Abstract
Purpose
Due to the scarcity of publications, guidelines, and harmonization among national regulations, biobanks and institutions face practical and theoretical issues when drafting a material transfer agreement (MTA), the fundamental tool to regulate the successful exchange of biosamples and information. Frequently researchers do not execute MTAs because of a general lack of knowledge about this topic. It is thus critical to develop new models to prevent loss of traceability and opportunities both for researchers and biobanks, their exposure to various risks, and delays in transferring biomaterials.
Methods
Through the involvement of institutional groups and professionals with multidisciplinary expertise, we have drawn up a ready-to-sign MTA for the CRO-Biobank (the biobank of the National Cancer Institute, CRO, Aviano), a standardized template that can be employed as a ready-to-use model agreement.
Results
The team identified the essential components to be included in the MTA, which comprise i) permissions, liability and representations; ii) custodianship and distribution limitations; iii) appropriate use of materials, including biosafety concerns; iv) confidentiality, non-disclosure, and publications; v) intellectual property protection for both the provider and recipient.
Conclusions
This paper aims to be an unabridged report (among the few works in the existing literature) providing a description of the whole process related to the formation of an MTA. Biobanks and institutions may consider adopting our ready-to-sign form as a standard model. The article discusses the most important issues tackled during the drafting of the document, thus proposing an operative approach for other institutions that face the same problems.
Introduction
In recent years, biobanks have been growing, representing the necessary structured organization to support research by providing access to biomaterials and related information. In fact, the availability of well-annotated, prepared and stored biological samples is crucial to the advancement of basic and translational research. Materials, whether or not institutionally managed, stored, and traced through biobanks, very often become instrumental in the context of several types of agreements. Among these agreements, the material transfer agreement (MTA) is definitely the best option, as it governs the transfer of one or more materials (1) from the owner or authorized licensee to third parties for research purposes.
The MTA regulates what is done when a third party asks to supply material to outside sources (MTA-out) or obtain material from such sources (MTA-in): it regulates the liability, sets distribution limitations, outlines the appropriate use of materials (including biosafety concerns), and ensures that proper credit is given to both the provider and recipient. It also provides a written record of the terms of a transfer (1), and this is why some institutions and governments are considering the number of executed MTAs as an indicator of scientific productivity (2). MTAs are also “important mechanisms for ensuring traceability of biospecimens and data, and transparency and accountability on the part of biobanks and their users”, as stated, among others, by the Australian National Health and Medical Research Council (3).
In most cases of biomaterial exchange among laboratories, researchers do not perform transfer agreements because of a general lack of knowledge and consistent scientific literature on this topic. In fact, whereas institutions are required to pursue harmonization and standardization as eligibility requirements or take part in international networks in the fields of biomedicine and biosciences, at the same time they face practical and theoretical issues when drafting an MTA due to the lack of harmonization among national regulations (4).
The International Society for Biological and Environmental Repositories (ISBER) developed guidelines for biorepositories that underline that “an MTA or similar agreement should be executed to document the obligations and responsibilities of parties involved in the transfer of materials from a repository prior to shipment. The agreement should be initiated as soon as possible, as additional time may be required for legal or regulatory approval prior to transfer” (5). Authoritative information is offered on international technology transfer portals such as Entente (http://entente-health.eu/) or the IPR Helpdesk (https://www.iprhelpdesk.eu/). These resources are undoubtedly useful for international support but are not specific to biosamples, and they require some prior knowledge in the technology transfer field in order to be used properly and be fully understood. Moreover, they provide institutional support templates but have been unable to define a common standard. The most authoritative template is perhaps the one developed by the US National Cancer Institute (NCI) specifically for biospecimen resources (6). The NCI template, however, does not mention one of the most critical issues of an MTA, that is, intellectual property (IP), whereas other sources do offer some specific definitions. This results in loss of traceability and loss of opportunity (both for researchers and biobanks), and it exposes the involved institutions and researchers to various risks. Unfortunately, the adoption of incorrect procedures could lead to irreparable damage in the future, e.g., concerning the ownership of intellectual property rights (IPR).
From this perspective it is important for biobanks and biomedical research centers to possess a standardized draft that could ideally be accepted as a ready-to-sign model agreement, which would facilitate researchers in addressing legal issues (7). Furthermore, it is necessary to set up a process that reduces long delays in the transfer (which are often due to a negotiating period to define the terms of agreement) and obtain the necessary signatures for MTAs from the legal representatives of institutions (8, 9).
Considering this framework, we have drawn up a ready-to-sign MTA for the CRO-Biobank (the biobank of the National Cancer Institute, CRO, Aviano) through the involvement of different institutional groups and professionals with multidisciplinary expertise, which may be suitable to researchers and institutions for different research purposes and collaborative projects. The additional goal of this paper is to outline all of an MTA's key clauses (Fig. 1) and to discuss the main issues and difficulties tackled during its creation, thus suggesting a model for other institutions and presenting an opportunity to develop the “culture of material sharing”.

Tag cloud generated using the text of our MTA. The numbers after the words and the choice of the font size show their recurrence in the text.
Methods
Our approach to drafting the CRO-Biobank MTA involved multiple resources and required the collaboration of different institutional groups and professionals with multidisciplinary expertise. The biobank staff interacted with the Scientific Directorate by cooperating with the Technology Transfer Office (TTO), consulted the General and Legal Affairs Office, and got involved with the Biobank Steering Committee and the legal representative of our institution (Fig. 2).
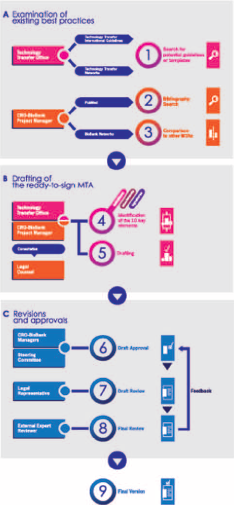
Flow chart showing the development process of our MTA. Our approach involved the collaboration of different institutional groups and multidisciplinary expertise professionals. (
Examination of Existing Best Practices
First, we performed a PubMed search for “Material Transfer Agreement” or “MTA” to get acquainted with the literature on the subject. Further information on existing biobank agreements and guidelines was gathered by the CRO-Biobank project manager, who interacted with biobank networks.
To gain a wider perspective, the TTO searched for non-biosample-specific guidelines and templates by exploring resources from networks and international organizations devoted to technology transfer such as the Alliance of Technology Transfer Professionals (ATTP) and the Association of University Technology Managers, as well as related initiatives such as the European IPR Helpdesk.
Drafting of the Ready-to-Sign MTA
All the information collected was instrumental in identifying the main conceptual sections to be included in an MTA and to structure them into a ready-to-sign document, taking also into account the advice on specific issues from legal professionals.
Revisions and Approvals
The final draft was presented and approved by the Steering Committee, which, according to its role, evaluates proposals concerning i) biomaterial requests from researchers; ii) operational and managerial issues; iii) scientific and legal ongoing compliance. The document was then submitted for review to the legal representative of our organization as the authorized person to execute the agreement. The MTA was subsequently sent to an external reviewer (an expert in the academic and corporate sector) with the necessary insight to ensure the document reflected the current needs of any private and public entities that might be interested in using biological materials.
Results
The multidisciplinary team identified the essential components to be included in an MTA, then drafted the MTA by dividing it into the following sections (see supplementary file 1, available online at www.biological-markers.com – Ready-to-sign MTA. This file shows the full version of the ready-to-sign MTA drafted by CRO-Biobank).
Definitions
The starting point of the teamwork for drafting the ready-to-sign agreement was the definition of “Biological Material”, as this is the core of the transfer itself and crucial to IPR controversies between parties.
For this reason we subdivided the concept of “Biological Material” into subgroups: i) “Original Biological Material”, which includes biobanked samples; ii) “Progeny”, which is Original Biological Material descendants (e.g. cell from cell); iii) “Unmodified Derivatives” such as subunits, purified or isolated components. To ensure that the use of the biological material and any substance created by its use would not lead to disputes between the provider and the recipient, we chose to add the more restrictive definition of “Modifications”. Any controversies will then be limited to those substances that are created by the recipients as a result of their research, which contain or incorporate the biological material (i.e., “Modifications”) (1). It is important to note that throughout the agreement, “Modifications” are not included when citing “Biological Material”.
This definition became pivotal to reducing potentially conflicting interpretations, which may have a significant impact, for instance, on the patentability of research results. Furthermore, this solution allows a double protection for both parties to the contract: in fact, the recipient's research team makes sure the provider will not claim ownership over the modifications since the former is able to claim title to any form of the biological material included in such modifications.
Even information related to the samples, such as clinical data, is treated and protected as material to be transferred.
Use of the Biological Material. Property and Rights
The main concept expressed in this paragraph is that the recipient accepts the liability pertaining to the custodianship of the original biological material, while the ownership remains with the provider. Custodianship is defined in the Definitions section as the “responsibility for safekeeping of biological material including associated information, control of its use, and eventual disposal in accordance with the terms and conditions of the agreement and the informed consent given by the participant”. Conversely, the provider does not claim ownership on modifications (see “Definitions”) made by the recipient.
Permissions
CRO-Biobank, as provider, make explicit its right to ask the recipient to provide the relevant project's approval by an ethics committee, in order to guarantee the respect of patients’ rights.
Custodianship
We commit ourselves to deliver samples in good condition as well as the appropriate related information and instructions for proper maintenance. From the moment of receipt, the researcher will be responsible for their use, storage and disposal under the terms and conditions of the agreement.
We decided to attach 2 forms (the empty informed consent form and the material transfer request form) as integral parts of the agreement because the provider, CRO-Biobank, is bound to respect and enforce them. Likewise, the receiver must abide by the same rules.
By signing the MTA, the recipient agrees not to transfer the biological material to any third party without written permission of the CRO-Biobank, otherwise there would be loss of custodianship control and appropriate compliance with the terms of the agreement.
Safety
Even when there are no issues concerning IP, most academic institutions use MTAs to perform all transfers given the other functions of these agreements (10), such as regulating several aspects regarding safety requirements, indemnification clauses, and transfer of biomaterials to third parties (11). In this section, we decided to make it explicit that samples may have hazardous properties, so the recipient must assume all liability to inform and train staff on its safe usage (12).
Liability and Representations
We included among the responsibilities of the provider, the collection and processing of samples according to all applicable laws, rules, and government regulations, including informed consent (which is attached to the agreement, see also the Custodianship section).
Moreover, the CRO-Biobank will ensure the protection of the patient's privacy. Samples are coded, so that under no circumstances the recipient will be supplied with the identity of the patient or be able to retrieve that information.
The provider and its employees and collaborators make no representations and assume no liability about any damages and claims in connection with the biological material, modifications or relevant information or their use by the recipient or its investigators.
Research Results
This clause states that if the research results in a discovery, invention, new use, or a product (collectively, “invention”), the recipient agrees to promptly disclose such invention to the provider on a confidential basis.
Moreover, to allow both parties to identify all potentially patentable inventions, the recipient also agrees to provide an annual written report including any results deriving from the use of the biological material until the expiration or termination of the agreement. This also allows the CRO-Biobank to ensure that the biological material be used under the terms of the agreement (i.e., complying with the aims enunciated in the informed consent, etc.). The CRO-Biobank cannot be held responsible, at any time, for the intentional misuse of the biological material by the recipient. Any educational institution party to the MTA has the right to use all inventions developed through the use of the biological material that is part of the agreement for its own internal research use, without the need to enter into license agreements or pay royalties.
Publications. Acknowledgment of Contribution
A key issue is often the unavoidable trade-off between appropriate confidentiality protection and researchers’ commitment to publish academic articles or make presentations. Starting from this consideration, our MTA states that it is not intended to prevent or unreasonably delay the publication of research results. In this context, our MTA also provides a mechanism of mutual protection for the provider and the recipient: prior to publication, the researcher has a minimum of 30 days prior to sending a copy of the information to be disseminated, to give the provider the opportunity to protect the outcome of the research results through any of the available and most suitable IPRs. Moreover, since researchers must try not to compromise the possibility for their affiliated institutions to profit from tech transfer operations, this additional check for patent eligibility by the provider's TTO represents another advantage for the recipient, as it may lead to the identification of recipients’ protectable rights, especially concerning the modifications (see section on “Definitions”).
Negotiated benefits shoud include information sharing, royalties, acknowledgment of the provider as the source of the biospecimens, publications, and transfer of technology or materials (13). We require the recipient to indicate the CRO-Biobank as the source of the biological material in all publications and presentations in the Methods section, according to the Bioresource Research Impact Factor's (BRIF) suggestion adopted by the European Association of Science Editors (14).
Confidentiality and Non-Disclosure
Similar to other common agreements, we ask the recipient to make reasonable efforts to keep confidential and not disclose to any third party any confidential information. We consider exceptions to the general principle information that was known to the recipient before receipt from the provider or that has become publicly known without any fault of the recipient. Other cases in which we envisioned an exception to the general rule are those in which information is obtained by the recipient from a third party in circumstances where the recipient has no reason to believe there has been a breach of a confidentiality agreement. Moreover, further exceptions apply if the recipient can establish by reasonable proof that information was substantially and independently developed by its affiliates or when the information must be disclosed following a court order or other judicial procedure. We ask confidentiality to be maintained for 5 years from the date of termination or expiration of the MTA.
Cost Recovery
The CRO-Biobank does not sell biological material; it only requests the reimbursement of transportation costs and a nominal fee covering storage and processing costs.
Term and Termination
It is recommended to set the date of expiration of the agreement and include the possibilities under which it can be terminated. We list 3 possibilities: i) a number of years from the date of signing the agreement (to be determined by mutual agreement); ii) upon completion of research with the biological material; iii) by 30 days’ written notice by either party to the other.
Upon expiration or termination of the agreement, the recipient should discontinue the use of the biological material and should return the original samples to the CRO-Biobank as the provider, which will destroy them. Such material will by no means be transferred to any other researchers because we cannot ensure it has been properly handled by the previous recipient.
Even if the agreement is terminated by one of the parties or expires, we specify that the parties still remain bound by some of its terms for an additional period of 5 years.
Signatures
In most cases MTAs are signed and executed by institutions rather than individual researchers (15). Our MTA must be dated and signed by a duly authorized representative of each party as well as the recipient's principal investigator. On the one hand, the additional signatures of the scientific staff are a valuable mechanism to remind them of the obligations they are agreeing to comply with; we believe this could enhance the researchers’ sense of responsibility when using the biomaterial. On the other hand, the existence of institutional policies authorizing specific individuals to execute a standard format MTA could be of great help in avoiding delays caused by unnecessary reviews and further procedures to obtain additional signatures.
Pilot Study
This template has been used by the CRO-Biobank for a pilot study (16). At present, 7 agreements have been signed. Neither the provider nor the recipient complained about the MTA template, and no delays occurred. However, more time is needed to evaluate the template's efficacy concerning IP issues and also concerning the development of a progeny or other time-related factors.
Discussion
The MTA is intended to facilitate the noncommercial transfer of materials and information between organizations (11). The current methods for drafting MTA terms and conditions are prone to delays and, in the worst-case scenario, may even prevent the parties from executing an agreement, thus generating a waste of resources and, in some instances, favoring the abandonment of a specific research project that relied on the requested materials (8, 17). All these obstacles and missed opportunities have the consequence of hampering the medical research system at large. The scarcity of regulations and homogeneous guidelines does not help to overcome these issues.
Transnational research activity involving institutes that manage human biosamples, in particular tissues, has been increasing rapidly over the last years, either kindled by direct collaborations or promoted by networks and funding programs. Existing MTA templates may, however, not address specific legal and ethical concerns of certain nations or even an entire continent, thus requiring further adaption of important concepts like informed consent, benefit-sharing arrangements, ownership, and IPRs (13).
With the increase in international transfers of human biospecimens and data, concerns are being raised about how donor wishes are protected in such circumstances. Our MTA acquires a new role that includes the respect of donor wishes under contractual terms. In fact, we attach to the MTA the CRO-Biobank empty informed consent form (see supplementary file 2, available online at www.biological-markers.com> – CRO-Biobank informed consent form), which is part of a pathway set up to enhance the awareness and understanding of patients’ participation (18), so that the researcher is bound to use the biospecimens in accordance with ethical guidelines. For example, samples collected by the CRO-Biobank must not be used in research studies unrelated to cancer, since this is not permitted by the informed consent form, i.e., by the patient. For the same reason, the next step could be the involvement of a research ethics committee in ensuring legal and ethical conduct, as recommended by Chalmers and colleagues (15).
It should also be noted that gathering a critical mass of patients/biological materials across national boundaries is an impact requirement for supported multicenter projects, even in the absence of comprehensive coverage of any concept relevant to the management of human tissues. In fact, networks aimed at opening research infrastructures (e.g., the European Research Area NETworks, ERA-NETs,) may involve ministries and executive agencies directly deputed to promote transnational applications in a broad range of topics from a variety of stakeholders, as human health and biobanks are mere parts of wider schemes. In this context, the promotion of MTA templates and subsequent adaptions help secure the added value of transnational research in biology. As an example, only minimum consortium agreement requirements including clarification of potential IPR matters for partnering institutions from the European Union, Latin America and the Caribbean countries (LAC) are set under the currently open Second LAC-ERANet Joint Call, Topic 8 (http://eranet-lac.eu/assets/moxiemanager/280116_GuideForApplicants_incPScreenshots.pdf), whereas any other information required by national regulations has to be made available by project partners upon request to the national agencies.
Several efforts have been made to streamline the process (8, 19) utilizing different approaches such as the use of a standardized language (20); however, none of these strategies has been widely adopted.
As a result of the analysis of MTA best practices and after careful comparison to specific sections from various types of agreements regulating knowledge and IP transfer in biotechnology, the CRO-Biobank was not in a position to readopt definitions and related IP clauses as suggested by other institutions. Support templates deal with different aspects of the MTA and are deficient in some. We consider the MTA reported in the NCI best practices for biorepositories (6) worthy of further implementation because of the importance of clearly defined original biological material, progeny and other biomaterial definitions that we set in our template, thus clearly stating any subsequent rights and obligations. In fact, we realized that a detailed IP framework is highly recommended, particularly when mid- to long-term planned research activities may imply complex biological events and ever-changing relationships among the teams.
Laws and regulations concerning IPRs on biospecimens are not well defined or are different among countries. The creation of commercial products from human samples has generated complex legal and ethical questions that have no commonly accepted answers. International case law suggests that, when these issues are dealt with, each situation must be determined individually (13), and strong efforts are required to adapt the tradition and precedent of the law to the innovations of the biotechnology era (21). MTAs could help in defining at least part of these questions before they arise, that is, before the research with biospecimens takes place.
A major criticism of MTAs is that their terms may expand the rights of the parties beyond those granted under formal IP laws (22). First, MTAs may regulate the use of biospecimens that would usually not be eligible for IP protection or where IP has expired (23). Second, MTAs may set limits to use in countries where the inventors/owners have not sought or been granted patent protection. Third, the terms of MTAs may assign rights over patented inventions that go far beyond those contemplated by patent policy (23). Such reach through rights is, however, discouraged by most guidelines and policies for best practices in licensing (24, 25).
One of the aims of this work is to help researchers understand the basic concepts contained in most of the agreements. Notably, the template we conceived might represent a useful tool for releasing biological materials (MTA-out) and can be adopted by researchers and biobanks or other research organizations involved in the storage and sharing of biosamples. Instead MTAs offered by the recipients (MTA-in) are on the whole unusual, as it is mainly in the interest of the provider to have a document regulating the use of the biological material and to have the recipient accept terms that are standardized by the provider.
Our solution relies on approaching the drafting of a ready-to-sign MTA through the involvement of combined expertise at distinct levels. This becomes even more important in research institutes, whose translational character pursued through multidisciplinarity is emphasized. In our case, it was to our advantage that our biobanking activities were already institutionalized within the Scientific Directorate, and that even the biobank project manager was in close contact with other staff, such as the TTO. The availability of a group or committee that is already accustomed to combining expertise with the planning required to manage a biobank has been of great help in creating procedures that are efficiency-driven, and in drafting documents such as the MTA.
The solution we propose is intended to be a fundamental resource for institutions aimed at fostering collaborative projects typically driven by the exchange of biomaterials, and particularly for biobanks whose main goal is the delivery of samples.
Moreover, we developed our template satisfying all ISBER guidelines and recommendations for biorepository MTAs (5). Altogether, our MTA model and its clauses are based on easily acceptable standards but certainly do not provide a solution to all cases in which IPRs play a major role. In any case, every biomedical institution, especially if engaged in translational research and thereby oriented towards technology transfer, should be ordinarily bound to manage inventive processes from the signing of the disclosure forms. Considering that this is an unavoidable step, our solution proved to allow the rapid transfer of biomaterials, limiting the chances of potential disputes, if any, to those cases in which the recipient needs to make a disclosure. Another possible limitation towards the adoption of a universal MTA template relates to the unavoidable differences among different national legislations.
Conclusions
This paper aims to be an unabridged study among the few works in the existing literature providing a description of the whole process related to the formation of an MTA, and also to present the most complete ready-to-sign template. Because of a general lack of knowledge about these kinds of agreements, researchers often do not use MTAs, and institutions underestimate the importance of executing them. The availability of a ready-to-sign template may thus increase the adoption of these tools and procedures, and deepen researchers’ awareness of the implications related to material sharing.
At the same time, both parties (the biobank provider and the researcher recipient) may take advantage of utilizing an MTA to manage their liability and ensure appropriate use or distribution of materials, while avoiding risks such as a loss of opportunity (e.g., concerning the ownership of IPRs). Moreover, the number of executed MTAs is also increasingly being used as an indicator of scientific productivity to obtain government funds.
Despite the crucial role of MTAs in increasing the number of opportunities offered by geographically distributed research, and the fact that institutions are constantly required to meet harmonization and standardization requirements, apparently there is no consistent literature on this subject. Therefore, the present work offers readers an important opportunity to increase their understanding of the issues surrounding the preparation of an MTA. Even if this template proved its efficacy in reducing delays, further time is needed to evaluate its worth concerning IP issues.
Even if our template is not adopted unconditionally, biobanks and institutions involved in the drafting of an MTA may still benefit from our multidisciplinary experience and solutions to practical and theoretical issues around the creation of their own MTAs.
Footnotes
Acknowledgment
We thank all professionals who contributed to the drafting of the CRO-Biobank MTA. We also thank Riccardo Bianchet for graphics support and Ms Anna Vallerugo, MA, for the English editing.
Abbreviations
Financial support: None.
Conflict of interest: The authors declare that they have no competing interests.
