Abstract
Background:
Genetic studies of ovarian cancer (OC) have historically focused on BRCA1/2 mutations, lacking other studies of homologous recombination repair (HRR). Poly (ADP-ribose) polymerase inhibitors (PARPi) exploit synthetic lethality to significantly improve OC treatment outcomes, especially in BRCA1/2 deficiency patients.
Objectives:
Our study aims to construct a mutation map of HRR genes in OC and identify factors influencing the efficacy of PARPi.
Design:
A retrospective observational analysis of HRR gene variation data from 695 OC patients from March 2019 to February 2022 was performed.
Methods:
The HRR gene variation data of 695 OC patients who underwent next-generation sequencing (NGS) in the First Affiliated Hospital of Zhengzhou University were retrospectively collected. Clinical data on the use of PARPi in these patients were also gathered to identify factors that may interfere with the efficacy of PARPi.
Results:
Out of 127 pathogenic variants in the BRCA1/2 genes, 104 (81.9%) were BRCA1 mutations, and 23 (18.1%) were BRCA2 mutations. Among the 59 variants of uncertain significance (VUS), 20 (33.9%) were BRCA1, while 39 (66.1%) were BRCA2 mutations. In addition to BRCA1/2, HRR gene results showed that 9 (69%) of 13 were HRR pathway pathogenic variants; and 16 (1.7%) of 116 VUS were Food and Drug Administration (FDA)-approved mutated HRR genes. Notably, the treatment regimen significantly influenced the effectiveness of PARPi, especially when using first-line maintenance therapy, leading to enhanced progression-free survival (PFS) compared to alternative protocols.
Conclusion:
Focusing on HRR gene mutations and supporting clinical research about PARPi in OC patients is crucial for developing precision treatment strategies and enhancing prognosis.
Introduction
Ovarian cancer (OC) ranks as the sixth most common cause of death among female malignant tumors, presenting a substantial risk to women’s lives and well-being. 1 GLOBOCAN 2020 data reveals that there were 313,959 new cases of OC reported globally, resulting in 207,252 annual deaths. 2 Currently, the occurrence of OC in China is on the rise. 3 The primary barrier to enhancing OC diagnosis is the absence of efficient early detection screening techniques. 4 Therefore, the disease is already at an advanced stage when it is diagnosed in many women with OC, and the 5-year survival rate is only 15%–25%. 5 75% patients with advanced illness have a relapse within 18–24 months, and the median progression-free survival (PFS) for OC is 16–21 months.6–8 In addition, chemotherapy resistance can easily lead to OC recurrence and a high mortality rate.9,10
Different pathological types of OC tend to have different types of genetic alterations. Genetic susceptibility accounts for 14%–24% of OC cases, primarily due to hereditary mutations in the BRCA1 or BRCA2 genes. 11 BRCA1 and BRCA2 proteins participate in the repair of double-strand DNA breaks through homologous recombination repair (HRR). 12 Dysfunction of BRCA1, BRCA2, or genes coding for proteins that interact with BRCA proteins, like BRIP1, RAD51C, RAD51D, and FANCM, results in genomic instability. 13 High-grade serous ovarian cancer (HGSOC) exhibits high recurrence and metastasis rates, 14 with over 95% of cases carrying somatic TP53 mutations. 15 Conversely, TP53 mutations are notably less prevalent in low-grade serous ovarian cancer (LGSOC) and serous borderline tumors. 16 Mutations in BRAF and KRAS were found in about 60% of LGSOC, but not in HGSOC. 17 Epithelial ovarian cancer (EOC) such as clear cell, endometrioid, and mucinous tumors, exhibit mutations in oncogenes KRAS and PI3K, as well as tumor suppressor genes PTEN and ARID1A. 18 Comparatively, major mutations in mucinous ovarian tumors are primarily KRAS mutations.19,20
Genetic mutations can easily cause the corresponding treatment to fail in OC. 21 Exploring innovative treatment approaches in recent years has led to the emergence of novel drugs, such as Poly (ADP-ribose) polymerase inhibitors (PARPi), which specifically target the repair of DNA damage in anticancer mechanisms. 22 PARPi, such as olaparib, niraparib, and rucaparib, target the synthetic lethality pathway, inhibiting the repair of single-strand breaks and causing cancer cell death through apoptosis. 23 Olaparib has been approved for the treatment of patients with advanced OC as the first-line maintenance therapy (FL-M), providing a significant PFS benefit (as demonstrated in the phase III SOLO1 clinical trial, NCT01844986).24–26 Niraparib has also been approved for the maintenance treatment of adult patients with newly diagnosed advanced (FIGO Stages III and IV) epithelial high-grade ovarian, fallopian tube, or primary peritoneal cancer who are in complete response (CR) or partial response (PR) following completion of first-line platinum-based chemotherapy.27,28 In recurrent OC, the European Society of Medical Oncology (ESMO) guidelines recommend niraparib for maintenance therapy regardless of BRCA mutation status, while for patients with BRCA mutations, olaparib has been recommended for maintenance therapy. 29 By contrast, American Society of Clinical Oncology (ASCO) guidelines suggest that maintenance treatment with niraparib in patients without germline or somatic BRCA mutations should carefully evaluate the potential PFS benefit against a possible overall survival decrement. 28 Therefore, the Food and Drug Administration (FDA), but not the European Medicines Agency, restricted niraparib indication to only patients with germline BRCA mutations.30,31 In addition, PARPi monotherapy should not be routinely offered for the treatment of recurrent platinum-sensitive EOC. 28 Overall, PARPi hold great promise for OC maintenance treatment, but in clinical practice, individualized treatment protocols should be developed based on the patient’s specific genetic mutation status, disease progression, and treatment response to maximize efficacy and minimize adverse effects. Future studies should continue to delve deeper into the optimal application strategy of PARPi to improve the prognosis of patients with OC.
In this study, the molecular characteristics of HRR genes in 695 OC patients who underwent next-generation sequencing (NGS) in the First Affiliated Hospital of Zhengzhou University were analyzed to identify different HRR gene mutations. In addition, the clinical diagnosis and treatment data of 180 patients who were treated with PARPi after HRR gene sequencing were collected. This study aims to provide a reference for clinical guidance of PARPi medication in OC by constructing an HRR gene map and exploring the factors that may affect the efficacy of PARPi in OC.
Materials and methods
Patient recruitment
A total of 695 patients who were newly diagnosed with malignant OC and underwent the NGS of HRR in the Department of Obstetrics and Gynecology of the First Affiliated Hospital of Zhengzhou University from March 2019 to February 2022 were collected to analyze the molecular distribution characteristics of HRR gene mutations including BRCA1/2. 32 Post-test genetic counseling to patients with positive germline mutation testing was provided to analyze the risks for their immediate relatives. The research was approved by the Ethics Committee of the First Affiliated Hospital, Zhengzhou University (2023-KY-1165-002). The inclusion criteria for this study were as follows: (i) at least 18 years old; (ii) patients with pathological diagnosis of OC and fallopian tube cancer; and (iii) PARPi treatment for at least 1 month. Due to its retrospective design and the use of anonymized data, the requirement for informed consent from the patients was waived. The reporting of this research conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental File 1). 33
Clinical assessments
Data were obtained from the patient’s medical records. Clinical data from patients participating in NGS, especially those using PARPi (including olaparib, niraparib, etc.), were collected. Baseline clinical characteristics of all patients were gathered. Furthermore, data on the indication for PARPi treatment were gathered, including maintenance treatment for patients with advanced ovarian, fallopian tube, or primary peritoneal cancer who achieved CR or PR to FL-M; maintenance treatment for patients with recurrent ovarian, fallopian tube, or primary peritoneal cancer who achieved CR or PR to platinum-based chemotherapy (maintenance therapy after platinum-sensitive recurrence (PSR-M)); and active treatment (AT) for patients with recurrent advanced OC. Relapses that happened over 6 months following the most recent platinum-based chemotherapy treatment were categorized as platinum-sensitive recurrences. BRCA mutation-positive (BRCAmut) was described as a pathogenic or possibly pathogenic mutation in either BRCA1 or BRCA2 that was found in a patient; BRCA wide type (BRCAwt) was the alternative option. Pathogenic and likely pathogenic mutations in various genes associated with the HRR pathway, such as ATM, BARD1, BRCA1, BRCA2, BRIP1, CDK12, CHEK1, CHEK2, FANCL, PALB2, RAD51B, RAD51C, RAD51D, and RAD54L were considered as HRR mutation positive,34,35 which were also FDA-approved mutated HRR genes. 36 The Common Terminology Criteria Adverse Events V 5.0 was used to grade adverse events (AEs) to adjust the dosage. PFS was determined by measuring the duration from the initiation of PARPi treatment to the occurrence of either disease progression or cause-specific death, whichever happened earlier.
Statistical analysis
The statistical analyses were conducted using R software (Lucent Technologies, version 4.3.1) and SPSS software (International Business Machines Corporation, version 22.0). The categorical variables, namely BRCA mutation status and HRR mutation status, were compared using the Fisher exact test. To analyze PFS, Kaplan-Meier curves were compared using a log-rank test, and the relationships between various factors and PFS were tested by determining the hazard ratio using a Cox proportional hazards regression model. Multivariable models were constructed using baseline variables that met the p < 0.05 threshold of significance in the univariable analysis. Statistical significance was set at p < 0.05.
Results
Cohort characteristics
The clinical and demographic characteristics of patients recruited in this study are outlined in Table 1. The median age at which OC was diagnosed was 52 years, with a range from 55 to 60. Histology of OC showed that most cases were HGSOC (590/695), followed by LGSOC (45/695), clear cell (40/695), endometrioid (4/695), mucinous (7/695), and others (9/695).
Clinical and demographic characteristics of our cohort.

Distribution of mutation in HRR gene
We first collected and analyzed the percentage of the BRCA1/2 gene, as shown in Figure 1a. Of the 127 pathogenic variants, 104 (104/127, 81.9%) occurred in BRCA1, and 23 (23/127, 18.1%) occurred in BRCA2. In addition, among the 59 cases of variants of uncertain significance (VUS) identified, 20 cases (20/59, 33.9%) were associated with VUS in the BRCA1 gene, and 39 cases (39/50, 66.1%) were associated with VUS in the BRCA2 gene. Other subjects were identified as having deleterious variants in non-BRCA genes (Figure 1b). Of the 13 pathogenic variants in all panels except BRCA1/2, 9 (9/13, 69%) involved pathogenic mutations in HRR genes. Among them, TP53, SDHA, FANCM, FANCA, BRIP1, PALB2, and RAD51D are mutant genes. There were 116 VUS and 16 (16/116, 1.7%) genes involved in HRR genes. Among them, ATM, BARD1, CDK12, CHEK2, FANCL, PALB2, RAD51B, RAD51C, RAD51D, and RAD54L are mutant genes approved by FDA. 36 In summary, the BRCA1/2 mutations continue to be the predominant mutations in OC within the realm of HRR genes.
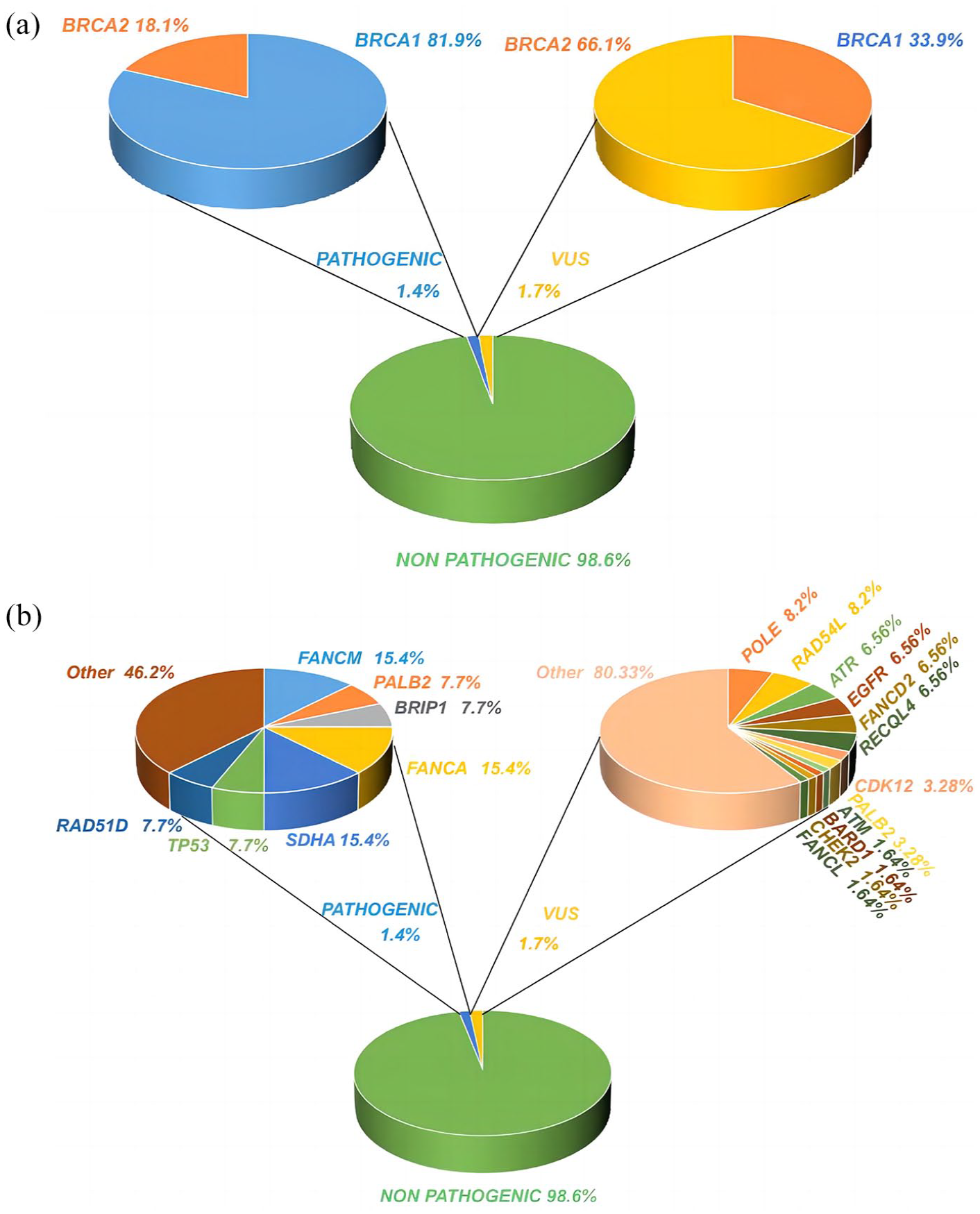
Percentage of pathogenic variations and VUS in the HRR gene test. (a) Percentage of pathogenic variants and VUS in testing BRCA1/2 gene. (b) Percentage of pathogenic variants and VUS testing for HRR genes other than the BRCA1/2 gene.
Characterization of DNA mutations and copy number variation
After analyzing the HRR mutation data, the top 30 genes with DNA mutations and the top 20 genes with copy number variation (CNV) are prominently displayed. Further detailed DNA mutation and CNV characteristics are present in Figure 2. The different colors in the figure represent various mutation types, including exonic, frameshift deletion, nonsynonymous single nucleotide variations (SNV), frameshift mutation, frameshift substitution, missense mutation, non-frameshift deletion insertion, non-frameshift substitution, nonsense mutation, splicing, stopgain, and mut-het. The top two mutated genes are BRCA1/2 with the primary types of DNA mutations being exonic variants and het mutations. The primary DNA mutation type for TP53 is nonsynonymous SNV. Furthermore, this study assessed the top 20 variations in CNV, including MYC, PRKCI, TERC, etc., primarily focusing on nonsynonymous SNV in DNA. DNA mutation and CNV are two distinct types of genomic alterations, each reflecting distinct levels of genomic variation. DNA mutation refers to alterations in single or multiple bases within the genome, whereas CNV involves changes in the copy number of one or more genes. 37 Both types of genomic variations play essential roles in understanding the mechanisms underlying OC and individual genetic diversity, contributing significantly to the advancement of personalized therapies for OC.

Characterization of DNA mutations and CNV in 695 samples. The figure depicts the top 30 genes with DNA mutations and the top 20 genes with CNV.
Analysis of the family history of BRCA germline
In addition to cross-sectional analysis of OC-related HRR gene mutation results, this study also conducted a longitudinal survey to analyze the BRCA gene mutation status of OC patients and their immediate family members. Based on the comparative analysis of genetic mutations of patients with BRCA mutations and their immediate relatives (Table 2), identical amino acid and coding sequence changes were observed among immediate family members. The occurrence of OC in two generations strongly supports a common origin, confirming the hereditary nature and familial clustering of OC. 38
Family history of BRCA germline.

CDS, coding sequence.
Characteristics of patients with PARPi treatment among the 180 individuals
Although we collected the data of NGS in 695 OC patients, focusing on variants in the HRR gene, only 180 patients received PARPi (olaparib and niraparib) treatment. We categorized and followed up on the clinical information of these 180 patients according to their original treatment plans. Among them, 100 patients (100/180, 55.6%) were in the FL-M group, 40 patients (40/180, 22.2%) were in the PSR-M group, and an additional 40 patients (40/180, 22.2%) were in the AT group (Table 3). Among the 100 patients in the FL-M group, 47 patients had a BRCAmut (34 patients had a BRCA1 mutation, while 16 patients carried a BRCA2 mutation), while 53 patients had BRCAwt. Besides the FL-M group, 13 others had mutations in HRR pathway genes other than BRCA mutations. Among the 40 patients in the PSR-M group, 14 patients (14/40, 35%) had a BRCA1 mutation, 2 patients (2/40, 5%) had a BRCA2 mutation, and 24 patients (24/40, 60%) had BRCAwt. In addition, four individuals in the PSR-M group experienced mutations in the HRR pathway genes other than BRCA1/2 genes. Of the 40 patients in the AT group, 11 had a BRCA1 mutation, and 5 had mutations in the HRR pathway other than BRCA mutations. Among them, 11 were treated with immunosuppressive drugs.
Characteristics of 180 patients in this study underwent PARPi treatment.

AT, active treatment; BMI, body mass index; CR, complete response; FL-M, first-line maintenance therapy; HRR, homologous recombination repair; NA, not available; PARPi, poly (ADP-ribose) polymerase inhibitors; PR, partial response; PSR-M, maintenance therapy after platinum-sensitive recurrence.
Treatment response and PFS for the included patients after PARPi treatment
We conducted follow-ups and monitored the efficacy of PARPi specifically in the 180 individuals who received PARPi treatment in this cohort study with NGS. Of the patients treated with PARPi, 74 had BRCAmut, and 106 had BRCAwt. The median PFS of BRCAwt was 23 months, while the median PFS of the BRCAmut was not reached. Besides, the data have shown that the 12-month PFS of the BRCAwt group was 58.8% (95% CI: 50.0–69.1), while the 12-month PFS of BRCA1/2mut was 70.1% (95% CI: 60.4–81.4) (Figure 3a). Results found that BRCAmut types had longer PFS and better prognosis compared to the BRCAwt group (p < 0.05). In addition, different treatment regimens of PARPi exhibited varying efficacies. Progression events were observed in 82 (82/180, 45.6%) patients, including 23 (23/100, 23.0%) in the FL-M group, 24 (24/40, 60.0%) in the PSR-M group, and 35 (35/40, 70.0%) in the AT group. Four patients died during follow-up due to disease progression in the FL-M group, and the 12-month PFS was 83.9% (95% CI: 76.9–91.4). The 12-month PFS for PSR-M patients was 54.9% (95% CI: 41.4–72.7), with 10 patient deaths. Among the AT patients, the 12-month PFS was 19.34% (95% CI: 10.01–37.4), and 21 patients died of disease progression during follow-up (Figure 3b). The findings indicate that the FL-M regimen of PARPi is linked to a more favorable prognosis, demonstrating superior PFS compared to the PSR-M and AT groups in OC.
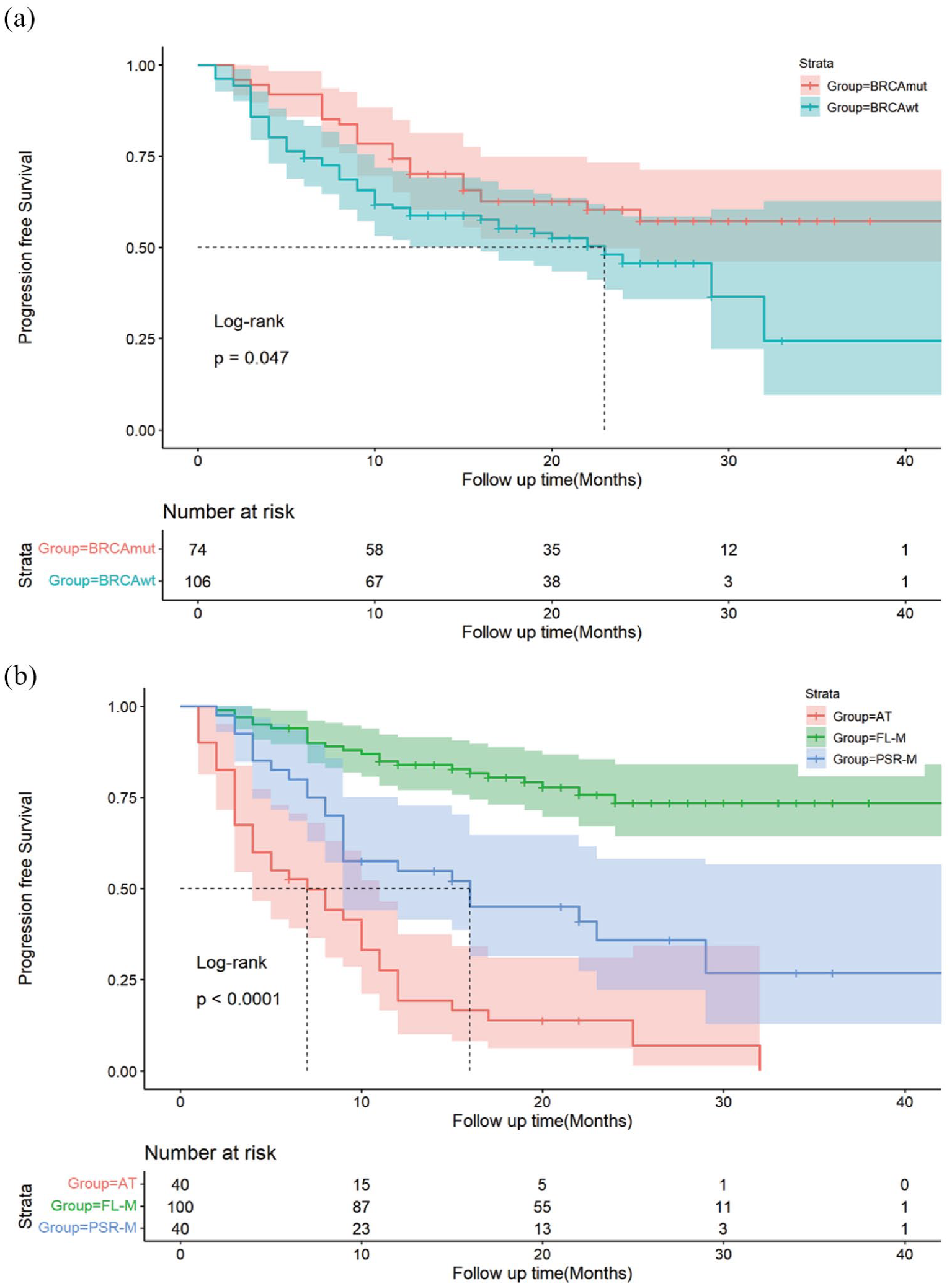
Kaplan-Meier curves for the PFS in PARPi patients. (a) Kaplan-Meier curves for the PFS in the BRCA1/2wt and BRCA1/2mut patients. (b) Kaplan-Meier curves for PFS of patients in the FL-M, PSR-M, and AT subgroups.
Influencing factors for PFS
In this study, the continuous variables, age and body mass index, were divided into two categorical variables according to the overall average value. The multivariate study suggests that the anti-angiogenic medication, the response to platinum-based chemotherapy (CR/PR), and whether or not a second surgery was performed could be predictors of the PFS in the FL-M group. It was discovered that the PFS of the patients in the PSR-M group was influenced by the tumor stage and the amount of time that had passed between the end of chemotherapy and the start of PARPi (months). The PFS in the AT group was significantly impacted by the use of anti-angiogenic medications, except bevacizumab (Table 4).
Factors linked to PFS in the three groups were analyzed using Cox regression.

AT, active treatment; BMI, body mass index; BRCAmut, BRCA mutation-positive; BRCAwt, BRCA wide type; CR, complete response; FL-M, first-line maintenance therapy; HR, hazard ratio; PARPi, poly (ADP-ribose) polymerase inhibitors; PFS, progression-free survival; PR, partial response; PSR-M, maintenance therapy after platinum-sensitive recurrence.
AEs associated with PARPi treatment
The AEs associated with PARPi treatment are critical concerns in the management of OC,26,39 and the detailed information on these events for the included patients is presented in Table 5. Among the 180 patients treated with PARPi, 34 experienced AEs. Gastrointestinal symptoms were the most common AE, such as nausea and vomiting. Besides, there were eight patients with hematological AEs, of which three were severely discontinued due to myelosuppression, experiencing symptoms such as anemia, reduced platelet count, and leukopenia, while two were improved after drug reduction. Instead of hematological AEs, two individuals discontinued the medication due to severe vomiting. In addition, mild AEs such as diarrhea, fatigue, constipation, anorexia, oral mucositis, and erythema were observed.
AEs for 180 patients treated with PARPi treatment.

AEs, adverse events; PARPi, poly (ADP-ribose) polymerase inhibitors.
Discussion
The elevated mortality and morbidity rates of OC underscore its significance as a critical area of study. 1 Notably, germline pathogenic variants, particularly in genes related to homologous recombination and mismatch repair pathways, are detected in about 22%–25% of EOC cases. 40 Extensive research has been directed toward the BRCA1/2 genes, focusing on their mutations and the therapeutic efficacy of PARPi. 41 Nevertheless, notable deficiencies in the study of other HRR genes also exist. Concurrently, advancements in NGS have enhanced gene detection capabilities, including those of BRCA1/2, thereby underpinning clinical decision-making and enabling more precise treatments, which may provide a novel theoretical basis for improving OC therapy.
This study gathered data on BRCA1/2 and other HRR gene mutations from 695 OC patients who were treated at the First Affiliated Hospital of Zhengzhou University between February 2019 and February 2022. The study aimed to assess the connection between gene mutation status and the effectiveness of PARPi treatment. Our findings suggest that BRCA1/2 mutations are still the main mutation form of OC, and the proportion of pathogenic variants is much higher than that of VUS. The pathogenic variants occur in BRCA1/2 (81.9% and 18.1% of total mutations for BRCA1 and BRCA2, respectively). In addition to BRCA1/2 gene mutations, pathogenic mutations were in TP53, SDHA, FANCM, FANCA, BRIP1, PALB2, RAD51D, etc., and VUS were in ATM, BARD1, CDK12, CHEK2, FANCL, PALB2, RAD51, RAD51C, RAD51D, RAD54L, etc. Simultaneously, an analysis of familial histories about BRCA germline mutations revealed the presence of identical genetic variations within direct family lineages, suggesting a shared genetic origin for OC across two generations.
In this research, the genetic profile of HRR in a large OC cohort was studied. HRR genes such as BRCA1/2 were detected in OC patients and a large number of gene mutations were found. Simultaneously, the relationship between patients’ genetic mutation profiles and the efficacy of PARPi was analyzed. It was found that compared to the BRCAwt group, PARPi significantly improves the PFS in patients with BRCA1/2mut. Simultaneously, the stratification of OC treatment protocols indicated that FL-M therapy was associated with enhanced PFS, demonstrating a notable advantage over PSR-M and AT groups. Factors potentially influencing PFS included sensitivity to cisplatin, the interval between the final chemotherapy session and initiation of PARPi treatment, and the administration of anti-angiogenic drugs, such as bevacizumab. AEs were included in this study, with hematological adverse reactions, including anemia, thrombocytopenia, and leukopenia, identified as the most common type. These AEs might require intervention through drug dose reduction or drug withdrawal to alleviate their impact in severe cases.
This study has limitations inherent in its observational nature. This study is a single-center study, limited to the population admitted to the First Affiliated Hospital of Zhengzhou University, and the population may not be representative of other regions. In addition, a larger number of the clinical samples is required for in-depth studies. Meanwhile, the type of PARPi used by the patients was not strictly differentiated in this study, and all patients received at least one approved PARPi treatment. Future studies should consider distinguishing between different types of PARPi to assess their specific impact on treatment efficacy and AEs. Overall, the relevant results of this study, including the influence of BRCA1/2 gene status, PARPi treatment plan, and other related factors on the prognosis of OC, as well as the occurrence of AEs, still provide references for the clinical treatment of OC.
Conclusion
In OC patients, prioritizing multi-gene HRR pathway mutations over BRCA1/2 testing can offer clinically significant insights. Integrating clinical data helps identify factors affecting PARPi effectiveness, hence directing clinical PARPi use in OC.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241271845 – Supplemental material for Genetic landscape of homologous recombination repair and practical outcomes of PARPi therapy in ovarian cancer management
Supplemental material, sj-docx-1-tam-10.1177_17588359241271845 for Genetic landscape of homologous recombination repair and practical outcomes of PARPi therapy in ovarian cancer management by Mengyu Chen, Ningjing Lei, Ruixia Guo, Liping Han, Qinghe Zhao, Yang Zhao, Luojie Qiu, Fengling Wu, Shan Jiang, Ningyao Tong, Kunmei Wang, Siyu Li and Lei Chang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
