Abstract
Background
When advanced, oral squamous cell carcinoma (OSCC) may involve adjacent non-epithelial structures, and the prognosis is worse for bone invasion. Human relaxin-2 is a peptide hormone that has recently been associated with cancer. It can induce human osteoclast differentiation and activation, suggesting a role in tumor-driven osteolysis. This study was a preliminary assessment of the prognostic role of relaxin-2 in surgical specimens of OSCC tissue and adjacent but uninvolved mandibular/maxillary bone.
Methods
Relaxin-2 immunohistochemical expression and reaction intensity were assessed in tumor and uninvolved adjacent mandibular/maxillary bone specimens from 23 operated OSCC patients.
Results
All OSCC specimens were positive for relaxin-2. The intensity of its reaction in OSCC correlated significantly with the pattern of the tumor's invasion front (p = 0.02), being higher with the infiltrative pattern. Mean relaxin-2 immunohistochemical expression was higher in patients whose OSCC recurred after treatment than those experiencing no recurrence (81.3% ± 22.6% vs. 59.5% ± 29.7%, respectively). A significant direct association emerged between relaxin-2 expression in OSCC specimens and recurrence rate (p = 0.049).
Conclusions
Relaxin-2 expression in OSCC should be further investigated as a potentially useful marker for identifying patients at higher risk of recurrence, who might benefit from closer follow-up and more aggressive adjuvant therapy. In other oncological settings, increasing evidence of relaxin being produced by cancer cells is prompting efforts to synthesize human relaxin-2 analogs capable of acting as antagonists and limiting tumor growth.
Introduction
In 2012, there were an estimated 300,400 new cases of oral cavity cancer (including lip cancer) worldwide, and 145,400 deaths (1). In its advanced stages, oral squamous cell carcinoma (OSCC) may involve adjacent non-epithelial structures: according to the literature, the incidence of mandibular invasion ranges from 12% to 56% (2). Whereas some authors have reported an association with adverse outcomes in univariate analysis, other studies did not support this finding (3). To what extent the prognosis is worsened by bone invasion has yet to be clearly established, mainly because of the limited data available (4). The pattern of tumor invasion may be infiltrative or expansive. In the former, the front of invasion is jagged, with islands and fingers of tumor cells penetrating inside the tissue layers they encounter. In the latter, the tumor has a broad, smooth invasion front that pushes against and displaces the affected structures.
Molecular changes occur in malignancies before any morphological changes become apparent, and they are responsible for the disease's biological behavior, prognosis, and response to therapy. Relaxin was discovered and named by Dr. Frederick Hisaw based on his studies on the reproductive endocrinology of the gopher and, later on, the guinea pig (5). Human relaxin-2 is a 6-kDa peptide hormone with a structure similar to that of insulin (6). It exerts its numerous effects by binding to the relaxin family peptide receptor 1. Relaxin is involved in a number of physiological and pathogenic processes, including collagen and cardiovascular regulation and tissue remodeling in pregnancy and cancer (7). It is only quite recently that relaxin-2 has been associated with cancer biology. Its overexpression has been putatively attributed a number of roles, including the modulation of tumor growth, neovascularization, metastasis and oncogenic progression (8). Relaxin-2 has been mainly implicated in tumors that metastasize to bone (9), but it has been associated with primary osteosarcoma too (10). In osteolytic metastases, bone destruction is mediated by osteoclasts induced by tumor cells that produce osteoclast-activating factors. It has recently been demonstrated that relaxin-2 is capable of inducing the complete differentiation and activation of human osteoclasts, pointing to a role for relaxin-2 in metastatic–tumor-driven osteolysis (11). Relaxin-2 also appears to have an important part to play in extracellular matrix degradation and tumor cell motility, modulated by matrix metalloproteinase expression (12). In 2013, Ma et al (9) studied the clinical and pathological roles of serum relaxin-2 and tissue relaxin-2 expression levels in human primary osteosarcoma. The incidence of advanced-stage cancer and hematogenous metastatic cancer in the groups with high relaxin-2 mRNA expression and high serum relaxin-2 levels was significantly higher than in the groups with low relaxin-2 expression and low serum relaxin-2 levels. Using immunohistochemistry to examine the expression of relaxin-2 and S100A4 in 130 specimens of primary osteosarcoma, Huang et al (13) concluded that relaxin-2 and S100A4 overexpression might be related to the likelihood of metastasis and a poor prognosis in osteosarcoma patients. In 1997, after the recognition of the mammotrophic action of relaxin, Bani (14) hypothesized that further research might show that this peptide also influences the behavior of breast cancer cells. To investigate the potential role of relaxin in the progression of breast cancer in vivo, Binder et al (15) measured serum concentrations of relaxin in 160 breast cancer patients being followed up after surgery. The authors concluded that the detection of high relaxin concentrations, especially in patients with metastases, supported the assumption of a role for this hormone in tissue remodeling during breast cancer progression. The part it plays in human thyroid carcinoma is still debated (12, 16). In 2013, Ren et al (17) assessed the prognostic value of serum human relaxin-2 levels in patients with esophageal squamous cell carcinoma. Patients with high relaxin-2 levels had a poorer prognosis than patients with low levels: furthermore, the serum human relaxin-2 level was correlated with survival and tumor-node-metastasis staging. Very recently, our group investigated relaxin-2 immunohistochemical expression in 25 consecutively operated patients with temporal bone carcinoma: their recurrence rates, disease-free survival (DFS) and disease-specific survival did not correlate with the relaxin-2 expression in their carcinoma specimens or pathologically negative adjacent bone specimens (18).
The present study investigated relaxin-2 expression in patients with primary OSCC with and without bone involvement. The aim of the study was to conduct a preliminary investigation into the prognostic role of relaxin-2 expression in surgical specimens of OSCC tissue and adjacent but uninvolved mandibular/maxillary bone.
Materials and methods
Patients
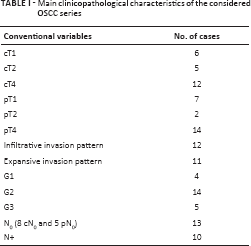
The study was conducted on specimens resected from 23 patients with primary OSCC who underwent surgery at the Otolaryngology Section or Maxillofacial Surgery Unit of the Neurosciences Department at Padua University (Italy). The present investigation was approved by the Otolaryngology Section's in-house committee. The study was conducted in accordance with the principles of the Helsinki Declaration. The patients included 14 men and 9 women with a mean age of 63.5 ± 13.4 years. Preoperatively, all patients underwent oral biopsy, upper aerodigestive tract endoscopy, neck ultrasonography (with or without fine needle aspiration cytology), head and neck contrast-enhanced computerized tomography (CT) and/or magnetic resonance imaging (5 cases), chest x-ray, and liver ultrasonography. Positron emission tomography (PET) was performed in selected cases. The primary OSCCs were classified as cT1 in 6 cases, cT2 in 5, and cT4a in 12, according to the seventh edition of the TNM classification of malignant tumors (19). No distant metastases were detected at diagnosis.
Before surgery, all patients signed a detailed informed consent form. Mandibulectomy was performed in 19 cases and partial maxillectomy in 4. Surgical reconstruction with a free flap was performed in 12 cases. Fifteen patients underwent cervical lymph node dissection. The surgical margins were negative on definitive histopathological examination in all cases. Pathological T staging established that 7 cases were pT1, 2 were pT2, and 14 were pT4. The regional lymph nodes were classified as N0 in 13 cases (8 cN0 and 5 pN0), and as pN+ in 10 (6 pN1, 1 pN2a, 2 pN2b, and 1 pN2c). Table I summarizes the main clinicopathological characteristics of the considered series. Postoperative radiotherapy was administered in 10 cases using conventional fractionation, i.e., 1.8-2 Gy/fraction once a day, with a total dose ranging from 50 to 70 Gy (median 60 Gy). Five patients were also given postoperative adjuvant chemotherapy.
Main clinicopathological characteristics of the considered OSCC series
The clinical follow-up (adjustable to patients’ individual characteristics) was scheduled as follows: once a month for the first year after treatment; every 2 months in the second year; every 3 months in the third year; every 4 months in the fourth year; every 6 months in the fifth year; and yearly thereafter. Neck ultrasonography and chest x-rays were also performed at least yearly. Contrast-enhanced CT or MRI of the oral cavity and of the neck in advanced cases, total body PET-CT, chest CT, and liver ultrasonography were repeated as necessary. The median follow-up for the cohort was 60.0 months (mean 63.9 ± 46.4 months).
Immunohistochemistry
Four-micron-thick sections were cut from each formalin-fixed and paraffin-embedded (FFPE) tissue block for immunohistochemistry. Immunostaining was done with a fully automated system (BOND-MAX; Leica Biosystems). Sections were incubated with rabbit anti-relaxin-2 (polyclonal; Phoenix Pharmaceuticals; dilution 1:350; citrate buffer) for 30 minutes at room temperature and detected using a compact horseradish peroxidase-conjugated polymer system according to the manufacturer's protocol.
Staining was visualized with 3,3’-diaminobenzidine and the slides were lightly counterstained with hematoxylin. Sections were then dehydrated, cleared and mounted. FFPE human placenta samples were used as positive controls, and serum without the primary antibody served as a negative control.
Both the tumor and the adjacent but uninvolved mandibular/maxillary bone samples were jointly assessed by 3 pathologists in the same fields. The percentage of positive cells was recorded and the intensity of the immunoreaction was scored on a 4-tier scale: 0 = no staining, 1 = weak, 2 = moderate, and 3 = strong staining. Agreement about the staining intensity was reached in all cases.
Statistical Analysis
The statistical tests applied were Fisher's exact test, the Kruskal-Wallis test, and Spearman's rank correlation test, as appropriate. The log-rank test and Cox's regression model were used to analyze DFS after stratifying patients in the light of the clinical, pathological and immunohistochemical variables considered. The receiver operating curve (ROC) approach (failure versus parameter) was used to establish the analytically best-fitting cutoff for binarizing the variables (relaxin-2 expression, relaxin-2 reaction intensity) according to the highest level of the positive likelihood ratio. The best performance coincides with an area under the ROC curve (AUC) of 1.0.
A p value <0.05 was considered significant. The STATA™ 8.1 (StataCorp LP) statistical package was used for all analyses.
Results
Patients’ Clinical Outcomes
Twelve patients developed local and/or regional recurrences of their OSCC after a mean period of 32.7 ± 34.8 months. The Kruskal-Wallis test ruled out any significant difference between mean follow-up (in months) in patients with and without recurrent disease (p = 0.91). Statistical analysis showed no significant associations between cT or cN classification and recurrence rate (Fisher's exact test, p = 0.22 and p = 0.82, respectively) or DFS (log-rank test, p = 0.68 and p = 0.81, respectively).
Fisher's exact test revealed a significant association between pT classification and lymph node status (N) (p = 0.005), but not between pT and pathological grade (p = 0.44) or recurrence (p = 0.51). The log-rank test identified no significant difference in DFS when patients were classified by pT (p = 0.68). Neoplastic bone involvement was confirmed histologically in 13 cases (with medullary involvement in 9). The pattern of primary tumor invasion (infiltrative vs. expansive), ascertained by conventional pathology in all cases, did not correlate significantly with pT classification (Fisher's exact test, p = 1), N classification (Fisher's exact test, p = 0.41), pathological stage (Fisher's exact test, p = 0.73), pathological grade (Fisher's exact test, p = 1), or recurrence rate (Fisher's exact test, p = 1). The log-rank test showed no significant difference in DFS when patients were stratified by pattern of primary OSCC invasion (p = 0.87). No significant associations were found between pathological grade and recurrence rate (Fisher's exact test, p = 0.09) or DFS (log-rank test, p = 0.08). N classification was significantly associated with recurrence rate (Fisher's exact test, p = 0.01) and DFS in months (log-rank test, p = 0.01), whereas pathological stage was unrelated to recurrence rate (Fisher's exact test, p = 0.31) or DFS (log-rank test, p = 0.68). Postoperative radiotherapy did not correlate significantly with recurrence rate (Fisher's exact test, p = 0.10) or DFS (log-rank test, p = 0.06).
Relaxin-2 Expression and Clinical and Pathological Features of OSCC
The subcellular localization of relaxin-2 was always cytoplasmic. The normal oral mucosa showed weak focal immunostaining for relaxin-2, while the normal bone exhibited a diffuse weak-to-moderate immunoreaction (Fig. 1). All the OSCC specimens were positive for relaxin-2 (Fig. 1). The less differentiated tumor areas showed moderate-to-strong relaxin-2 immunostaining, while the intensity of the immunoreaction was weak in the well-differentiated neoplastic areas.
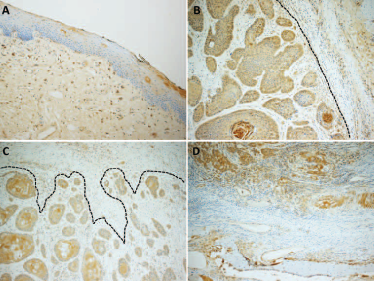
Representative images of relaxin-2 immunohistochemistry from our series: normal oral mucosa (
The percentage of immunostained cells and the relaxin-2 intensity scores were assessed in specimens of both OSCC and adjacent but uninvolved mandibular/maxillary bone (Fig. 1). The distribution of the positive cells in the carcinomas was always diffuse.
Statistical analysis ruled out any significant associations between the percentage of OSCC cells staining positive for relaxin-2 and pT classification (Kruskal-Wallis test, p = 0.25; Fig. 2), pattern of primary tumor invasion front (Kruskal-Wallis test, p = 0.42), pathological grade (Kruskal-Wallis test, p = 0.56), N classification (Kruskal-Wallis test, p = 0.24), or pathological stage (Kruskal-Wallis test, p = 0.40). The intensity of the relaxin-2 reaction in OSCC correlated significantly with the pattern of tumor invasion (Kruskal-Wallis test, p = 0.02). On the other hand, the Kruskal-Wallis test revealed no significant associations between relaxin-2 reaction intensity in OSCC and pT classification (p = 0.25; Fig. 2), pathological grade (p = 0.61), N classification (p = 0.71), or pathological stage (p = 0.26).
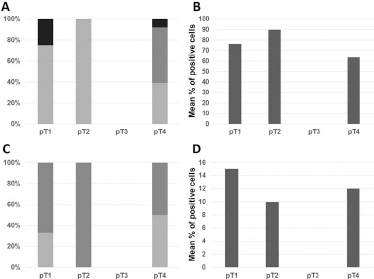
The graphs show the distribution of the weak (light gray), moderate (dark gray), and strong (black) immunoreaction for relaxin-2 among the pT categories in oral squamous cell carcinoma (
As for the pathologically negative surgical specimens of adjacent bone, relaxin-2 expression was unassociated with pT classification (Kruskal-Wallis test, p = 0.77; Fig. 2), pattern of primary tumor invasion (Kruskal-Wallis test, p = 0.65), pathological grade (Kruskal-Wallis test, p = 0.53), N classification (Kruskal-Wallis test, p = 0.35), or pathological stage (Kruskal-Wallis test, p = 0.77). Statistical analysis ruled out any significant association between relaxin-2 reaction intensity in adjacent but uninvolved mandibular/maxillary bone and pT classification (Kruskal-Wallis test, p = 0.29; Fig. 2), pattern of primary tumor invasion (Kruskal-Wallis test, p = 0.86), pathological grade (Kruskal-Wallis test, p = 0.29), N classification (Kruskal-Wallis test, p = 0.60), or pathological stage (Kruskal-Wallis test, p = 0.68).
The expression of relaxin-2 in OSCC did not correlate significantly with its expression in the surgical specimens of adjacent bone (Spearman's rank correlation test, p = 0.80), nor did the relaxin-2 reaction intensity in OSCC correlate significantly with its intensity in adjacent but uninvolved mandibular/maxillary bone (Spearman's rank correlation test, p = 0.98).
Relaxin-2 Expression and Prognosis in OSCC
The mean levels of relaxin-2 expression in samples of OSCCs that did and did not recur after treatment were 81.3% ± 22.6% and 59.5% ± 29.7%, respectively. The Kruskal-Wallis test revealed a significant direct association between recurrence rate and relaxin-2 expression in the OSCC specimens (p = 0.049). On the other hand, statistical analysis found no significant association between recurrence rate and relaxin-2 reaction intensity in OSCC (Kruskal-Wallis test, p = 0.26). The Kruskal-Wallis test also ruled out any associations between recurrence rate and relaxin-2 expression or reaction intensity in pathologically negative surgical specimens of adjacent bone (p = 0.34 and p = 0.27, respectively).
Our statistical analysis also found no significant differences in DFS (in months) when patients were stratified by their relaxin-2 expression in OSCC (the best-fitting cutoff calculated was 60%, AUC 0.74, 95% confidence interval [CI] 0.53-0.95; Cox's regression model, p = 0.43), their relaxin-2 reaction intensity in OSCC (best-fitting cutoff 2, AUC 0.37, 95% CI 0.15-0.59; Cox's regression model, p = 0.50), their relaxin-2 expression in pathologically negative adjacent bone (best-fitting cutoff 70%, AUC 0.36, 95% CI 0.06-0.66; Cox's regression model, p = 0.18), or their relaxin-2 reaction intensity in pathologically negative adjacent bone (best-fitting cutoff 3, AUC 0.36, 95% CI 0.12-0.60; Cox's regression model, p = 0.08).
Discussion
The main strength of this study lies in the homogeneity of the series of patients considered, given that (i) all patients underwent primary surgery, and (ii) only surgical specimens (not biopsies) of OSCC and pathologically negative adjacent bone were considered for immunohistochemical examination. In addition, the pathologists in our research group had already gained specific experience of measuring and analyzing relaxin-2 immunohistochemical expression in squamous cell carcinoma of the head and neck (18). The main weaknesses of our investigation concern (i) the retrospective setting, and (ii) the limited number of OSCC cases involved.
In several academic and non-academic institutions, head and neck surgeons, biologists, and pathologists are working towards a scenario in which molecular markers may support standard clinical and pathological classifications in predicting the risk of carcinoma recurrence (20). Further efforts are needed to elucidate the mechanisms behind the locoregional recurrence of head and neck cancers despite their rational curative treatments (21, 22). Relaxin has been implicated as an important autocrine/paracrine factor in tumor biology. It has been involved in various mechanisms associated with tumor cell growth, local invasion and metastasis in several malignancies, especially those metastasizing to bone. Neoplastic destruction of bone is primarily mediated by osteoclasts (the cells that resorb bone), and the bone microenvironment plays a crucial part in osteoclast differentiation and activation. It has been reported that relaxin is capable of inducing human osteoclast differentiation and activation, and relaxin-induced osteoclasts seem to be fully differentiated and capable of reabsorbing bone (11). In the available literature, relaxin-2 expression has been investigated in a few oncological settings, mainly involving osteosarcoma and carcinoma of the breast, prostate, liver, and thyroid. To the best of our knowledge, relaxin-2 expression and its potential prognostic value in OSCC had never been investigated before.
Hypothesizing that hidden microscopic pathways of spread in OSCC may be involved in this malignancy's potential for aggressiveness and high local recurrence rates, we investigated the prognostic role of relaxin-2 immunohistochemical expression in surgical specimens of OSCC and pathologically negative adjacent bone. The relatively high local recurrence rate of OSCC (even in patients with a definitive pathology report of free surgical margins) is a necessarily debated and difficult issue that might reasonably also relate to neoplastic bone invasion escaping conventional pathological investigations. Our study found higher mean immunohistochemical expression of relaxin-2 in patients whose OSCC recurred after treatment than in patients experiencing no recurrent disease (81.3% ± 22.6% vs. 59.5% ± 29.7%, respectively). Our statistical analysis showed a significant direct association between relaxin-2 expression and recurrence rate in the OSCC specimens considered (p = 0.049). The reaction intensity of the relaxin-2 in OSCC also correlated significantly with the pattern of the tumor's invasion front (p = 0.02), being higher in the case of an infiltrative pattern. Tumor invasion patterns have been widely described as important predictors of local recurrence and survival in surgically treated patients with OSCC. In a series of previously untreated patients who underwent surgery for squamous cell carcinoma of the oral tongue, Spiro et al (23) concluded that more infiltrative patterns at the tumor/host interface were associated with a higher incidence of nodal and distant metastasis, as well as a significantly shorter survival. Our present – albeit very preliminary – findings seem to support the possibility of a mechanism of local aggressiveness in OSCC that is also mediated by relaxin-2. On the other hand, as found recently in the case of temporal bone carcinoma (18), no associations emerged between recurrence rate or DFS and relaxin-2 expression in pathologically negative surgical specimens of bone adjacent to OSCC in the present study.
In conclusion, relaxin-2 expression needs further investigation based on larger and prospective series to confirm that in OSCC it could be a prognostic marker potentially capable of identifying patients at higher risk of recurrence after treatment, who might benefit from closer follow-up protocols and more aggressive adjuvant therapy. It is well known that, in the setting of investigations into molecular markers involved in the biology of OSCC and their potential clinical applications, the more ambitious goal should be to identify targets for therapeutic agents. The development of integrated therapeutic strategies (including targeted treatments) could be crucial to improving the prognosis for patients with advanced OSCC. In other oncological settings, the increasing evidence of relaxin being produced by cancer cells and possibly taking action in an autocrine manner on the relaxin family peptide 1 receptors, thereby promoting the growth and invasiveness of several types of neoplasm (6), is prompting efforts to synthesize human relaxin-2 analogs that might act as antagonists and limit tumor growth. Generally speaking, attempts to interfere with relaxin are justifiable because it affects a variety of pathophysiological mechanisms (angiogenesis, extracellular matrix remodeling, cell proliferation, apoptosis) and may therefore be involved on several levels in the development and progression of neoplasms.
Footnotes
Acknowledgment
The authors thank Frances Coburn for correcting the English version of this paper.
Financial support: This study was partly supported by grants No. 60A07-1341/12 (G. Marioni) and No. 60A07-8983/13 (A. Martini) from the University of Padua, Italy.
Conflict of interest: None.
