosciences) following the protocol and analyzed by FACScan. The percentages of cells in G0/G1, S and G2/M phases were counted and compared.
Transwell assay
Transwell assay was performed to determine the effect of PANDAR on migration and invasion of the CRC cells. For the migration assay, transfected cells were seeded on the upper chamber of the transwell apparatus and 500 μL RPMI 1640 containing 10% FBS was added to the lower chamber. After incubation for 24 hours, cells adhered to the lower chamber were fixed with methanol and then stained with 1% crystal violet. For the invasion assay, cells were treated with the same method except the membrane was precoated with Matrigel. Finally, the stained cells were counted manually in 7 randomly selected fields.
Statistical analysis
All statistical analyses were performed with SPSS 17.0 software. The association between PANDAR expression and clinicopathological features was analyzed by chi-square test. Kaplan-Meier curves were plotted to describe the overall survival rate of CRC patients. The prognostic value of PANDAR was evaluated by multivariate Cox regression analysis. A p value <0.05 was considered as statistically significant.
Results
LncRNA PANDAR expression was up-regulated in CRC tissues and cell lines
The relative expression level of lncRNA PANDAR was determined by qRT-PCR in a total of 102 patients with CRC. As shown in Figure 1A, PANDAR was up-regulated in CRC tissues compared with adjacent nontumor tissues (p<0.05). Furthermore, we explored the expression of PANDAR in 4 CRC cell lines (HCT116, HT29, SW480 and DLD-1) and a normal colonic cell line NCM460. Our data showed that PANDAR expression was increased in all 4 CRC cell lines compared with the normal colonic cell line NCM460 (Fig. 1B; p<0.05). Among the CRC cell lines, HT29 and DLD-1, with the high PANDAR expression, were chosen for the next study. These results indicated that abnormal PANDAR expression might be related to CRC progression.
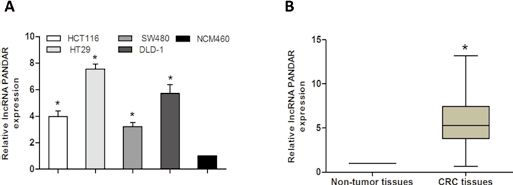
Long noncoding RNA (lncRNA) PANDAR expression was up-regulated in colorectal cancer (CRC) tissues and cell lines. (A) Relative expression of PANDAR in 102 pairs of CRC tissues and adjacent nontumor tissues was determined by quantitative real-time polymerase chain reaction (qRT-PCR) analysis. (B) Relative expression of PANDAR in 4 CRC cell lines and a normal colonic cell line NCM460 was determined by qRT-PCR analysis. GAPDH was used as an internal control (*p<0.05).
Relationship between lncRNA PANDAR expression and clinical features of CRC patients
To explore the correlation of lncRNA PANDAR expression levels with clinicopathological features in CRC patients, we divided the 102 CRC patients into a high PANDAR expression group (n = 51) and a low PANDAR expression group (n = 51) according to the median value of relative PANDAR expression. The relevance of PANDAR and clinicopathological features were analyzed by chi-square test. We found that high PANDAR expression was significantly correlated with local invasion, lymph node metastasis and advanced TNM stage (Tab. I; p<0.05). However, there was no significant correlation of PANDAR expression with other clinicopathological features such as age, sex, tumor size and histological grade (Tab. I; p>0.05). Thus, these results indicated that increased expression of PANDAR might have important roles in CRC progression and development.
Associations of PANDAR expression with clinicopathological features of CRC patients
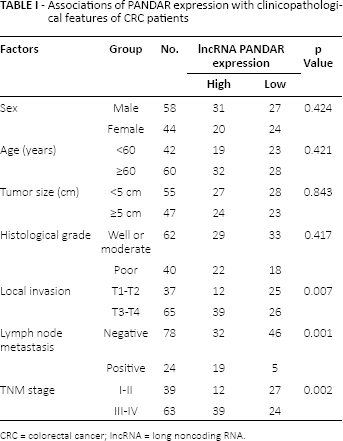
CRC = colorectal cancer; lncRNA = long noncoding RNA.
LncRNA PANDAR up-regulation associated with poor prognosis in CRC patients
The association between lncRNA PANDAR expression and overall survival of CRC patients was investigated by Kaplan-Meier analysis and log-rank test. The results indi cated that patients who had high PANDAR expression had a shorter overall survival than patients who had low PANDAR expression (Fig. 2). Multivariate Cox regression analysis was performed to estimate the prognostic value of PANDAR and various clinicopathological features in CRC patients. Our data showed that PANDAR expression, local invasion, lymph node metastasis and TNM stage were independent prognostic factors for overall survival of patients with CRC (Tab. II; hazard ratio [HR] = 3.083, 95% confidence interval [95% CI], 0.843-7.891; p<0.05).
Multivariate Cox regression analysis PANDAR for overall survival of patients with CRC
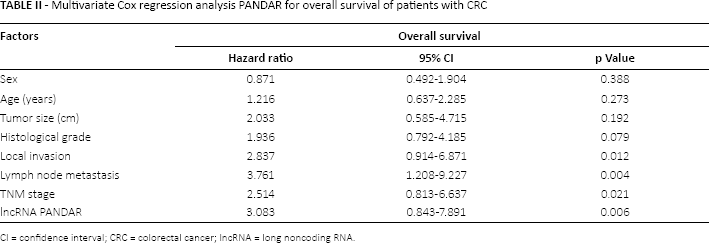
CI = confidence interval; CRC = colorectal cancer; lncRNA = long noncoding RNA.
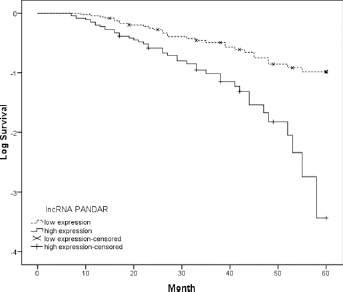
Kaplan-Meier survival analysis of association between lncRNA PANDAR expression and overall survival of 102 colorectal cancer (CRC) patients. Patients with high PANDAR expression showed poor overall survival compared with patients with low PANDAR expression (p = 0.001; log-rank test).
Silencing lncRNA PANDAR inhibited CRC cell proliferation and induced cell cycle arrest
To further explore the biological effects of lncRNA PANDAR in CRC progression, we transfected 2 si-PANDARs (si-PANDAR-1 and si-PANDAR-2) into CRC cells. The qRT-PCR assays revealed that PANDAR expression was significantly reduced in both HT29 and DLD-1 cells (Fig. 3A; p<0.05). In the further investigation, we used the higher efficiency si-PANDAR-1 to explore lncRNA PANDAR function. MTT assay showed that the proliferation of cells transfected with si-PANDAR-1 was decreased compared with that of cells transfected with si-NC controls (Fig. 3B; p<0.05). To further explore whether the effect of PANDAR on cell proliferation reflected a cell cycle arrest, cell cycle analysis was performed, and flow cytometric analysis revealed that reduced expression of PANDAR showed a significant blocking of cells at the G1/G0 phase (Fig. 3C; p<0.05). Thus, these findings suggested that PANDAR effectively retarded cell growth by blocking the G1/S transition in CRC cells in vitro.
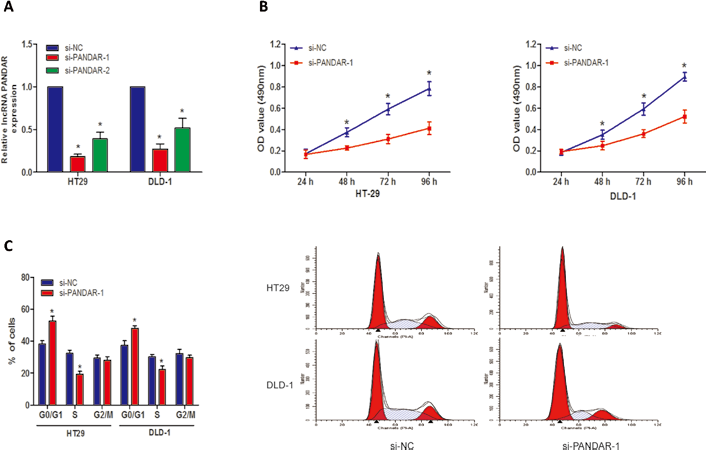
Knockdown of lncRNA PANDAR inhibited colorectal cancer (CRC) cell proliferation in vitro. (A) PANDAR expression levels were determined by quantitative real-time polymerase chain reaction (qRT-PCR) after HT29 and DLD-1 cells were transfected with si-PANDAR-1 or si-NC. (B) The percentage cell proliferation of HT29 and DLD-1 cells transfected with si-PANDAR-1 or si-NC was detected by MTT assay. OD = optical density. (C) Cell cycle distribution for HT29 and DLD-1 cells transfected with si-PANDAR-1 or si-NC was detected by cell cycle analysis (*p<0.05).
Silencing lncRNA PANDAR inhibited CRC cell migration and invasion
To investigate the effect of lncRNA PANDAR on the motility of CRC cells, transwell migration and invasion assays were performed. The results showed that reduced expression of PANDAR significantly inhibited cell migration (Fig. 4A; p<0.05, for both HT29 and DLD-1 cells) and invasion (Fig. 4B; p<0.05, for both HT29 and DLD-1 cells). Thus, these results suggested that decreased expression of PANDAR could inhibit CRC cell migration and invasion in vitro.
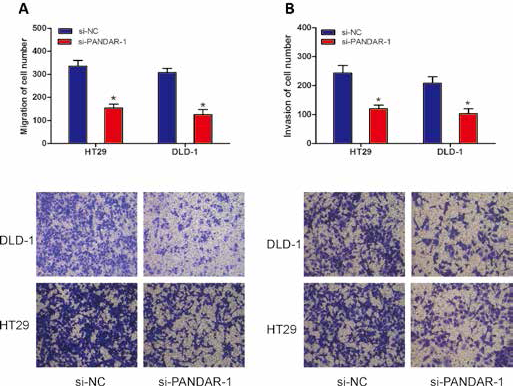
Knockdown of lncRNA PANDAR inhibited cell migration and invasion in vitro. (A) Cell migration ability of HT29 and DLD-1 cells transfected with si-PANDAR-1 or si-NC was detected by transwell migration assay. (B) Cell invasion ability of HT29 and DLD-1 cells transfected with si-PANDAR-1 or si-NC was detected by transwell invasion assay (*p<0.05).
Discussion
CRC remains one of the leading causes of cancer-related death worldwide, so finding new molecular targets for its diagnosis, prognosis and treatment has the potential to improve the clinical strategy and outcome of this disease (10). In the present study, there were 4 significant findings in our results: First, PANDAR expression in CRC tissues and cell lines was significantly higher than that in adjacent nontumor tissues and the normal colonic cell line NCM460; second, increased expression of PANDAR was significantly associated with local invasion, lymph node metastasis and advanced TNM stage of CRC patients; third, multivariate Cox regression analysis revealed that PANDAR expression was a significant risk factor affecting the overall survival of CRC patients. Fourth, down-regulated PANDAR expression could inhibit CRC cell proliferation, migration and invasion in vitro. These data demonstrated that PANDAR expression could be a valuable biomarker of the progression and the prognosis in CRC.
LncRNAs are a new class of transcripts recently discovered to be pervasively transcribed in human genome and to play a critical role in epigenetic regulation (11). In addition, as with microRNA, lncRNAs may function as tumor markers for the prediction of tumor prognosis (12). Recent studies indicated that the expression of lncRNAs is dysregulated in different kinds of tumors, including CRC. For example, Shi et al showed that low expression of the novel lncRNA RP11-462C24.1 suggested a biomarker of poor prognosis in CRC (13). Han et al found that increased expression of the lncRNA AFAP1-AS1 promoted tumor growth and metastasis in CRC (14). Liu et al demonstrated that overexpression of the lncRNA DANCR was associated with advanced tumor progression and poor prognosis in patients with CRC (15). Shen et al showed that lncRNA SPRY4-IT1 promoted CRC metastasis by regulating the epithelial-mesenchymal transition (16). These findings suggested that deregulation of lncRNAs may play vital roles in the tumorigenesis and progression of CRC. More extensive investigations are required to elucidate the roles of lncRNAs in the development of CRC so as to identify those lncRNAs that may serve as novel prognosis predictors or as therapeutic targets for CRC.
LncRNA PANDAR (promoter of CDKN1A antisense DNA damage activated RNA), 1,506 nt in length and located at chromosome subband 6p21.2, which we focused on in this study, was firstly reported by Hung et al (17). They found that PANDAR was induced in a p53-dependent manner and interacts with the transcription factor NF-YA to limit the expression of proapoptotic genes in normal human fetal lung fibroblasts (17). Recently, Ma et al showed that PANDAR was dramatically up-regulated in gastric cancer tissues compared with adjacent normal tissues, and the increased expression of PANDAR was positively correlated with tumor invasion, TNM stage, lymphatic metastasis and patients' outcomes (18). Peng et al showed that PANDAR was highly expressed in hepatocellular carcinoma tissues and could serve as an independent prognostic marker. In addition, they found that PANDAR knockdown significantly repressed cell proliferation, colony formation and cycle progression of hepatocellular carcinoma in vitro (19). Sang et al showed that PANDAR was up-regulated in breast cancer tissues and cell lines, and regulated the G1/S transition of breast cancer cells by suppressing p16(INK4A) expression (20). Those studies indicated that PANDAR plays important roles in tumor progression. In our study, we showed that PANDAR was up-regulated in CRC tissues and cell lines. In addition, overexpression of PANDAR was associated with local invasion, lymph node metastasis and advanced TNM stage, which raises the possibility that PANDAR might have an important role in the development or pathogenesis of CRC. More importantly, we proved that PANDAR expression was significantly associated with overall survival of patients with CRC. Multivariate Cox regression analysis illustrated that PANDAR was an independent factor for prognosis of CRC patients. Finally, in in vitro assays, we found that PANDAR knockdown could inhibit CRC cell proliferation by blocking the G1/S transition in CRC cells. In addition, we showed that PANDAR inhibition also suppressed CRC cell migration and invasion in vitro. These studies expanded the function of PANDAR in tumor progression and development.
In conclusion, we demonstrated that PANDAR might play a vital role in the development and progression of CRC. The development of PANDAR-based therapeutic strategies for the down-regulation of such oncogenic lncRNAs may provide a novel and promising alternative therapeutic approach for future cancer treatment.
Footnotes
Financial support: No grants or funding have been received for this study.
Conflict of interest: None.
