Abstract
Background
Through analyzing apparent diffusion coefficient (ADC) values and morphological evaluations, this research aimed to study how magnetic resonance imaging (MRI)-based breast lesion characteristics can enhance the diagnosis and prognosis of breast cancer.
Methods
A total of 118 breast lesions, including 50 benign and 68 malignant lesions, from 106 patients were analyzed. All lesions were measured with both diffusion-weighted (DW) and dynamic contrast-enhanced (DCE) MRI. The average ADC of breast lesions was analyzed at
Results
A
Conclusions
Both ADC values and MRI morphological assessment could be used to distinguish malignant breast lesions from benign ones.
Keywords
Introduction
Breast cancer is a leading cause of cancer-related deaths among women under the age of 50 years worldwide (1). Breast cancer, a heterogeneous complex disease, has distinct morphological appearances, molecular features and clinical characteristics (2). Factors such as heredity, obesity, alcoholism and smoking have been associated with breast carcinoma (3). From the molecular biology point of view, breast carcinoma comprises 4 major subtypes: luminal A (estrogen receptor [ER] and/or progesterone receptor [PR] positive, human epidermal growth factor receptor 2 [HER2] negative); luminal B (ER and/or PR positive, as well as HER2 positive or Ki67 14%); HER2-enriched (HER2 amplified, ER and PR negative); and triple-negative (ER, PR and HER2 negative) (4). Breast cancer is generally accompanied by clinical manifestations of a breast mass, nipple discharge, changed skin, nipple and/or areola abnormalities, and enlarged axillary lymph nodes; it has similar survival rates when radical mastectomy or breast-conserving surgery is performed (5). Diagnostic methods for breast cancer include mammography x-ray examination, computed tomography (CT), magnetic resonance imaging (MRI), positron emission-CT (PET-CT) and ultrasonography and tissue biopsy. Clinical practitioners often use multiple methods in order to obtain an explicit diagnostic conclusion (6).
Based on the nuclear magnetic resonance principle, MRI analyzes the internal structure of objects by applying electro-magnetic waves emitted by a gradient magnetic field. Due to its low cost and wide availability, MRI plays a vital role in both clinical practice and medical research, especially in the prevention, diagnosis, treatment and prognosis of heart disease, lung cancer, gastric carcinoma and breast cancer (7). Because of its ability to assess tissue vascularization and distinguish between key tumor and fibrotic tissue, MRI is an extremely useful method in determining the real extent of the disease. MRI also is able to visualize different morphological and kinetic features of different biology and histology (5). Comparing the 3 breast imaging methods (mammography, high-frequency breast ultrasound and MRI), MRI is currently considered as a promising preoperative staging method with the best specificity and sensitivity in detecting breast tumors (6). In recent years, diffusion-weighted MRI (DW-MRI), a noninvasive method that uses the diffusion of water in vivo to generate contrast in MR images, has also been used in breast cancer analysis. DW-MRI can accurately portray the microscopic anatomy of a target tissue or organ by measuring the motion of water molecules and providing dynamic MRI with supplementary data (8). The water mobility in tissue mainly depends on the number and morphology of cells, their relative arrangement, and the extracellular matrix in which the cells are embedded. The apparent diffusion coefficient (ADC) is the unit of measurement used to quantify water diffusion in tissue (9–12).
Dynamic contrast-enhanced MRI (DCE-MRI) is another prospective method for the detection, diagnosis and treatment of breast carcinoma. It allows to obtain and analyze both morphological features and functional aspects related to malignant growth by comparing the temporal and spatial uptake of contrast medium and estimating the degree of malignancy in detecting margins of breast masses (13). Meanwhile, predictive and prognostic cancer factors including ER, PR, HER2 and Ki-67 have been regularly connected with MRI features to offer sufficient information for better diagnosis and treatment in patients with breast cancer (13).
In this study, we analyzed the correlation of DCE/DW-MRI imaging features and ADC values with cell molecular phenotypes for the sake of evaluating the diagnostic value of MRI in breast cancer.
Materials and methods
Subjects and lesions
This study included 117 female patients who underwent preoperative breast DW/DCE-MRI between May 2014 and November 2016 at Zhejiang Cancer Hospital. Among these patients, those who showed a non-mass enhancement pattern on MRI (n = 8) and those with inflammatory breast lesions (n = 3) were excluded, resulting in a total of 106 female study participants. Out of these 106 patients, 118 lesions were analyzed and 12 patients had 2 or more lesions proved pathologically. The study was conducted with the approval of the institutional review board and ethics committee of Zhejiang Cancer Hospital. All patients were informed about the procedures prior to examination and signed the corresponding informed consent forms.
MRI image acquisition
MRI was performed on a 1.5T MR System (GE Medical Systems), using a bilateral 8-channel breast coil. Patients were scanned in the prone position, with the bilateral breasts naturally suspended in the breast coil. The main parameters of the scanning protocol are depicted in Table I. Intravenous injection was started with Gd-DTPA (0.1 mmol/kg, Schering Corporation) and continued with the same volume of normal saline using an indwelling needle at an injection rate of 2.0 mL/s.
Main parameters of scanning MRI protocol
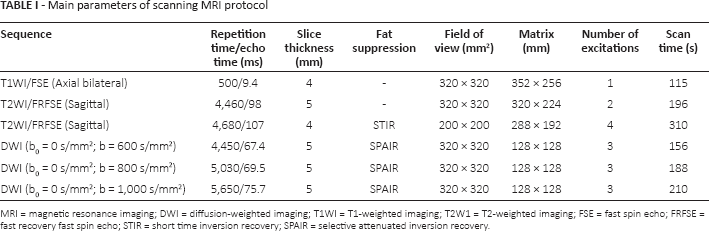
MRI = magnetic resonance imaging; DWI = diffusion-weighted imaging; T1WI = T1-weighted imaging; T2W1 = T2-weighted imaging; FSE = fast spin echo; FRFSE = fast recovery fast spin echo; STIR = short time inversion recovery; SPAIR = selective attenuated inversion recovery.
ADC measurement
The ADC values of each lesion were automatically measured using the Functool 2 software of the GE ADW 4.1 workstation through placing regions of interest (ROIs) manually on the consistent lesion of ADC maps. An oval-shaped ROI was created as large as possible while avoiding hemorrhagic and necrotic portions. The ADC values were measured at
Morphological evaluation
Two researchers who had more than 5 years' experience in performing breast MR imaging reviewed the retrospective MR images in consensus. The readers were blinded to the clinical history and pathology results before DCE-MRI analysis. The malignant lesions' margins (i.e., smooth, lobulated, irregular and speculated) and enhancement patterns (i.e., homogeneous, heterogeneous and rim) were assessed on the basis of the Breast Imaging Reporting and Data System (BI-RADS) for MRI (14).
In DCE-MRI, assessment of the dynamic curve type was conducted using a semiquantitative method that had been illustrated in a previous work (15). The dynamic curves were categorized into 3 types (persistent enhancement, plateau and washout), with 5% as the cutoff value. A signal intensity percentage change >5% was classified as persistent enhancement, while a change within 5% was regarded as a plateau and a change <5% was classified as washout.
Statistical analysis
All of the observed data were analyzed using the SPSS 17.0 statistical software in conjunction with the GraphPad Prism software. Continuous data were presented as means ± standard deviations and compared using the paired Student's
Results
Clinical demographics
A total of 68 (57.6%) tumors found in 58 patients were confirmed to be malignant and the remaining 50 (42.4%) lesions detected in 48 patients were diagnosed as benign (Tab. II). Patients with malignant lesions and those with benign lesions were matched in mean age (p = 0.387) and menopausal status (p = 0.642). In addition, malignant and benign lesions were distributed evenly in laterality (p = 0.667), yet were significantly disparate in mean size (p<0.001). According to lesion histology, the 68 malignant lesions were categorized into 44 invasive ductal carcinomas (IDCs), 10 invasive lobular carcinomas (ILCs), 6 ductal carcinomas in situ (DCIS), and 8 others. Based on tumor node metastasis (TNM) stage, 32.35%, 45.59% and 22.06% of lesions were classified as stage I, stage II and stage III, respectively. Furthermore, a large portion of malignant lesions were found to have positive ER status (66.18%), PR status (57.35%) and Ki-67 status (72.06%), while negative HER2 status was observed in the majority (67.65%) of malignant lesions.
Demographic and clinical characteristics of study participants
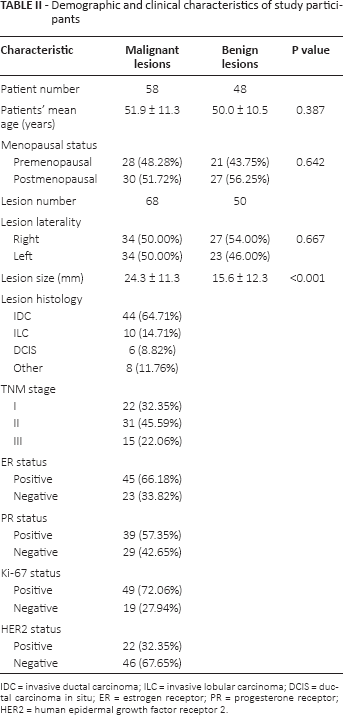
IDC = invasive ductal carcinoma; ILC = invasive lobular carcinoma; DCIS = ductal carcinoma in situ; ER = estrogen receptor; PR = progesterone receptor; HER2 = human epidermal growth factor receptor 2.
Analysis of ADCs
As demonstrated in Figure 1A, among the 3
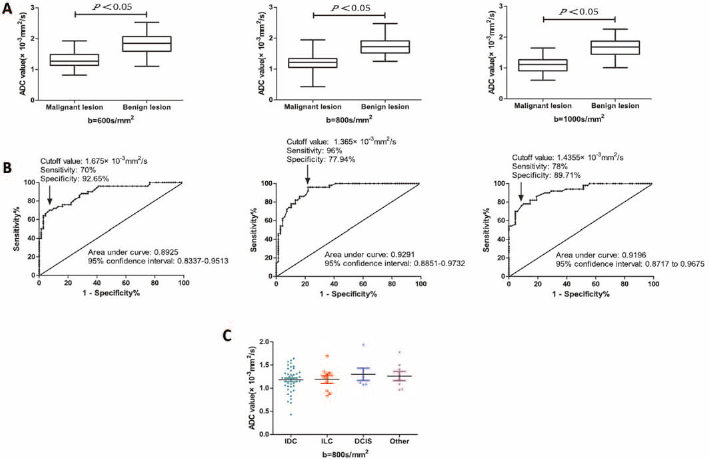
Box plot showing the comparison of average ADC values between malignant and benign breast lesions at
MRI morphological evaluation
As shown in Table III, lesion margins, lesion enhancement patterns and dynamic curve types were different between malignant and benign breast lesions. Compared with benign lesions, malignant lesions were more likely to have spiculated margins (55.88% vs. 20.00%) and heterogeneous enhancement (54.41% vs. 26.00%) (both p<0.001). In dynamic imaging, malignant lesions were more likely to exhibit washout curves (57.41% vs. 20.00%), while benign lesions were more prone to exhibiting persistent enhancement (64.00% vs. 14.71%) (p<0.001).
Comparison of MRI-based characteristics between malignant and benign breast lesions
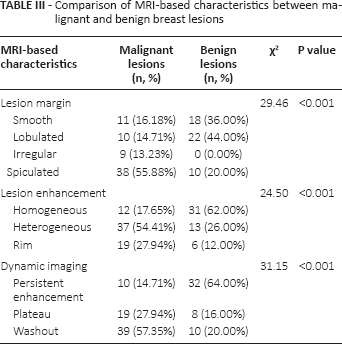
Table IV presents the relationship between MRI morphological features and clinical characteristic of malignant lesions, such as histology and TNM stage. The proportion of spiculated margins among IDC lesions (70.46%) was much larger than that among ILC and DCIS lesions (30.00% and 33.34%, respectively). Irrespective of the histological type of carcinoma, heterogeneous enhancement appeared as the major pattern of lesion enhancement (≈50.00%). In dynamic imaging, invasive carcinomas (IDC and ILC) mainly displayed washout imaging features, while persistent enhancement, plateau and washout were evenly distributed in DCIS. Furthermore, with increasing TNM stages lesions were more characterized by spiculated margins (TNM I: 36.36%, TNM II: 67.74%, TNM III: 60.00%), heterogeneous enhancement (TNM I: 54.54%, TNM II: 48.39%, TNM III: 66.67%) and washout imaging features (TNM I: 40.91%, TNM II: 61.29% TNM III: 73.33%). Lesions with positive ER status exceeded those with negative ER status in the proportion of smooth margins, homogeneous enhancement and persistent enhancement imaging; these tendencies were similar with respect to PR status. Ki-67 positivity was more likely to be found among lesions with spiculated magins, rim enhancement and washout imaging, while HER2 status seemed to be evenly distributed regardless of lesion margins, lesion enhancement and dynamic imaging.
Comparison of MRI morphological features and clinical characteristic of malignant breast lesions
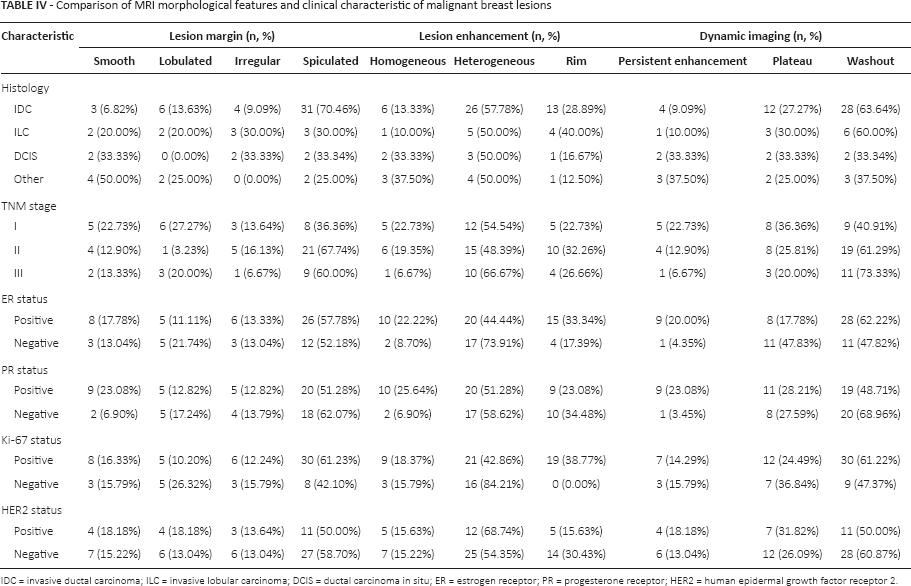
DC = invasive ductal carcinoma; ILC = invasive lobular carcinoma; DCIS = ductal carcinoma in situ; ER = estrogen receptor; PR = progesterone receptor; HER2 = human epidermal growth factor receptor 2.
Spearman correlation analyses
ADC values were uncorrelated with histological types and TNM stages of breast cancer (Tab. V), and significant correlations were also not found between ADC values and immunohistochemical features including ER, PR, Ki-67 and HER2 (all p>0.05). There was a significantly negative correlation between PR status and dynamic imaging (rs = -0.269, p = 0.027), which indicated that malignant lesions with stronger PR expression are more likely to show persistent enhancement curves in dynamic imaging. In addition, a significant positive correlation was found between Ki-67 status and lesion enhancement (rs = 0.243, p = 0.045), which revealed that malignant lesions with stronger Ki-67 expression were more likely to show rim enhancement. Furthermore, distinct histologies of breast cancer also rendered lesion margins specific (rs = -0.444, p<0.001), and dynamic imaging could suggest TNM stages of breast cancer patients in advance (rs = 0.253, p = 0.037).
Spearman's correlation analyses of MRI-based features and tumor characteristics (histology and TNM stage) as well as immunohistochemical features (ER, PR, Ki-67 and HER2 status)
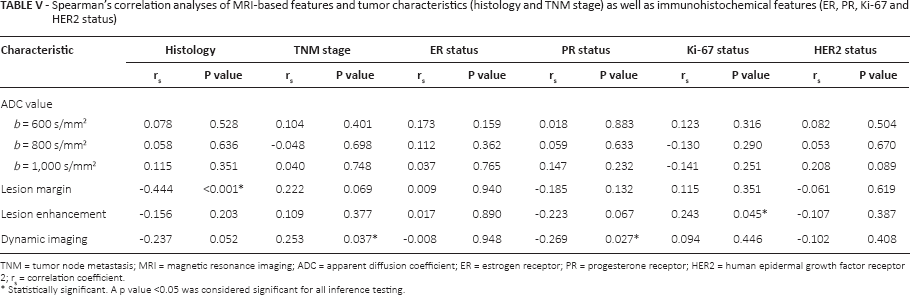
TNM = tumor node metastasis; MRI = magnetic resonance imaging; ADC = apparent diffusion coefficient; ER = estrogen receptor; PR = progesterone receptor; HER2 = human epidermal growth factor receptor 2; rs = correlation coefficient.
Statistically significant. A p value <0.05 was considered significant for all inference testing.
Discussion
The present study investigated the relationship between pathological characteristics and diffusion imaging in breast cancer based on the assumption that different tumor micro-environments might be related to different diffusion properties of water molecules (17). Several previous articles have reported the relationship between breast cancer DCE-MRI features and ADC or MRI morphological or immunohistochemical features (17, 18). Our study based on a quantitative analysis of the MRI data might provide more objective and detailed information on the MRI-based features of breast cancer and their diagnostic and prognostic values.
In this study, we demonstrated that lesion sizes and ADC values in benign and malignant lesions had significant differences. Specifically, the mean ADC value of the malignant group was lower than that of the benign group, and Belli et al (9) reported similar results with respect to ADC values. Our study also found that ADC values were significantly different between IDC and DCIS, which is consistent with previous findings (17). The result was reinforced by the ROC curve, which identified 800 s/mm2 as the optimal cutoff value that maximized the sensitivity of detecting lesions. Intriguingly, the cutoff value of
Furthermore, analyzing ADC values with DCE-MRI would contribute to the understanding of the accordance between comprehensive radiological and pathological findings, and ADC values can also be a prospective prognostic parameter (9). Apart from that, several studies have illustrated that lower ADC values in breast malignancies were mainly due to higher cell density, histological grade, mitotic rates and nuclear/cytoplasmic ratios (17, 22). In tumor tissues with higher cell density the extracellular space decreases, which in turn restricts the microcellular water motion, and thus MRI displays lower ADC values (12, 17, 23, 24). It was also suggested that the growth of malignant tumors not only depended on the cancer cells themselves but was also affected by the tumor microenvironment, which was featured by a complicated system including tumor cells, extracellular matrix and stromal cells (e.g., fibroblasts, adipocytes, infiltrating immune and endothelial cells) (17). According to the findings of other studies, the stroma in breast cancer is different from that in normal breast, exhibiting a unique pattern that could stimulate cell proliferation, adhesion, invasion and extracellular matrix remodeling (25–28). It was also suspected that all the features of the tumor microenvironment played a part in restraining the diffusion of water molecules (17, 22, 29, 30).
In this study, we also discovered that malignant lesions were more likely to have spiculated margins, heterogeneous enhancement and washout curves, especially invasive tumors such as IDC and ILC. It was suggested that the existence of augmented vessel density and arteriovenous anastomoses with fast outflow based on biology and pathophysiology could account for the washout phenomenon (29). Kuhl et al (31) pointed out that the washout curve is a significant indication of malignancy, though its predictive power is not sensitive. The heterogeneous rim enhancement may be explained by massive angiogenesis in the tumor periphery, central necrosis and desmoplasia. These results demonstrated that ADC values and MRI morphological differences could be used to further enhance the diagnostic results.
Finally, we also found that lesion enhancement for roundness of breast tumor was positively correlated with Ki-67 index status, a biomarker of cell proliferation (32). This finding is concordant with previous biological reports which suggested that higher expression of Ki-67 was associated with stronger proliferative ability, higher cellularity and inflammatory host reactions (33). In addition, there was a significant positive correlation between Ki-67 status and lesion enhancement, which may reveal that malignant lesions with stronger Ki-67 expression were more likely to show rim enhancement. This study might imply that malignant lesions with stronger PR expression were more likely to show persistent enhancement curves. Leong et al (34) reported that the type of MRI dynamic curve was related to the PR expression. This study also demonstrated that irregular tumor morphology could predict negative expression of ER, which is consistent with the previous report indicating that a higher grade of heterogeneity was associated with negative ER status. ER status in breast cancer is currently used to select patients for hormonal therapy and it also provides rich prognostic information (35). Moreover, ER status was also reported to be associated with high angiogenesis in the tumor periphery, which may be explained by central tumor necrosis (36).
There were several limitations to this research. For instance, this was a relatively limited study which included few tumor subtypes. Moreover, we analyzed only a few prognostic factors without exploring the molecular profiles of single breast cancers. Finally, DW-MRI of breast cancer should be validated using larger studies in clinical practice since this was merely a preliminary study.
Conclusion
In conclusion, both ADC values and MRI morphological assessment could be used to distinguish malignant breast lesions from benign ones. Such an approach provides a reasonable foundation for assessing the type and histological grade of breast cancer.
Footnotes
Abbreviations
Financial support: This work was supported by a grant from Cultivation of High-level Innovation Health Talents of ZheJiang (#2012-241).
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
