Abstract
Objective
Homologous recombination (HR) is frequently impaired in sporadic high-grade serous ovarian carcinoma (sHGSOC) due to deficiencies in BRCA1/2 genes, a situation associated with hypersensitivity to platinum compounds. Alterations in other genes can also cause HR deficiency. Preclinical data show that RAP80 is an HR–pathway-related gene that influences BRCA1 activity. RAP80 has been reported to affect outcome in some solid neoplasms. This study investigates the role of RAP80 in sHGSOC survival.
Methods
mRNA expression of RAP80 was analyzed in tumor samples from 35 patients who postoperatively received standard platinum-based chemotherapy. The effects of RAP80 expression on progression-free survival (PFS) and overall survival (OS) were examined by means of Cox regressions. The clinical variables known to have prognostic value (FIGO stage, residual disease at surgery, and debulking surgery) were included as covariates in the analysis. BRCA1 was analyzed given the moderate correlations with RAP80.
Results
Median follow-up, PFS and OS were 61.3, 20.2 and 62.8 months, respectively. Low RAP80 expression levels were associated with shorter PFS (HR = 1.449, p = 0.007) and OS (HR = 1.331, p = 0.047).
Conclusions
This is the first study to show a potential prognostic role of RAP80 expression in patients with HGSOC. The results suggest that HR deficiency due to low RAP80 expression is not associated with hypersensitivity to platinum compounds in sHGSOC.
Introduction
High-grade serous ovarian cancer (HGSOC) is the most lethal gynecological malignancy (15,000 deaths per year in the US alone) (1) and accounts for 70% of all ovarian cancers. The standard of care for HGSOC is systematic pelvic and abdominal debulking surgery aiming to remove all peritoneal implants usually present, followed by chemotherapy with carboplatin and paclitaxel, which has been clearly shown to extend survival (2). However, 5-year overall survival (OS) remains just 40%-45% (3). Two main molecular features of HGSOC are TP53 mutations, found in 96% of HGSOCs, and a defective homologous recombination pathway, which may be present in up to 50% of all cases (4). Both features contribute to the high level of genome instability shown by HGSOC, which is a major characteristic of this disease.
Homologous recombination (HR), the main repair mechanism of DNA double-strand breaks (DSBs) including those caused by platinum, acts by building a homology-directed copy of the sister chromatid during the S and G2 phases. The genes BRCA1 and, to a lesser extent, BRCA2 play a key role in HR. BRCA1 is present in different protein macrocomplexes localized at the DSBs and is involved in specific steps of the DNA damage response (5, 6). BRCA1/2 deficiency is associated with decreased HR and with breast and ovarian carcinogenesis (5). The study of these genes was originally linked to the hereditary syndrome of breast and ovarian cancers. Indeed, germline BRCA1/2 mutations are present in 50% of hereditary ovarian cancers (which, in turn, account for 10%-15% of all HGSOCs). In addition, a small proportion of sporadic HGSOCs (<7%) also show truncating somatic mutations (4). Strong evidence indicates that BRCA1/2 mutations confer sensitivity to platinum compounds and improved survival on HGSOC patients (7, 8), and they are the only clear predictive factor of response to olaparib, which is the first PARP inhibitor approved for HGSOC treatment (9).
However, BRCA1 inactivation in sporadic HGSOC is mainly driven by promoter hypermethylation, which occurs in ∼15% of these cases (4, 10, 11). BRCA1 promoter hypermethylation is usually associated with loss of heterozygosity and total BRCA1 inactivation leads to very low or undetectable BRCA1 expression (12). Preclinical data in several solid tumors indicate that low BRCA1 expression confers sensitivity to platinum (13, 14) but resistance to taxanes (15, 16), suggesting that BRCA1 expression can differentially modulate response to chemotherapy (17, 18). These preclinical findings have been borne out by several clinical reports. While low BRCA1 mRNA expression levels have been demonstrated to be associated with longer OS after cisplatin-based neoadjuvant chemotherapy in patients with non-small cell lung cancer (NSCLC) and bladder cancer (19, 20), higher BRCA1 mRNA levels were related to longer survival in docetaxel-treated patients with metastatic NSCLC or gastric cancer (21). To date, however, the prognostic impact of BRCA1 expression in sporadic HGSOC after first-line chemotherapy remains unclear, suggesting a different survival impact derived from BRCA1 silencing in comparison to BRCA1/2 mutations (22–24). It is relevant to remark that standard first-line therapy for HGSOC is the carboplatin and paclitaxel combination, as stated earlier, and this fact could interfere in this association.
On the whole, data from the Cancer Genome Atlas (TCGA) show that ∼20% of sporadic HGSOCs harbor some kind of BRCA1 or BRCA2 deficiencies. Nevertheless, abnormalities in other genes (such as EMSY or PTEN) that also operate in the HR pathway (mutation, rearrangement, DNA methylation, or attenuated mRNA expression) would be the cause of HR inactivation in another 20% of HGSOCs (4). The current increasing interest in this research could expand the therapeutic possibilities of PARP inhibitors beyond germline or somatic BRCA1/2 mutations. Our work is focused on RAP80: its proteins act on the RAP80/Abraxas1/BRCA1 (BRCA1A) macrocomplex related to the DNA damage response around the DSBs (6) and its mechanism of action has been clearly documented to affect HR in preclinical studies.
Importantly, after the BRCA1 accrual at the HR foci in a canonical H2AX-dependent manner, it is stabilized through its interaction with RAP80 in the BRCA1A complex. This complex allows the accumulation of BRCA1 at the damage-induced foci and also controls the G2-M checkpoint, temporarily inhibiting entry into mitosis to avoid aberrant chromosome segregation (25–27). Cellular experiments have shown that RAP80 deficiency affects the accumulation of BRCA1 (compromised) and CtIP and RAD51 (prolonged) at the HR foci, thus inducing excessive but inefficient hyperactivation of HR and subsequent genome instability (28, 29). The clinical relevance of RAP80 is endorsed by a report on genetic predisposition to hereditary breast cancer syndrome related to a particular RAP80 mutation that impairs the DNA damage response (30). In addition, in a phase II trial including wild-type epidermal growth factor receptor (EGFR) NSCLC patients treated with platinum or taxane compounds, RAP80 expression levels were identified as potential modifiers of the BRCA1 effect on prognosis (31), supporting preclinical evidence on the importance of RAP80 for BRCA1 repair function (27). The role of RAP80 expression in the prognosis of HGSOC has not been studied so far.
In summary, HR is frequently impaired in sporadic HGSOC, not only due to BRCA1 and BRCA2 deficiencies (∼16% and ∼4%, respectively) but also due to alterations in other HR–pathway-related genes (∼20%). These latter mechanisms are partially unexplored. It is well documented that RAP80 influences BRCA1 activity in the BRCA1A complex, and its alterations have been shown to affect normal HR. RAP80 has been reported to affect outcome of some solid neoplasms. We aim to elucidate the potential effect of RAP80 on outcome of chemotherapy in HGSOC. With this purpose, we analyzed mRNA expression levels of RAP80 (as well as BRCA1) isolated from paraffin-embedded tumor samples of surgical patients with sporadic HGSOC who received platinum-based chemotherapy as first line, and then correlated our findings with progression-free survival (PFS) and OS in these patients.
Materials and methods
Patient population and study design
We retrospectively retrieved formalin-fixed paraffin-embedded tissue blocks obtained at time of primary surgery (performed or attempted) from patients with sporadic HGSOC (BRCA1/2 wild-type patients or patients without clinical suspicion of hereditary breast and ovarian cancer syndrome and undetermined BRCA1/2 status) who received platinum-based chemotherapy as first line between 2000 and 2009 at the Hospital Germans Trias i Pujol, Badalona, Spain, to ensure a minimum follow-up of 5 years. Chemotherapy previous to tissue sample obtainment was an exclusion criterion. The tissue blocks were thus chemo-naïve. Clinical data on each patient were collected from medical files. The clinical variables selected for inclusion in our analyses were: age at diagnosis, International Federation of Gynecology and Obstetrics (FIGO) stage (early vs. advanced), residual disease at surgery, performance of systematic staging and cytoreductive debulking, date of relapse or progression, and survival information. Although the currently accepted standard is that absence of macroscopic residual disease should be the optimal goal of surgery (32), we used the former threshold of <1 cm vs. >1 cm since discrimination between 0 and 1-10 mm was not included in older medical files. Systematic staging and debulking surgery was defined as hysterectomy, bilateral oophorectomy and salpingectomy, omentectomy, exploration and biopsy of all peritoneum surfaces, resection of all suspicious implants, and exploration of pelvic and paraaortic nodes (with excision of at least the enlarged nodes) (33).
Because of the close biochemical interrelation between BRCA1 and the studied gene, the expression levels of BRCA1 were also analyzed given the moderate correlations with RAP80 that could be a confounding factor for the results.
The research was approved by the ethics committee of our institution (CEIC-Hospital Universitari Germans Trias i Pujol). Data can be retrieved upon request and approval by our ethics committee.
Tumor sample processing and gene expression analyses are detailed in the supplementary materials (see Supplementary Table S1 - Primers and probes. Available online at www.biological-markers.com).
Statistical analyses
Gene expression levels were considered continuous variables, but natural partition into tertiles was also planned for the main results. PFS was calculated from the date of surgery to the date of progression or death from any cause. OS was calculated from the date of surgery to the date of death from any cause. Median PFS and OS, with their 95% confidence intervals (CIs), were estimated using the Kaplan-Meier method. Normality was tested with the Shapiro-Wilk test. Spearman's correlation coefficient was used to measure the strength of association between 2 variables. The association between the expression levels of each gene and PFS and OS was assessed by means of Cox regression analyses adjusted for the clinical prognostic factors of FIGO stage, residual disease, and performance of systematic staging and debulking surgery (32, 34, 35) in order to avoid confounding effects and maximize the statistical power. All analyses were performed using the Statistical Package for the Social Sciences (SPSS) for Windows version 17. Significance was set at p<0.05.
Results
Patient characteristics and clinical outcome
Tumor samples were collected from 35 patients, whose demographic characteristics are shown in Table I. Chemotherapy consisted of intravenous (IV) carboplatin plus paclitaxel in 31 patients (88.57%) and other IV platinum-based combinations in 4 patients. The median number of cycles was 6. No maintenance drugs or consolidation schemes were prescribed.
Baseline patient characteristics (n = 35)
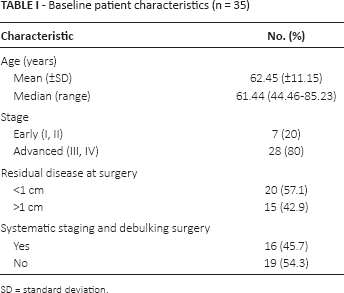
SD = standard deviation.
Median follow-up (from surgery until death or last date of follow-up) was 61.31 months (range: 24.15-74.81). At the time of data collection, 26 patients (84.6%) had progressed or relapsed, and 23 patients (65.7%) had died, 2 from non–cancer-related causes. Median PFS was 20.205 months (95% CI 10.076-30.335) and median OS was 62.784 months (95% CI 35.618-89.951). Most relapses were platinum sensitive (more than 6 months after the last platinum dose) (n = 22, 62.9%).
Univariate analysis for PFS and OS showed that age, advanced-stage disease, residual disease at surgery >1 cm, and absence of systematic staging and/or debulking surgery were significantly associated with worse outcomes, thus confirming the relevance of these variables as clinical prognostic factors (Tab. II).
Univariate analyses of clinical prognostic factors
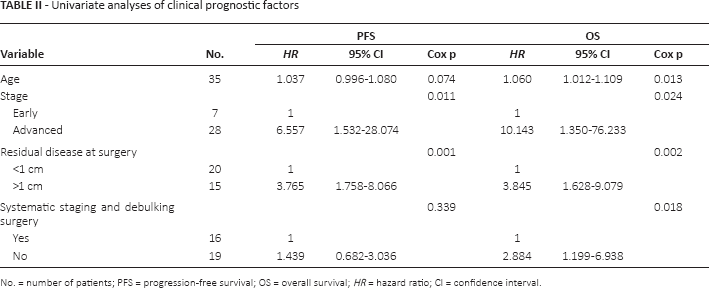
No. = number of patients; PFS = progression-free survival; OS = overall survival; HR = hazard ratio; CI = confidence interval.
Gene expression analyses
Gene expression analysis was successful in 71.4% of cases for BRCA1 and 91.4% of cases for RAP80. Median and mean expression levels are shown in Supplementary Table S2 (Results of gene expression analyses. Available online at www.biological-markers.com). Distribution of BRCA1 expression levels failed to show normality (Supplementary Table S3 - Shapiro-Wilk normality test. Available online at www.biological-markers.com). Moderate correlation was observed between BRCA1 and RAP80 expression levels (ρ = 0.56, p = 0.01) (Supplementary Table S4 - Spearman rho correlations between gene expression levels. Available online at www.biological-markers.com). Gene expression levels were not related to age, FIGO stage or residual disease at surgery (Supplementary Table S5 - Spearman rho correlations between gene expression levels and clinical characteristics. Available online at www.biological-markers.com).
Gene expression and PFS and OS
As mentioned in the Statistical Analyses section, the association between each gene expression level and outcome was assessed by means of Cox regression multivariate analyses adjusted for the 3 clinical prognostic factors that were found to be significant in the previous univariate analysis (FIGO stage, residual disease, and performance of systematic staging and debulking surgery). Only RAP80 proved to be significantly associated with outcome. Specifically, low RAP80 expression levels were associated with poorer PFS (HR = 1.449, 95% CI 1.107-1.901, p = 0.007) and OS (HR = 1.331, 95% CI 1.003-1.767, p = 0.047) (Tab. III). In order to further examine the association of RAP80 levels and PFS and OS, patients were classified according to the tertile of RAP80 expression (high, intermediate, low). Again, after adjusting for the 3 clinical prognostic factors, patients in the highest tertile of RAP80 expression had longer PFS and OS than those in the intermediate tertile (PFS: HR = 0.235, p = 0.020; OS: HR = 0.430, p = 0.195), who in turn had longer PFS and OS than those in the lowest tertile (PFS: HR = 0.465, p = 0.171; OS: HR = 0.491, p = 0.302), indicating a direct positive correlation between RAP80 expression levels and survival benefit (Fig. 1).
Cox regression of outcomes according to gene expression, adjusted for covariates of clinical prognostic factors (stage, residual disease at surgery, performance of systematic staging and debulking surgery)
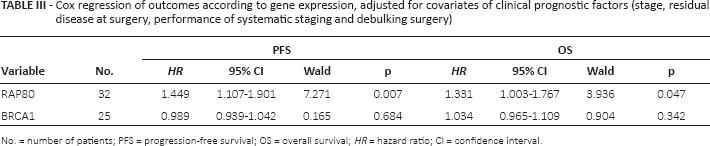
No. = number of patients; PFS = progression-free survival; OS = overall survival; HR = hazard ratio; CI = confidence interval.
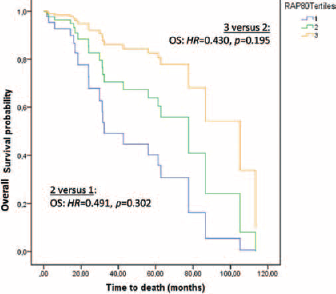
Overall survival plot of RAP80 expression in tertiles, adjusted for clinical prognostic factors as covariates (stage, residual disease at surgery, performance of systematic staging and debulking surgery). OS = overall survival; HR = hazard ratio.
In the exploratory subgroup analyses (FIGO stage [early vs. advanced], residual disease [<1 cm vs. >1 cm], and performance of systematic staging and debulking surgery [yes vs. no]), using Cox multivariate regression, the adjusted effects of RAP80 on PFS and OS were consistent across all subgroups, although statistical significance was only reached for the group with advanced disease (Supplementary Table S6 - Subgroup analyses. Available online at www.biological-markers.com).
Discussion
The present study is the first to examine the effect of RAP80 expression on outcome in ovarian cancer patients. Our findings indicate that decreased RAP80 expression levels have a deleterious effect on both PFS (HR = 1.4, p = 0.007) and OS (HR = 1.3, p = 0.047) in patients with resected sporadic HGSOC treated with platinum and taxane combinations after surgery.
Although the BRCA1 effect on outcome was not a main endpoint of our study, we believe it is mandatory to discuss this issue to better contextualize our findings. Our study did not find any association between BRCA1 expression and outcome. Indeed, the predictive impact of BRCA1 activity on sporadic HGSOC treated with platinum-based chemotherapy is still a matter of debate and could differ between tumors with somatic BRCA1/2 mutations and tumors with BRCA1 silencing. Interestingly, low/intermediate levels of BRCA1 expression predicted longer OS in a study of fresh-frozen tissue samples from 70 sporadic ovarian cancer patients treated with platinum-based chemotherapy (combined with a taxane in 34.6% of patients) (22). However, in a TCGA Research Network study of 316 HGSOC patients, 94% of whom had received a platinum-taxane combination, BRCA1 methylation conferred no survival advantage (HR = 1.06, 95% CI 0.62-1.81) (23). Expression of BRCA1 assessed by immunochemistry was studied in the 393 patients included in the phase III GOG-172 trial, which compared the intraperitoneal (IP) regimen (IV paclitaxel on day 1, IP cisplatin on day 2 and IP paclitaxel on day 8) versus the standard IV carboplatin + paclitaxel regimen as first line after surgery. Decreased expression (≤10% staining) of BRCA1 was associated with significantly longer survival in patients treated intraperitoneally, while the opposite trend was observed in patients in the IV arm (24). The authors attributed these findings to the superiority of IP platinum over the IV arm and its greater effect in patients with aberrant BRCA1 expression, while the effect of IV taxanes would be greater in patients with normal expression. However, immunochemistry is not a standardized method for assessing BRCA1 expression, due to the insufficient sensitivity of monoclonal antibodies against the N-terminus and the difficulty in interpreting results. The results of the present study support the hypothesis that the effects of IV platinums and taxanes may counteract with each other in different subpopulations of HGSOC defined by BRCA1 expression levels.
Limited information is available on RAP80-deficient tumors. As previously stated, RAP80 was found to be a potential modifier of BRCA1 survival impact in a phase II trial with wild-type EGFR NSCLC patients who were differently treated as per BRCA1 expression levels (31). Specifically, increasing levels of RAP80 correlated with decreasing survival in the subgroup with low BRCA1 expression (treated with cisplatin + gemcitabine), while increasing levels of RAP80 correlated with increasing survival in the subgroup with intermediate-high BRCA1 expression (treated with cisplatin + docetaxel and docetaxel alone, respectively) (31). Our finding of a positive correlation between RAP80 expression and survival is in line with this study in NSCLC, taking into account that ∼80% of sporadic HGSOCs retain functional levels of BRCA1/2 (4, 10) and that the potential favorable outcome of the low-BRCA1 subgroup after platinum could be overcome by concomitant paclitaxel.
Preclinical data show that RAP80-deficient mice are more prone to developing spontaneous or radiation-induced tumors, indicating that RAP80 acts as a tumor suppressor (36, 37). As mentioned earlier, RAP80 deficiency affects the accumulation of BRCA1 (compromised) and CtIP and RAD51 (prolonged) at the HR foci, thus inducing excessive but inefficient HR. It has been shown that either insufficient or excessive HR induces genomic instability, which paves the way to tumorigenesis (29). However, these 2 mechanisms differ: RAP80 deficiency does not necessarily derogate BRCA1 function, as BRCA1 is initially recruited at the DSB sites in an H2AX-dependent manner and may, at least temporarily, retain some of its functions, such as its effect on cell survival (6, 28, 29). Therefore, the phenotypes and magnitude of defects in RAP80-deficient cells are mild compared with those in BRCA1-deficient cells, probably due to some functional redundancy among BRCA1 complexes (6). Taking into account these facts and our results, we can thus speculate that RAP80-deficient sporadic HGSOC tumor cells may show increased genome instability but not necessarily compromised cell survival or hypersensitivity to platinum-based chemotherapy, with overall more aggressive behavior than tumors with low BRCA1 expression levels. This hypothesis is also supported by the finding that mice with concomitant absence of p53 and RAP80 presented particularly aggressive carcinogenesis (37), which may be a relevant fact taking into account that the loss of p53 expression is a main feature of HGSOC. Whether PARP inhibitors may have any role in RAP80-deficient HGSOC or not remains unexplored.
The present study has some limitations, including its retrospective and exploratory nature and the limited sample size (n = 35). However, it is a homogeneously treated sample and all biopsies were obtained before any chemotherapy was delivered. In addition, our results indicate a significant positive impact of high RAP80 expression levels on both PFS and OS, consistent across the subgroup analysis. In addition, taking into account the follow-up period of our study (2000-2014), the definition “sporadic” was applied to BRCA1/2 wild-type patients or patients without clinical suspicion of hereditary breast and ovarian cancer syndrome and undetermined BRCA1/2 status. BRCA1/2 mutational status assessment was not routinely performed at that time in most Spanish centers. Thus, we cannot rule out that a few cases would have been classified as germline BRCA1/2 mutations using the current systematic genetic analyses. However, this supposition is unlikely to alter our results.
To the best of our knowledge, this is the first study to highlight a prognostic role for RAP80 in sporadic HGSOC. The influence of RAP80 expression on outcome observed in sporadic HGSOC may well vary in other cancers. Future research in a larger sample of wild-type BRCA HGSOC is warranted to clearly determine the prognostic role of RAP80 in this disease. We expect that our findings will provide the basis for further investigation of RAP80 and its role in HGSOC, and that they will help to elucidate the role of HR alterations in sporadic ovarian cancer.
Footnotes
Acknowledgment
The authors thank R. O'Brate and K. Williams, independent medical writers, for their editorial assistance.
Financial support: The work in Dr Rosell's laboratory is partially supported by grants from Fundació La Caixa and the Redes Temáticas de Investgación en Cáncer (RTICC; grant RD12/0036/0072). Neither of these funding organizations had any input in the design of the study, the collection of data, or the decision to publish.
Conflict of interest: The authors declare that there are no conflicts of interests.
