Abstract
Background
Lung cancer ranks first both in morbidity and mortality in malignancies, but prognostic biological markers are lacking. The neutrophil-lymphocyte ratio (NLR) was proposed as a convenient biological marker. This study aimed to explore the prognostic value of NLR in advanced non-small cell lung cancer (NSCLC).
Methods
This retrospective study screened patients admitted from October 2007 to October 2014. Patients had histopathologically confirmed, treatment-naïve, metastatic NSCLC, and were prescribed platinum doublet chemotherapy. NLR and demographic data were collected, together with the outcome of chemotherapy. Progression-free survival (PFS) and overall survival (OS) were analyzed using the Kaplan-Meier method and Cox regression model.
Results
A total of 325 patients were enrolled. The cutoff value for NLR (3.19) was determined by receiver operator characteristic analysis. Patients were dichotomized into high (≥3.19) and low (<3.19) NLR groups. Both groups had similar demographic features. However, the low-NLR group had longer PFS (6.1 months) and OS (22.3 months) than the high-NLR group (5.1 months, p = 0.002; 13.1 months, p<0.001, respectively). Multivariate analysis confirmed that NLR was inversely related to the prognosis of these patients (HR = 1.684, 95%: 1.297-2.185, p<0.001).
Conclusions
This study argues that NLR is a convenient prognostic biological marker for advanced NSCLC patients treated with first-line chemotherapy and warrants further validation.
Introduction
Lung cancer ranks first both in morbidity and mortality in malignancies (1). Eighty percent of lung cancer cases are non-small cell lung cancer (NSCLC) (2). More than half of the patients are diagnosed at an advanced stage of the disease, with median overall survival (OS) of merely 10-12 months when standard platinum-based chemotherapy is given (3). Whereas targeted therapy achieves better efficacy, drug resistance inevitably follows. Therefore, there is an urgent but unmet need to identify prognostic factors for these patients to improve their prognosis.
As one of the hallmarks of cancer, inflammation facilitates the progression of cancer in various ways, including the promotion of angiogenesis, exacerbation of DNA damage, and facilitation of invasion (4). Inflammation-related factors including the neutrophil-lymphocyte ratio (NLR) have been proposed as prognostic markers for cancer (5, 6). Although the prognostic significance of NLR has been reported in rectal cancer, gastric cancer and esophageal cancer (7-8-9), it remains largely elusive in advanced NSCLC. Up to now, merely preliminary studies have been reported (10, 11). For the bulk of patients with advanced NSCLC, platinum doublet chemotherapy remains the standard first-line treatment. The outcome of this cohort of patients is critical to improve the prognosis. The present study aimed to investigate the prognostic value of baseline (pre-chemotherapy) NLR in these patients.
Materials and methods
Patients
This was a retrospective study conducted in patients admitted to the West China Hospital (a tertiary referral center) from October 2007 to October 2014. The inclusion criteria were 1) histopathologically confirmed NSCLC; 2) metastatic disease; 3) patients had to be treatment naïve and were prescribed platinum doublet chemotherapy (with paclitaxel, gemcitabine or pemetrexed). The exclusion criteria were 1) comorbidities affecting the hematological index such as serious infection, hematological disease or autoimmune disease; 2) concomitant other cancer; 3) maintenance therapy, either chemotherapy or targeted therapy; 4) patients who had their target lesions resected were excluded due to difficulties in defining progression. The study was approved by the ethics committee of Sichuan University.
Tumor staging was based on the American Joint Committee on Cancer Staging Manual (seventh edition; or sixth edition for patients admitted before 2009). Staging was amended to the new edition where the old one was used, e.g., those with “wet” IIIB were restaged to metastatic disease. Demographic and clinicopathological data were collected. The data censored time was July 2015.
Determination of NLR
The complete blood count results were obtained within 7 days prior to the initiation of chemotherapy. White cell count, neutrophil count, lymphocyte count and hemoglobin were recorded. NLR was calculated as follows: neutrophil count/lymphocyte count (11).
Statistical Analysis
OS was defined as the interval between the start of chemotherapy and the date of death. Progression-free survival (PFS) was defined as the time between the start of chemotherapy and the first sign of disease progression. Follow-up was defined from the initiation of chemotherapy to the censored date or the date of death, whichever came first. The “reverse” Kaplan-Meier curve was used to estimate the median follow-up time. Receiver operating characteristic (ROC) analysis and relative area under the curve (AUC) statistics were used to optimize the cutoff value of NLR. To perform ROC analysis, the NLR value was plotted against the survival status for each patient. Those patients who were lost to follow-up were excluded. Because this was a retrospective exploratory study, no primary end point was prespecified. The best cutoff value was selected with the maximum Youden's index (sensitivity + specificity-1). Student's t-test and the chi-square test were used for continuous and categorical variables separately. Survival analysis was done with the Kaplan-Meier method. Multivariate survival analysis was performed by means of the Cox regression model by considering variables with p<0.1 in univariate analysis. Proportional hazard assumption was tested graphically (Kaplan-Meier curve) to determine whether variables in the Cox proportional hazards model were constants that do not depend on time. All statistical analyses were conducted with SPSS 19.0 (IBM Inc.). P values less than 0.05 were considered statistically significant.
Results
Patient Characteristics
A total of 325 patients were enrolled in this study based on the inclusion and exclusion criteria (Fig. 1). The baseline demographic characteristics are shown in Table I. The median age was 59 years (range 28-82), and 218 (67.1%) of the patients were male. The most prominent histological type was adenocarcinoma (56.9%), followed by squamous carcinoma (25.8%). Other pathological types included adenosquamous carcinoma and poorly differentiated NSCLC (17.3%).
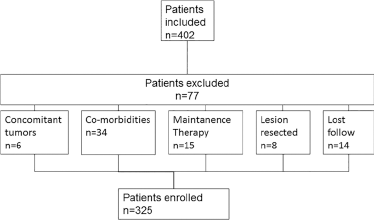
Flowchart of patient enrolment.
Patient characteristics
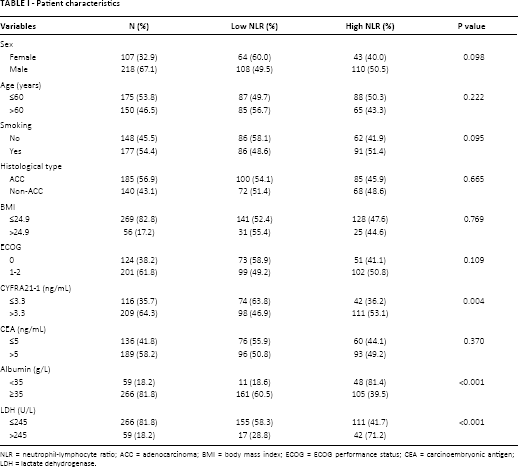
NLR = neutrophil-lymphocyte ratio; ACC = adenocarcinoma; BMI = body mass index; ECOG = ECOG performance status; CEA = carcinoembryonic antigen; LDH = lactate dehydrogenase.
Hematological Profiles
For the whole cohort, the median neutrophil count was 4.33 × 109/L (range 1.09-18.9), and the median lymphocyte count was 1.38 × 109/L (range 0.21-7.28). A wide range of NLR distribution was observed (range 1.0-21.81), and the median NLR was 3.09 (Fig. 2A-B-C). No relationship could be found between NLR and demographic features including age, sex, smoking, histological type, and Eastern Cooperative Oncology Group (ECOG) performance status (data not shown).
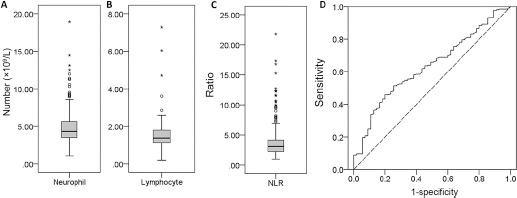
Distribution of neutrophils (
Patient Grouping
According to the ROC analysis, the recommended cutoff value for NLR was 3.19 with an AUC of 0.63 (sensitivity 51.5%, specificity 74.5%, Fig. 2D). The patients were dichotomized into a high-NLR (≥3.19) and low-NLR (<3.19) group. Both groups had similar demographic features. NLR was positively correlated with the NSCLC-associated antigen CYFRA21-1 and with lactate dehydrogenase (LDH), and negatively correlated with albumin (Tab. I).
NLR and Prognosis
For the whole cohort, the median duration of follow-up was 16.7 months (range 0.3-75.4 months). At the time of preparation of this manuscript (Aug 2015), 270 patients had died and the 5-year survival was 4.6% (Fig. 3A).
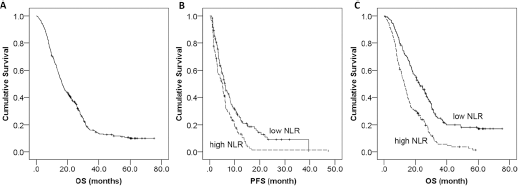
Overall survival (OS) of the whole cohort (
The median PFS was 6.1 months (95% CI: 4.9-7.2 months) and 5.1 months (95% CI: 4.1-6.1 months) for the low-NLR and high-NLR groups, respectively (p = 0.002, Fig. 3B). In addition, patients in the low-NLR group lived longer (22.3 months, 95% CI: 18.2-26.4 months) than those in the high-NLR group (13.1 months, 95% CI: 14.7-18.7 months, p<0.001, Fig. 3C).
In the univariate analysis, NLR, ECOG performance status, CYFRA21-1 and albumin were all found to be significant factors for both PFS and OS. LDH was related to OS but not PFS (Tab. II). In the multivariate analysis, ECOG performance status and CYFRA21-1 remained significant for both PFS and OS. NLR was significantly associated with OS but not PFS (Tab. III).
Univariate analysis in relation to OS and PFS
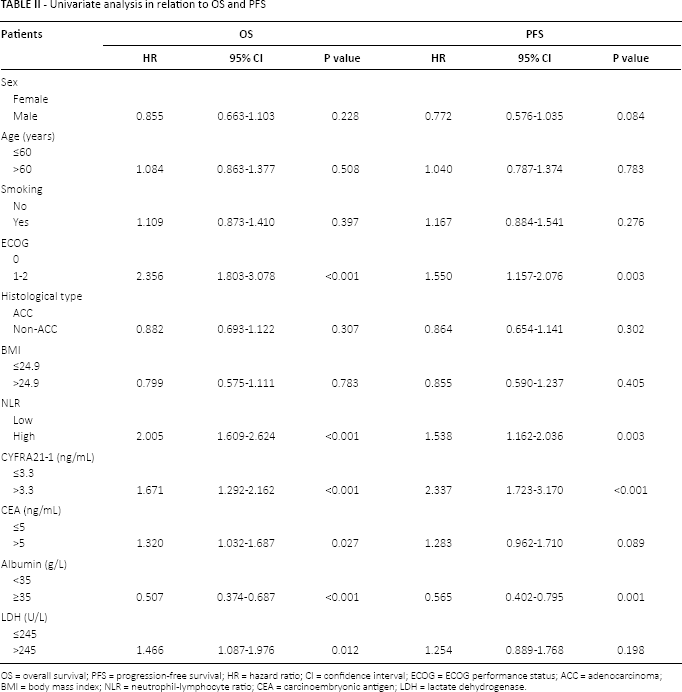
OS = overall survival; PFS = progression-free survival; HR = hazard ratio; CI = confidence interval; ECOG = ECOG performance status; ACC = adenocarcinoma
BMI = body mass index; NLR = neutrophil-lymphocyte ratio; CEA = carcinoembryonic antigen; LDH = lactate dehydrogenase.
Multivariate analysis in relation to OS and PFS
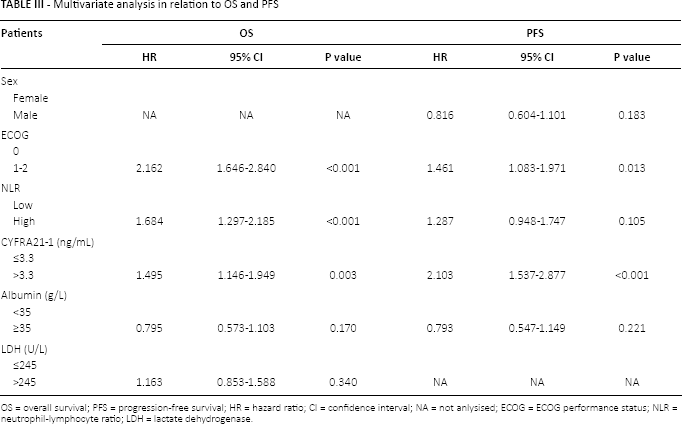
OS = overall survival; PFS = progression-free survival; HR = hazard ratio; CI = confidence interval; NA = not anlysised; ECOG = ECOG performance status; NLR = neutrophil-lymphocyte ratio; LDH = lactate dehydrogenase.
Discussion
We conducted a retrospective study to explore the relationship between NLR and the prognosis of patients with advanced NSCLC, enrolling a cohort of 325 patients treated with first-line platinum doublet chemotherapy between 2007 and 2014. NLR showed no correlations with any of the demographic features but was found to have prognostic value. Multivariate analysis confirmed that NLR was an independent prognostic marker.
The value of NLR as a prognostic marker had been previously explored in various tumors including gastric cancer (12), esophageal cancer (5, 6), colorectal cancer (13), oral squamous cell carcinoma (14), pancreatic cancer (15), rectal cancer (16), and hepatocellular carcinoma (17). In most of the reports, higher NLR was associated with more aggressive tumor behavior and worse prognosis. The prognostic value of NLR was also explored in NSCLC. For example, 2 groups from the UK and Japan independently assessed its prognostic value in patients with operable NSCLC, and both reported an independent negative impact of NLR on survival (18, 19). However, these were only small-scale studies, one of which was performed years ago using the old staging system, while the other enrolled confounding patients with locally advanced disease as well. Compared with these reports, the present study strongly supports the independent prognostic significance of NLR in patients with metastatic NSCLC treated with chemotherapy.
The negative prognostic value of NLR may have a mechanistic explanation. Cancer educates the regional immune response and releases inflammatory factors such as vascular endothelial growth factor and interleukin-18 (4, 20). The notorious contribution of chronic inflammation to carcinogenesis and cancer progression is well documented (21). The increased neutrophils suppress the activities of natural killer cells and cytotoxic T cells, and provide supplies of inhibitory cytokines such as tumor necrosis factor, interleukin-1 etc. (22, 23). Meanwhile, the reduced lymphocytes hamper the cellular immune response against cancer (24). Thus a high NLR (elevated neutrophils and/or decreased lymphocytes) might promote tumor growth and lead to a worse prognosis.
NLR was found to be a prognostic biomarker capable of predicting survival. However, it did not correlate with the PFS of chemotherapy-treated patients in the Cox regression model. Our results implied NLR was related to long-term survival, independent of the effectiveness of first-line chemotherapy. Our results, together with those of others (10), were in good concordance with its supposed role in chronic inflammation. NLR might represent a convenient, inexpensive biomarker of inflammation for cancer.
We also checked the influence of metabolic markers such as albumin, body mass index, etc. These were proposed as prognostic factors in some reports, but the results have remained largely inconclusive (25-26-27-28). The present study failed to prove their prognostic value. However, we found a positive relationship between CYFRA21-1 and survival, an observation that was supported by other reports (28).
In this study, positive correlations were found between NLR and LDH, albumin, and CYFRA21-1. Similar observations were reported by others (10, 29, 30). The mechanisms underlying these relationships are unclear at the moment, but it could be reasoned that they were all aspects of chronic inflammation. The interaction among them must be complex, especially in cancer. One could infer that all these factors contribute to tumor progression.
One problem in the validation of NLR as a prognostic factor has been the lack of a definite cutoff value. Arbitrary values (3.0, 4.0 or 5.0) were adopted in the literature (10, 29, 30). This study used ROC analysis to determine the optimal cutoff value. A similar analysis was used in another report (31). Actually, we also examined other cutoff values. The median value (NLR = 3.09) perfectly dichotomized patients into good- and poor-prognosis groups (p = 0.049, data not shown). These results further confirmed its prognostic value regardless of the cutoff value. More studies are warranted to facilitate the translation of NLR to clinical practice.
It should be noted that NLR can be influenced by other factors as well, such as infection and coronary disease (32). In addition, the sample size of this study was relatively small and bias was inevitable due to the study's retrospective nature. However, the findings of this study strongly argue that baseline NLR could be a convenient, cost-effective marker for everyday clinical practice. Patients with high NLR had a poor prognosis, and would be candidates for other aggressive treatments. Those who benefited from chemotherapy would be selected and in such a way the outcome of NSCLC would probably improve. Further prospective and large-scale studies are warranted to confirm the prognostic significance of NLR.
Footnotes
Conflict of interest: The authors have no conflict of interest to disclose.
Financial support: This work was supported by the National Natural Science Foundation of China (81272684, 81200640 and 81301493).
