Abstract
Background:
The prognostic value of immune cells expressing programmed cell death 1 (PD-1) and PD-1 ligand 1 (PD-L1) in cancer are controversial, and the potential differential impact of using tissue microarrays and whole tissue sections to assess the positivity of immune cells has not been addressed.
Methods:
The current study included 30 eligible studies with 7251 patients that evaluated the relationship between tumor-infiltrating lymphocytes expressing PD-1/PD-L1 and overall survival and disease-free survival, or progression-free survival. Subgroup analysis was based on the tissue type of cancer and the type of tissue sampling (tissue microarray or whole tissue section).
Results:
In the meta-analysis, PD-1-positive and PD-L1-positive tumor-infiltrating lymphocytes had a positive effect on disease-free survival or progression-free survival (hazard ratio [HR] 0.732; 95% confidence interval [CI] 0.565, 0.947; and HR 0.727; 95% CI 0.584, 0.905, respectively). PD-L1-positive tumor-infiltrating lymphocytes had a positive impact on overall survival in studies using tissue microarray (HR 0.586; 95% CI 0.476, 0.721), but had a poor impact when only whole tissue sections were considered (HR 1.558; 95% CI 1.232, 1.969). Lung cancer was associated with good overall survival and disease-free survival (HR 0.639; 95% CI 0.491, 0.831; and HR 0.693; 95% CI 0.538, 0.891, respectively) for PD-1-positive tumor-infiltrating lymphocytes, and colorectal cancer showed favorable disease-free survival (HR 0.471; 95% CI 0.308, 0.722) for PD-L1-positive tumor-infiltrating lymphocytes.
Conclusion:
Immune cells expressing PD-1 and PD-L1 within tumors are associated with the prognosis. However, the correlation may vary among different tumor types and by the type of tissue sampling used for the assessment.
Introduction
The immune system plays a central role in the tumor microenvironment. 1 Evidence indicates that a signaling pathway involving programmed cell death 1 (PD-1) and PD-1 ligand 1 (PD-L1), namely the PD pathway, acts as an important component of immunosuppression, and that blocking the PD pathway may lead to successful treatment of tumors via immune checkpoint inhibition. 2 Multiple clinical trials targeting PD-1 or PD-L1 are currently enrolling 3 and several inhibitors of PD-1 and PD-L1 have been approved by the U.S. Food and Drug Administration (FDA) for the treatment of specific advanced cancers. 4 However, it remains uncertain which biomarker best predicts the response to anti-PD immunotherapies.
Previous studies have identified immune cells, including tumor-infiltrating lymphocytes (TIL) and tumor-associated macrophages (TAM), as putative prognostic markers in solid malignant tumors such as gastric, colorectal, urothelial, and breast cancers.4-6 TIL and TAM also play a major role in PD-1/PD-L1 expression and in the prediction of response to anti-PD pathway therapies. 7 However, unlike their tumor cell counterpart, the prognostic implications of PD-1/PD-L1 expression in immune cells have not been thoroughly evaluated through a meta-analysis. Furthermore, the potential impact of tissue sampling, tissue microarray (TMA) or whole section, on immunohistochemical analysis of PD-1 and PD-L1 expression on immune cells has not been addressed.
Our goal here is to comprehend the predictive role of PD-1/PD-L1 expression on immune cells as a biomarker across multiple tissue types of cancer. Therefore, we performed a meta-analysis to evaluate the relationship between PD-1/PD-L1 expression on immune cells and prognosis in solid tumors.
Material and methods
Literature search and study selection
Articles relevant to the subject were retrieved from PubMed, Embase, and Web of Science. The search terms were “PD-1”, “programmed cell death 1”, “PD-L1”, “PD-1 ligand”, “B7-H1”, “CD274”, “carcinoma”, “malignant tumor”, “immune cell”, “survival”, and “prognosis”.
Studies eligible for inclusion in this meta-analysis were those that analyzed the relationship between the survival of patients with solid malignant tumor and the PD-1/PD-L1 expression status of immune cells within tumors, specifically TILs. The studies that did not specify the type of tumor-infiltrating immune cells with expression of PD-1/PD-L1 were also included and classified as studies evaluating the prognostic implications of PD-1/PD-L1 expression on immune cells not otherwise specified (ICNOS). The expression of PD-1/PD-L1 within the immune cells was determined by immunohistochemistry and was divided into positive (high) or negative (low) categories according to the criteria of each article. Patient prognosis was assessed by overall survival (OS) and disease-free survival (DFS) or progression-free survival (PFS). We only considered full articles published in English. Studies with insufficient survival data, insufficient pathological data, and tumors evaluated after neoadjuvant therapy (including previous history of PD-1 or PD-L1 inhibitors) were excluded.
Data extraction and quality assessment
Two authors (Y.K. and X.W.) independently searched the relevant databases according to the inclusion and exclusion criteria. We extracted the following data from the eligible studies: name of the first author, year of publication, primary organ examined, subtype of cancer, type of evaluated immune cells, type of outcome, clone of antibody used for immunohistochemistry, criteria for positivity, number of evaluated patients, the usage of TMA or whole tissue section, the number of cores for each sample when TMA was used, and survival associated with PD-1/PD-L1 expression on immune cells (TIL or ICNOS).
Survival data were primarily extracted as hazard ratio (HR) and 95% confident intervals (CI) from Cox univariate analysis. If the HR and 95% CI were not directly available from the articles and supplementary data, we contacted the author of each article to request this information or estimated HR and 95% CI from the Kaplan–Meier survival curves using the Engauge Digitizer software version 9.8 (http://digitizer.sourceforge.net/) and the spreadsheet provided by Tierney et al. 8
Statistical analysis
Comprehensive meta-analysis software Version 3.3 (Biostat, Englewood, NJ, USA) was used for the statistical analysis. Statistical heterogeneity was assessed by the Cochran’s Q and I2. When more than four studies were available, we considered P values <0.05 or I2 >50% as statistically significant heterogeneity and applied the random effects model. Subgroup analysis was conducted when three or more effect sizes (ES) shared a common section type (TMA or whole tissue section), or a common tissue type of cancer with the same outcome. Sensitivity analysis was applied by eliminating each ES one at a time. A graphical funnel plot was used to evaluate potential publication bias.
Results
Eligible studies and study characteristics
After searching PubMed, Embase, and Web of Science, 2608 records were found. Among these, 1913 titles were excluded due to lack of relevance, and 207 titles were excluded because they were not full-length research articles written in English. The remaining 488 articles were reviewed in detail, and 30 articles were selected as eligible for the meta-analysis (Figure 1).
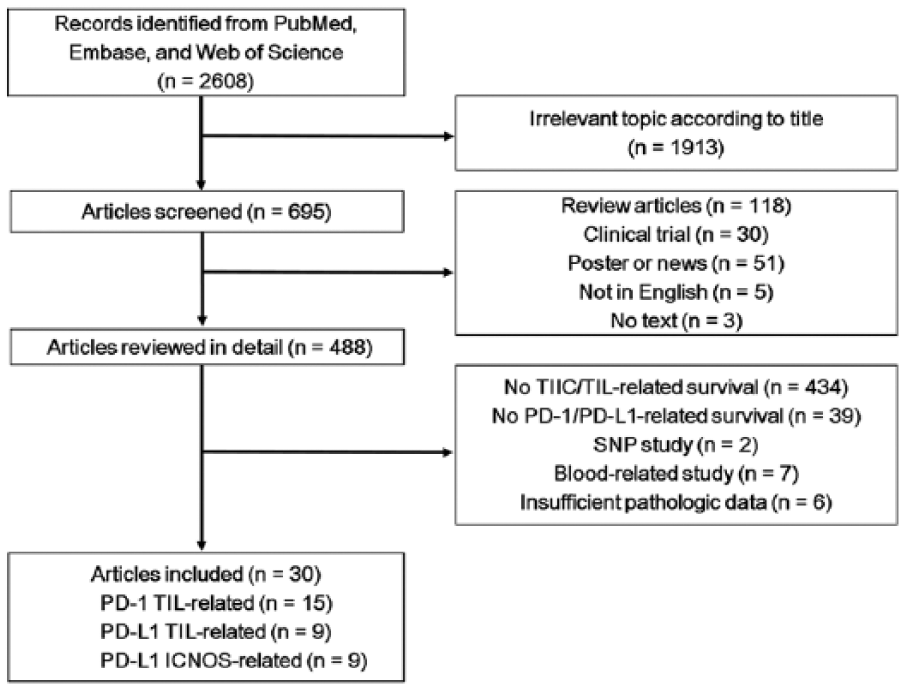
Flow chart of literature search and study selection.
A total of 27 ES were extracted from 15 studies to evaluate the correlation between prognosis and PD-1-positive TIL (Table 1). A total of 25 ES were extracted from 18 studies (nine PD-L1 TIL-related studies and nine PD-L1 ICNOS-related studies) that showed the correlation between prognosis and PD-L1-positive immune cells (Table 2). Patient numbers ranged from 36 to 660 for each study, and 7251 patients were included in the total analysis. Most studies focused on a single cancer patient cohort, but studies by Li et al. 1 and Lee et al. 27 analyzed two separate groups of colorectal carcinoma (CRC).
Characteristics of studies eligible for examination of PD-1 expression in immune cells.
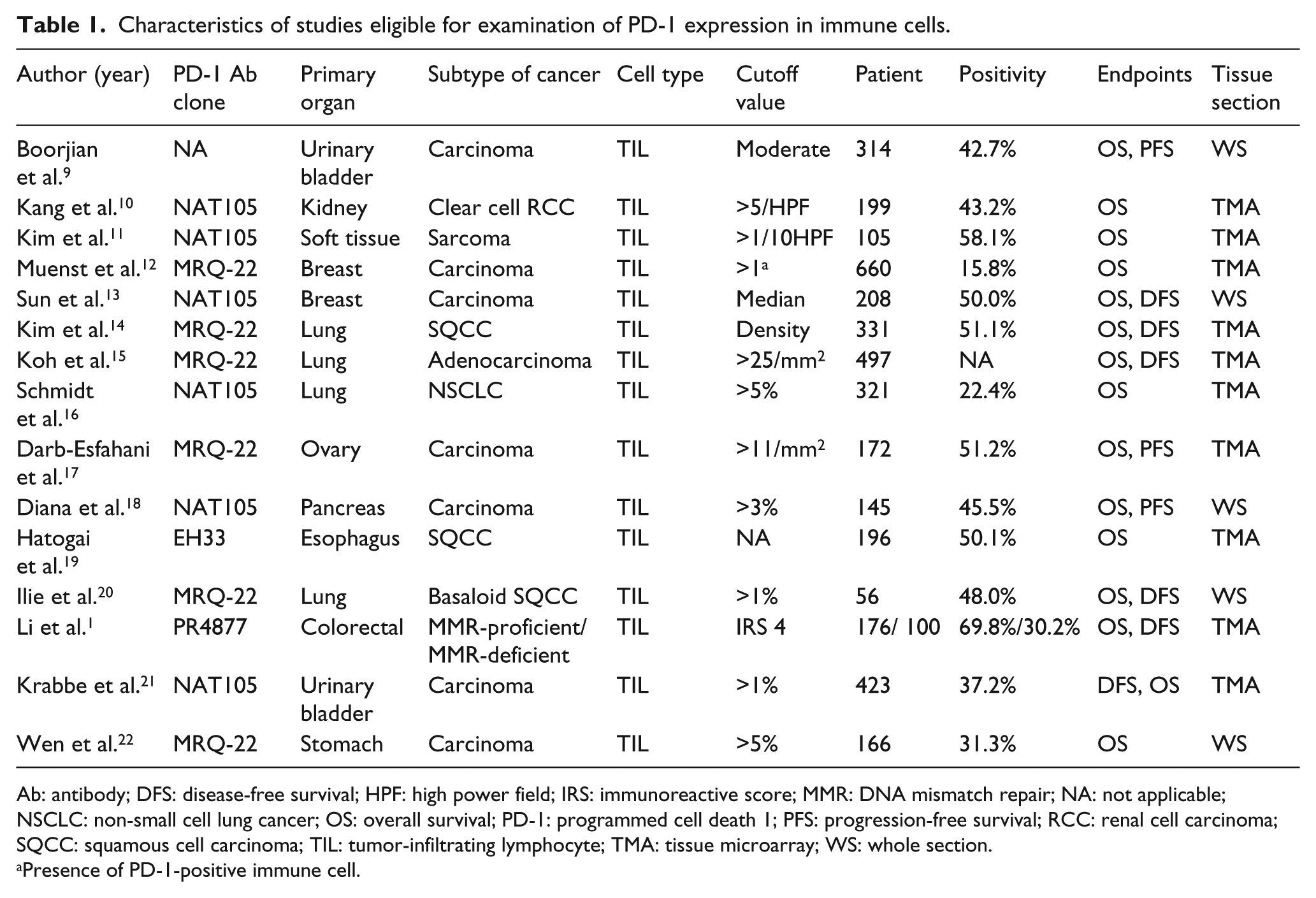
Ab: antibody; DFS: disease-free survival; HPF: high power field; IRS: immunoreactive score; MMR: DNA mismatch repair; NA: not applicable; NSCLC: non-small cell lung cancer; OS: overall survival; PD-1: programmed cell death 1; PFS: progression-free survival; RCC: renal cell carcinoma; SQCC: squamous cell carcinoma; TIL: tumor-infiltrating lymphocyte; TMA: tissue microarray; WS: whole section.
Presence of PD-1-positive immune cell.
Characteristics of studies eligible for examination of PD-L1 expression in immune cells.
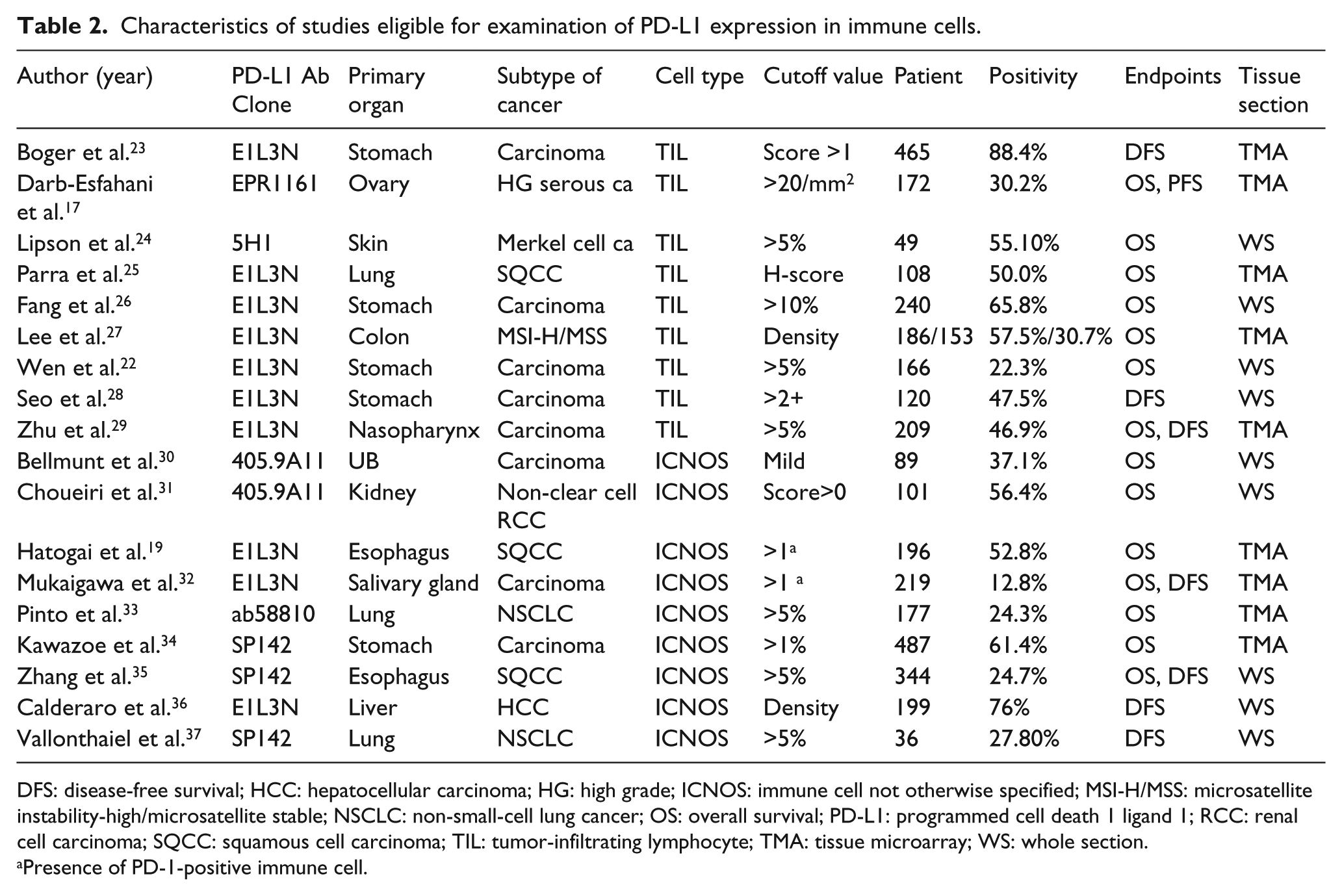
DFS: disease-free survival; HCC: hepatocellular carcinoma; HG: high grade; ICNOS: immune cell not otherwise specified; MSI-H/MSS: microsatellite instability-high/microsatellite stable; NSCLC: non-small-cell lung cancer; OS: overall survival; PD-L1: programmed cell death 1 ligand 1; RCC: renal cell carcinoma; SQCC: squamous cell carcinoma; TIL: tumor-infiltrating lymphocyte; TMA: tissue microarray; WS: whole section.
Presence of PD-1-positive immune cell.
Meta-analysis
In the overall population, 16 ES did not show a significant correlation between PD-1-positive TILs and OS (HR 1.123; 95% CI 0.781, 1.613; Figure 2(a)). Eleven ES extracted from 10 studies demonstrated a significant correlation between PD-1-positive TILs and good DFS or PFS (HR 0.732; 95% CI 0.565, 0.947; Figure 2(b)). Similarly, 9 ES showed that PD-L1-positive TILs were not associated with OS (HR 0.708; 95% CI 0.477, 1.052; Figure 2(c)) and 4 ES showed that PD-L1-positive TILs were associated with good DFS or PFS (HR 0.727; 95% CI 0.584, 0.905; Figure 2(d)) in the overall population. PD-L1-positive ICNOSs were not associated with the prognosis regardless of survival type (Supplementary Figure 1).
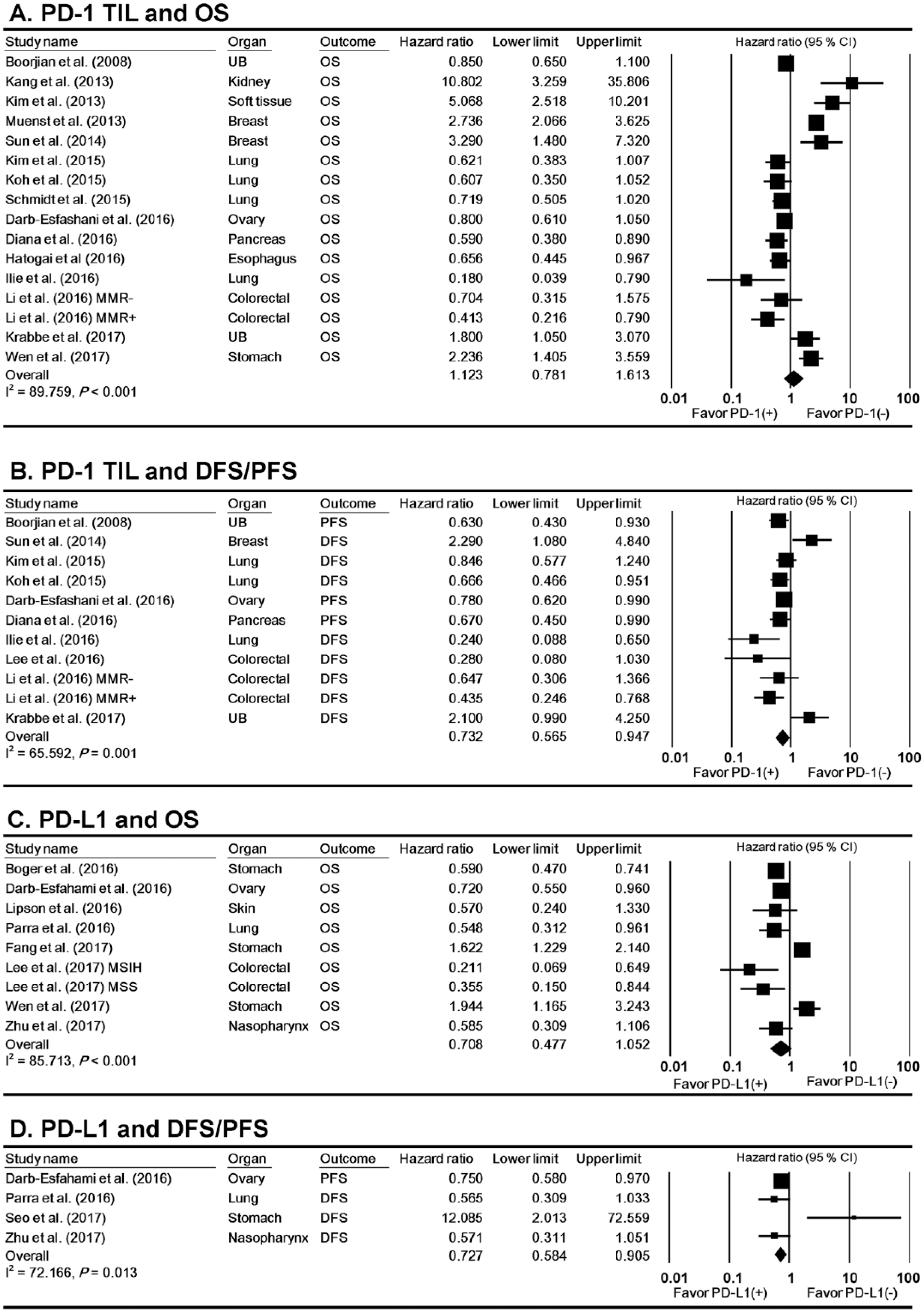
Forest plot describing the correlation between PD-1-positive TILs and OS (a) and DFS or PFS (b) as well as the correlation between PD-L1-positive TILs and OS (c) and DFS or PFS (d).
Subgroup analysis, sensitivity analysis, and publication bias
TMA has been commonly used for the evaluation of cancer samples due to the understanding that molecular targets with therapeutic meaning will be relatively uniformly expressed in cancer tissues, 4 but it is unclear if a small number of TMA cores is suitable for analyzing the heterogeneity present in intratumoral immune cells. However, 56.7% (17/30) of the eligible studies were based on TMA, and 50.0% (15/30) of the eligible studies used only one or two cores per sample. To this end, a subgroup analysis was conducted for studies with identical slide material (TMA or whole tissue section) to propose potential discrepancies. The subgroup analysis—according to the type of tissue sampling—did not have a significant impact on the correlation between PD-1-positive TIL and survival (Table 3). However, PD-L1 positive TILs showed contrasting results in the relationship with survival between TMA and whole tissue sections; PD-L1-positive TILs were associated with favorable OS (HR 0.586; 95% CI 0.476, 0.721) in studies using TMAs, but were associated with unfavorable prognosis in studies using whole tissue sections (HR 1.558; 95% CI 1.232, 1.969). In contrast with the prognostic implications of PD-L1-positive TILs in studies using whole tissue sections, patients with PD-L1-positive ICNOSs showed favorable OS (HR 0.733; 95% CI 0.578, 0.928) and favorable DFS or PFS (HR 0.644; 95% CI 0.500, 0.830) in studies using whole tissue sections (Supplementary Figure 2).
Subgroup analysis of included studies.
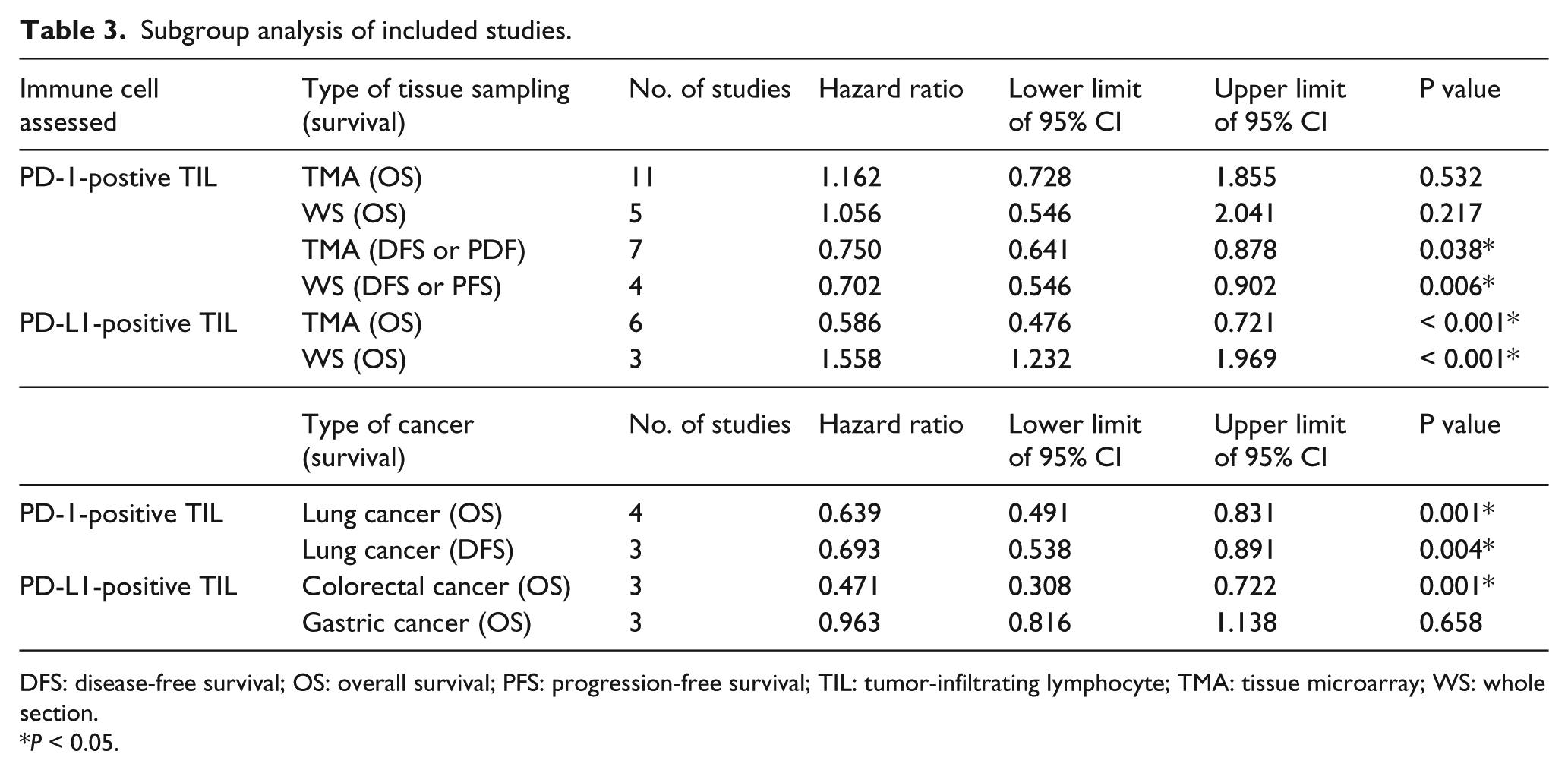
DFS: disease-free survival; OS: overall survival; PFS: progression-free survival; TIL: tumor-infiltrating lymphocyte; TMA: tissue microarray; WS: whole section.
P < 0.05.
Subgroup analysis was further conducted according to tissue type of cancer (Table 3). In lung cancer, PD-1-positive TILs were associated with favorable OS (HR 0.639; 95% CI 0.491, 0.831) and DFS (HR 0.693; 95% CI 0.538, 0.891). PD-1-positive TILs were correlated with favorable DFS in CRC (HR 0.471; 95% CI 0.308, 0.722) but failed as a prognostic marker in gastric cancer (HR 0.963; 95% CI 0.816, 1.138).
A sensitivity analysis indicated that exclusion of a single study did not affect the correlation between PD-1-positive TILs and survival with the exception of a study by Li et al. 1 (Supplementary Figure 3). When we eliminated studies that used whole tissue sections, the positive correlation between PD-L1-high TILs and OS became evident. Funnel plots for both PD-1 TILs and PD-L1 TILs results suggest that no publication bias is present in this study (Supplementary Figure 4).
Discussion
It remains uncertain which marker is most efficient for predicting the effect of the PD pathway blockade. Putative candidate markers include Bim, interferon-Ɣ, CTLA-4, PD-1, and PD-L1. 3 To complicate matters, some studies propose that PD-L1 expression on immune cells, especially TILs, better predicts the response of anti-PD-L1 than PD-L1 expression on tumor cells in multiple tissue types of malignancy. 5 For atezolizumab—an anti-PD-L1 drug approved by the FDA for metastatic bladder cancer—PD-L1 expression on TIL was considered as the only biomarker that predicted treatment response and outcome. 6 Unfortunately, providing the PD-1/PD-L1 expression status of immune cells is much more difficult than determining the PD-L1 status of tumor cells and requires a larger amount of tissue. In this study, we conducted a meta-analysis to determine the relationship between PD-1/PD-L1-postive immune cells and prognosis within solid tumors while considering whether TMA or a whole tissue section was used for immunohistochemical analysis.
In the meta-analysis of the overall population, a positive correlation between PD-1-positive TILs and DFS or PFS was observed. This partially contradicts findings by Zhang et al., 7 which showed an inverse correlation between PD-1-positive TILs and survival. However, the primary outcome of our findings was specific to DFS or PFS, while the primary outcome of the study conducted by Zhang et al. 7 was OS. Moreover, our study included all studies analyzed by Zhang et al. 7 and additional ES, which indicate that there was no significant correlation between PD-1-positive TILs and OS.
While PD-L1 expression in tumor cells indicates poor prognosis,7,29,38-42 the prognostic implications of PD-L1-positive immune cells (TILs and ICNOSs) were different between TMA and whole tissue sections evaluations and became significant when studies using whole tissue sections were separated from studies using TMA. These findings indicate that prognostic implications measured by TMAs and whole tissue sections could be considered as separate entities since TMA only represents a punched fraction of the whole slide, which could lead to increased subjectivity of TMA results especially if the number of cores per sample is limited. These differences could undermine the correlation between PD-L1-positive immune cells and prognosis when evaluated together. PD-L1-positive TILs in gastric cancer were not associated with prognosis. However, this is mostly due to an insufficient number of studies that use whole tissue sections, which further emphasizes the importance of whole sections in evaluating PD-L1 in immune cells. The mechanism of PD-L1-expressing TILs impacting prognosis is still elusive. Since PD-L1 expression on TILs has been strongly correlated with the density of CD4- or CD8- positive TIL,27,43 it has been suggested that the expression of PD-L1 on TILs may represent effective host immune responses in the presence of a favorable immune microenvironment abundant with CD4- and CD8-positive T cells and its cytokine milieu, which may lead to restraining of tumor growth.17,44
The limitation of our study is that none of the studies used the double staining immunohistochemistry method to distinguish PD-1/PD-L1-positive TIL from other immune cells (including macrophages and dendritic cells), which could have led to an overestimation of positive samples. Moreover, we calculated some HRs from Kaplan–Meier survival curves, which could have made the results less reliable.
In conclusion, we confirmed that PD-1/PD-L1 expression on tumor-infiltrating immune cells could be used as a biomarker to predict the prognosis of patients with cancer in clinical practice; however, the type of tissue section used for immunohistochemistry could have an impact on the result.
Supplemental Material
Supplementary_Figures – Supplemental material for Intratumoral immune cells expressing PD-1/PD-L1 and their prognostic implications in cancer: a meta-analysis
Supplemental material, Supplementary_Figures for Intratumoral immune cells expressing PD-1/PD-L1 and their prognostic implications in cancer: a meta-analysis by Younghoon Kim, Xianyu Wen, Nam Yun Cho and Gyeong Hoon Kang in The International Journal of Biological Markers
Footnotes
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the National Research Foundation (NRF) funded by the Korean Ministry of Science, ICT, and Future Planning (2011-0030049) (2016M3A9B6026921) and a grant (2320160090) from the SNUH Research Fund.
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
