Abstract
Aim
Breast carcinoma (BCA) and diabetes mellitus (DM) are two major health problems in women and the general population. Cullin-1 is reported to be an important tumor-related protein involved in cell-cycle progression, signal transduction and transcription. The aim of this work is to investigate the role of Cullin-1 in the development of BCA and to find potential relationships between Cullin-1 and diabetes in BCA patients.
Methods
To evaluate the function of Cullin-1, we entered 168 patients with primary invasive BCA in this study. Pairs of BCA tissues and adjacent noncancerous tissues from these patients were collected between 2006 and 2008. We used immunohistochemistry to analyze the correlation between Cullin-1 expression and clinicopathological variables and patient survival. In addition, we investigated the role of Cullin-1 in BCA cell proliferation.
Results
Cullin-1 expression was upregulated in BCA tissues. Enhanced immunoreactivity for Cullin-1 in BCA tissues was inversely correlated with overall survival and disease-free survival, which suggested a poor prognosis in BCA patients. Strong expression of Cullin-1 was more frequently observed in patients with estrogen receptor negativity and HER2 positivity. We also found that Cullin-1 expression was increased in BCA patients with a previous diagnosis of diabetes.
Conclusions
Our results demonstrate that increased Cullin-1 expression is significantly correlated with poor prognosis in patients with BCA. Cullin-1 might regulate BCA cell proliferation through the ubiquitin-proteasome system. Thus, Cullin-1 might be an important marker and a therapeutic target in BCA.
Introduction
Breast carcinoma (BCA) is the leading neoplastic malignancy among women worldwide. Several BCA risk factors have been known for many years. Among these, age, a family history of BCA, early age at menarche, late age at menopause, nulliparity, late age at first birth, tall height, high body mass index, and alcohol intake are established risk factors (1). Another major health problem in industrial countries is diabetes, which shows a rising trend (2). The incidence of diabetes is higher in BCA patients at 20% compared to the general population, suggesting that blood glucose control may be important for BCA control (3). In several cohort studies it has been shown that diabetes increases both the overall and cancer-specific mortality in the general population and in BCA patients (4, 5). Evidence supporting the association between preexisting diabetes and overall mortality in BCA is extensive. However, the potential interrelations between diabetes mellitus and BCA are complicated and remain to be elucidated.
The cullin family of proteins was first identified in 1996 as being required for cell cycle exit in Caenorhabditis elegans (6) and for the G1-to-S-phase transition in budding yeast (7). Members of the cullin family (Cullin-1, Cullin-2, Cullin-3, Cullin-4A, Cullin-4B, Cullin-5, Cullin-7, PARC and APC2) provide the scaffold of Cullin-RING ubiquitin ligase (CRL) complexes responsible for a large portion of ubiquitin-proteasome system (UPS)-mediated proteolysis. The family members are characterized by an evolutionarily conserved cullin-homology domain, and serve as molecular scaffolds to facilitate the assembly of multimeric CRLs with SCF/CRL-1 as the founding member. Cullin-1 is the most extensively characterized member of the cullin family and a key component of SCF E3s, which play an indispensable role in both cell cycle progression and early embryogenesis.
This work was aimed at identifying the key molecules involved in diabetes mellitus and BCA so as to find potential targets for BCA therapy. To elucidate the function of Cullin-1, we used immunohistochemistry and analyzed the correlation between Cullin-1 expression and clinicopathological variables and patient survival. In addition, we investigated the role of Cullin-1 in BCA cell proliferation.
Materials and methods
Study Participants
A total of 168 patients with primary invasive BCA were included in the study. BCA tissues and adjacent noncancerous tissues (NCTs) were collected between 2006 and 2008 from Nanjing Hospital of Traditional Chinese Medicine, the Second Affiliated Hospital of Soochow University, and the Affiliated Hospital of Nanjing University of Chinese Medicine. Tissue samples were immediately snap-frozen in liquid nitrogen. All human materials were obtained with informed consent. The clinical information of the BCA patients is presented in Table I. The study population included 62 patients with a previous history of diabetes. Patients with a previous history of malignant tumors were excluded from the study. The Clinical Research Ethics Committee of Nanjing University approved the research protocols and written informed consent was obtained from the participants.
RNA Extraction and Quantitative Real-Time Reverse Transcription Polymerase Chain Reaction (qRT-PCR)
Total RNA was extracted using the TRIzol reagent (Invitrogen) according to the manufacturer's instructions. The concentrations were determined using a NanoDrop 1000 spectrophotometer (Thermo Scientific). cDNA was synthesized with the PrimeScript RT reagent kit (TaKaRa) using 500 ng total RNA as the template. qRT-PCR analyses were conducted to quantitate the relative mRNA expression using SYBR Premix Ex Taq (TaKaRa) with beta-actin as internal control. The results were defined from the threshold cycle (Ct), and relative expression levels were calculated by means of the 2-∆∆Ct method (8). PCR was performed using an ABI 7900HT instrument (Applied Biosystems). The primers used for PCR analysis are listed in the supplementary material (Supplementary Table S1 - Primer sequences. Available online at www.biological-markers.com).
Immunohistochemical Staining
Tissue arrays were constructed using the 168 paired BCA tissues and NCTs. Immunohistochemical staining was performed on 4-μm sections of paraffin-embedded tissues. Cullin-1 antibody (Abgent) and estrogen receptor (ER), progesterone receptor (PR) and HER2 antibodies (Roche) were prepared. To determine the expression level of Cullin-1 protein, slides were incubated in Cullin-1 antibody diluted 1:200 at 4°C overnight. The subsequent steps were performed using the EnVision FLEX High pH visualization system (Dako) according to the manufacturer's instructions. The ER, PR and HER2 proteins were tested using the Benchmark XT (Roche) according to the manufacturer's instructions. The HER2-positive score was based on the 2013 ASCO guidelines.
Cell Culture and Cell Proliferation Assay
Two human BCA cell lines, MDA-MB-231 and BT-549, were purchased from the American Type Culture Collection. MDA-MB-231 and BT-549 cells were cultured in RPMI 1640 medium supplemented with 10% fetal bovine serum (Invitrogen). HEK-293T cells were cultured in DMEM. The cells were incubated with 5% CO2 at 37°C. Cell proliferation was quantified using the Cell Counting Kit-8 (CCK-8, Dojindo Laboratories) according to the manufacturer's instructions. Two thousand cells were planted into 96-well plates and OD450 was tested every 24 hours.
Vector Construction and Oligonucleotide Transfection
The open reading frame (ORF) of Cullin-1 was amplified by nested PCR and cloned into the pLVX-IRES-Neo vector (Clontech). The primers and endonuclease sites used for the vector constructs are shown in the supplementary material (Supplementary Table S1). Virus particles were harvested 48 hours after cotransfecting pLVX-Cullin-1 with the packaging plasmid ps-PAX2 and the envelope plasmid pMD2G into HEK-293T cells using Lipofectamine 2000 reagent (Invitrogen). MDA-MB-231 and BT-549 cells were infected with recombinant lentivirus-transducing units plus 6 μg/mL polybrene (Sigma). Small interfering RNAs (siRNAs) of Cullin-1 were synthesized (Ribobio). Oligonucleotide transfection was performed using Lipofectamine 2000 reagent according to the manufacturer's instructions. The final siRNA concentration in the transfection mixture was 50 nM.
Xenograft Tumor Model
Male BALB/c athymic nude mice at 5 weeks of age (purchased from Vital River) were bred in a licensed SPF (special pathogen-free) grade laboratory. A total of 3 × 106 cells (MDA-MB-231 and BT-549, stably expressing Cullin-1 or the vector control) in 200 μL of RPMI 1640 were injected subcutaneously in the right flank of each mouse. After transplantation, the growth of the subcutaneous tumors was assessed twice a week. Tumor size was monitored by measuring the length and width with calipers, and volumes were calculated with the formula (L × W2) × 0.5, where L is the length and W the width of the tumor. The mice were sacrificed after a period of 6-7 weeks, and the weights of the subcutaneous tumors were recorded. All protocols were approved by the Nanjing University animal ethics committee.
Statistical Analyses
The results are presented as mean values ± standard errors of the mean. The association between Cullin-1 staining and the clinicopathological parameters of the BCA patients were evaluated by the χ2 test. Overall survival rates were calculated actuarially according to the Kaplan-Meier method with the log-rank test and were measured from the day of surgery. Differences between groups were estimated using the χ2 test, Student's t-test, the Mann-Whitney U-test, and repeated-measures ANOVA test. Relationships were explored by Spearman's correlation. P values <0.05 were considered statistically significant.
Results
Clinicopathological Features of the Patients
One hundred sixty-eight patients with primary invasive BCA were included in the study. All patients were pathologically diagnosed as having invasive ductal carcinoma. Sixty-two patients had also been diagnosed with type 2 diabetes. The clinicopathological features of the study group are summarized in Table I.
Patient information

IDC = invasive ductal carcinoma.
Cullin-1 Expression in Human BCA Tissues
The expression level of Cullin-1 was remarkably upregulated in 110 of the 168 (65.5%) BCA tissues when compared to the corresponding NCTs by qRT-PCR (Fig. 1A, 1B). Only 17 cases (10.1%) were downregulated while 41 cases (24.4%) were unchanged. To further demonstrate Cullin-1 protein expression levels in BCAs, immunohistochemistry was performed. The Cullin-1 protein showed typical nuclear staining in BCA tissues. The cases were scored from 0 to 3 according to the percentage of positive cells (score 1: 0%-25%, score 2: 25%-50%, score 3: 51%-75%, score 4: 76%-100%) (9). The BCAs scored as 1-2 were designated as Cullin-1 low expression while those scored as 3-4 were designated as Cullin-1 high expression (Fig. 2A). Importantly, enhanced immunoreactivity of Cullin-1 in BCA tissues was inversely correlated with overall survival (OS) and disease-free survival (DFS), which suggested a poor prognosis for these BCA patients (Fig. 2B, 2C).
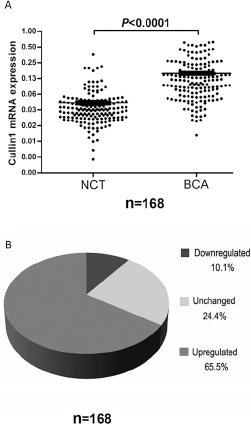
Cullin-1 mRNA expression in BCA patients. (
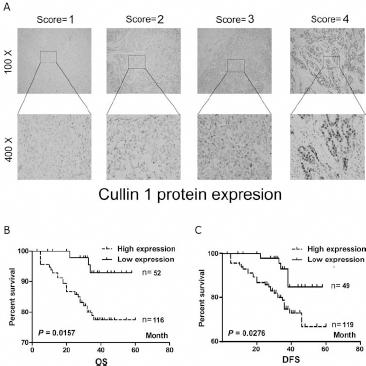
Cullin-1 protein expression in BCA patients. (
Correlation of Cullin-1 Expression and ER, PR and HER2 in BCAs
ER, PR and HER2 status are significant parameters for the classification, clinical treatment and outcome of BCA. For the purpose of defining the relationship between Cullin-1 expression and ER, PR and HER2 status, protein expression was tested and the results are summarized in Table II. High expression of Cullin-1 was more frequently observed in patients with ER negativity and HER2 positivity. No association was observed between the intensity of Cullin-1 staining and PR status.
Correlation of Cullin-1 expression with ER, PR and HER2 status
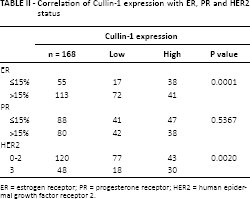
ER = estrogen receptor; PR = progesterone receptor; HER2 = human epidermal growth factor receptor 2.
Cullin-1 Promotes BCA Cell Proliferation in Vitro and in Vivo
The high expression of Cullin-1 in BCA tissues suggests it contributes to tumorigenesis. To confirm this inference, a cell proliferation assay revealed that overexpression of Cullin-1 significantly enhanced the growth rate of MDA-MB-231 and BT-549 cells (Fig. 3A, 3B). In contrast, silencing of Cullin-1 expression with siRNA significantly repressed the growth of MDA-MB-231 and BT-549 cells (Fig. 3C, 3D). To further assess the function of Cullin-1 in vivo, Cullin-1 overexpressing stable cell lines (MDA-MB-231 and BT-549) were constructed and tumor formation assays were run in a nude mouse model. Compared with the control group, overexpression of Cullin-1 was found to promote tumor growth with a remarkable increase in tumor volume and weight in nude mice (Fig. 4A, 4B).
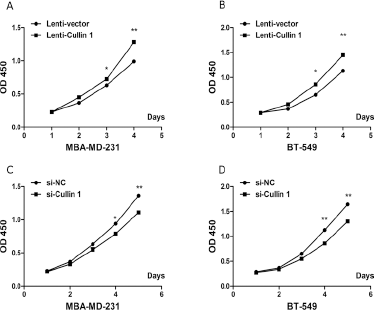
Cullin-1 promotes cell proliferation in vitro. (
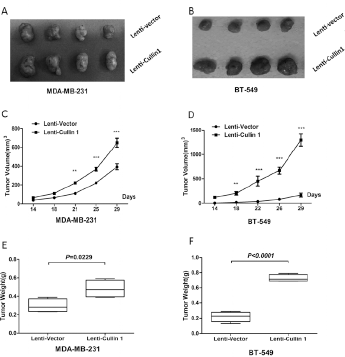
Cullin-1 enhances BCA cell growth in vivo. (
Cullin-1 Expression in BCA Patients Diagnosed with Diabetes before BCA
In patients with BCA, diabetes diagnosed prior to cancer is associated with advanced cancer stage and increased mortality (2). We therefore analyzed Cullin-1 mRNA and protein expression in tissues of patients with BCA only and patients with diabetes and BCA (D-BCA). Using qRT-PCR, we found that Cullin-1 was significantly upregulated in 62 D-BCA tissues compared with the corresponding NCTs (p<0.0001, Fig. 5A). Moreover, high expression of Cullin-1 was observed in D-BCA tissues compared with BCA tissues (Fig. 5B). Cullin-1 protein expression was also frequently increased in D-BCA tissues compared with BCA tissues (Fig. 5C, 5D).
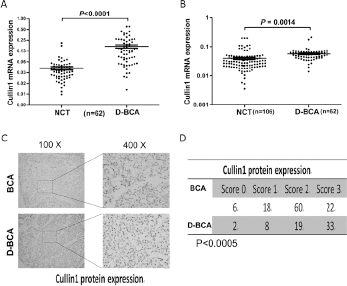
Cullin-1 expression in D-BCA patients. (
Discussion
BCA is a common malignancy in women (10). In many countries and regions, it is the most common malignant tumor in women, and as a result it is one of the main causes of death in women (11, 12). Diabetes is an endocrine disease with an increasing annual incidence rate. A report has suggested that the incidence rate increases by 7% every 10 years (4). Epidemiological evidence suggests preexisting diabetes is associated with increased risk of BCA (13), advanced cancer stage at diagnosis (4, 14), altered treatment regimens (3, 15), chemotherapy toxicity (3), BCA mortality in the general population (5, 16), and overall mortality in those diagnosed with BCA (17).
The ubiquitin-proteasome system plays an important role in maintaining the balance between normal growth and uncontrolled proliferation by regulating a large variety of cellular proteins (18). The cullin family of ubiquitin ligases, including Cullin-1, 2, 3, 4A, 4B, 5 and 7, represents the largest class of E3 ligases, with Cullin-1 being the most extensively characterized family member and a key component of SCF E3s (18). This is the first study to investigate Cullin-1 in BCA and compare its expression between BCA tissues and D-BCA tissues. In this study, High expression of Cullin-1 in BCA tissues was inversely correlated with OS and DFS. Which was similar to the results reported by Bai et al (19). Furthermore, higher expression of Cullin-1 was observed in D-BCA tissues than BCA tissues, and Cullin-1 protein expression was more frequently increased in D-BCA tissues than BCA tissues.
ER, PR and HER2 status all influence the prognosis of BCA patients and the likelihood of response to systemic therapies (20). Endocrine therapy is probably the most important systemic therapy for hormone receptor-positive BCA (21). The loss of ER expression is thought to be involved in endocrine therapy resistance. In addition, Min et al (9) reported that Cullin-1 expression was correlated with histological grade, negative ER, and positive HER2, and that it showed a significant association with poor overall survival in BCA. Similarly, our study showed that high expression of Cullin-1 was more frequently observed in patients with ER negativity and HER2 positivity. No association was observed between the intensity of Cullin-1 staining and PR status.
Cullin-mediated substrate degradation involves a wide range of cellular processes such as proliferation, apoptosis and differentiation. Once the cell regulatory mechanisms of cullin encounter malfunctions or perturbations, accumulation of oncoproteins or excessive degradation of tumor suppressors will inevitably occur, which may provoke cells into malignant transformation and tumorigenesis (18). We found that Cullin-1 promotes BCA cell proliferation in vitro and in vivo.
Conclusion
Our results demonstrate that increased Cullin-1 expression is significantly correlated with poor prognosis in patients with BCA. Cullin-1 might regulate BCA cell proliferation through the ubiquitin-proteasome system. Thus, Cullin-1 might be an important marker and a therapeutic target for BCA.
Footnotes
Financial support: Youth Scientific Funding of the National Natural Science Foundation of China, grant number NSFC-81402487.
Conflict of interest: None.
