Abstract
This report describes the organization of the Biobank of the CRO Aviano National Cancer Institute, Aviano (CRO- Biobank), Italy, implemented as a structured facility dedicated to collecting human biological samples. It describes a particular disease-specific biobank and the integration of a research biobank in a clinical setting. The CRO-Biobank's mission is rooted in supporting and implementing cancer research, with its main focus on optimizing technical and quality processes, while also investigating ethical, legal and IT topics.
The CRO-Biobank has implemented processes aimed at guaranteeing the safety of the providers, protecting patient privacy and ensuring both the traceability and quality of its samples. Our 8 years of experience allow us to offer insights and useful suggestions that may solve theoretical and practical issues that can arise when starting up new biobanks or developing existing biobanks further.
Introduction
The accessibility of homogeneous and well-characterized biological samples is a prerequisite for valuable research. Therefore, biobanks need to organize large specimen and data collections to overcome limitations due to inadequate availability of quality samples and lack of information (1).
Different types of biobanks are being developed worldwide (e.g., population banks, disease-oriented biobanks) which differ in sample types (e.g., serum, urine, blood cord, tissues, cells, saliva) and source (e.g., human, animal, microorganisms). Irrespective of the type of biobank, it is essential to develop a management strategy prior to the start of sample collection, in order to avoid the risk of collecting inadequate samples.
As a matter of fact, biobanking involves more than a mere collection of samples: it requires respect for ethical and legal regulations (privacy in particular) and international guidelines, along with a design enabling traceability, as well as rooms and facilities, long-term economic plans, standard operating procedures, quality controls, sample information recovery (e.g., biological, clinical and personal data) and appropriate IT. Most of these fundamental items are overlooked in the literature, resulting in open questions and unsolved problems (2–4).
This report is an overview of the organization of the Biobank of the CRO Aviano National Cancer Institute, Aviano, Italy (CRO-Biobank), focusing on the main topics mentioned above. It describes a particular disease-specific biobank and the integration of a research biobank in a clinical setting. Based on our 8-years experience, we propose suggestions, ideas and practical solutions that may be useful to those who are starting up new biobanks, and for those who wish to further develop existing biobanks.
Mission
CRO-Biobank is an institutional cancer biobank established in 2006 with the aim of collecting and preserving human biological samples, such as tissues, fluids and blood derivatives, for research purposes in cancer (or related fields). Samples are obtained from both healthy subjects and patients histologically diagnosed with cancer or precancerous lesions. The CRO-Biobank functions as a lasting source of human biological samples, collected at diagnosis and at subsequent therapeutical stages, with records of associated sociodemographic, clinicopathological, biological and lifestyle information. The statistically significant number of samples and their corresponding data available from the CRO-Biobank is an invaluable resource for research on cancer prevention, diagnosis and treatment. Particular focus is placed on identifying factors (e.g., biomarkers) that may predispose to cancer, studying the efficacy of new therapies and vaccines, and discovering new pharmacological targets (5). When put into practice, human tissue sampling is particularly important to fulfill the promise of personalized medicine derived from next-generation sequencing advances (6) or disease- and patient-specific induced pluripotent stem cells (7).
Organizational Overview
The Biobank team collaborates with the whole institutional staff (physicians, nurses, technicians, IT expert, secretaries, etc.) (Fig. 1). This organizational system optimizes the cost to benefit ratio, since it exploits the resources and capabilities of already available personnel. At the same time, it guarantees high-quality standards, as it is maintained by dedicated personnel specifically trained in biobanking.
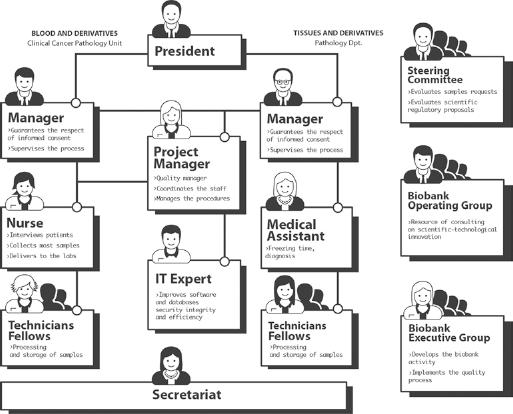
Organizational chart of the CRO-Biobank showing the main functions of the staff.
The CRO-Biobank president is the CRO's scientific director. Two Biobank managers (a pathologist and a clinical pathologist) supervise the entire process of collecting and storing samples, and ensure that informed consent is respected, according to the Ministerial Biobank Guidelines (8). The Biobank project manager (employed full-time) is responsible for quality control (as quality manager [QM]), implementing proper procedures and coordinating the Biobank staff. The CRO-Biobank full-time staff consists of a biologist, a nurse, a lab technician and a fellow. The institutional personnel who are involved part-time in biobanking activities comprise a pathologist in charge of assuring the standardization of the tissue sample collection, an IT expert and a scientific directorate secretary. Other members of the institutional staff (surgeons, pathologists, medical oncologists) contribute by providing biological samples, performing histological diagnoses or providing clinical data, maintaining their specific research activities, while daily operational activities of the Biobank are conducted by specifically dedicated personnel.
A wide variety of samples are collected at multiple clinical phases: (a) at different therapeutical stages: at diagnosis, before/after treatment (surgery, chemotherapy, hormone therapy and radiotherapy); (b) at different sites at the same time: from primary tumors and metastases, (c) at different times – for instance, at first, second and subsequent relapses. The collection is thus representative of the whole “biological history” of the disease and is useful in a large variety of studies (e.g., comparative studies on metastasis onset).
Once collected, the biological materials are processed in the laboratories of the Clinical Pathology Unit and the Pathology Department, then rapidly stored in the Biobank storage room.
The CRO-Biobank has 3 working groups: the steering committee (SC), the operative group (OG) and the executive group (EG). The SC is composed of researchers from CRO departments involved in sample collection and research programs. The committee meets monthly to evaluate sample requests submitted by investigators. The mission of the OG, formed by experts in different disciplines such as molecular biology, pathology, hematology, clinical oncology, microbiology, surgery and forensic medicine, is to promote continuous updating of all of the scientific and technological aspects of biobanking at the CRO-Biobank. The EG, which comprises the biobank managers, the project manager and QM, the IT expert and the laboratory technicians’ coordinators of the clinical laboratories where samples are handled, serves to update technical procedures to maintain high quality standards in all CRO-Biobank activities.
Biobanking also has to consider the important concepts of accountability and financial considerations: operating costs are periodically reviewed by the managers and the president to allocate pertinent resources and ensure economical sustainability (see also the section “Sustainability considerations”).
Ethical and Legal Framework
Patients
The CRO-Biobank acts in compliance with ethical and privacy regulations. Its organization is meant to reduce the discomfort of the patient: e.g., to avoid whenever possible additional venipuncture blood samples being taken at the same time as the routine blood examination.
Human biological material and associated genetic data are treated in ways that ensure respect for the dignity, rights and personal freedoms of the donors, who sign a detailed informed consent for the storage and further use of their biological material (9).
As required by most governmental laws, enrolled patients must be informed by a physician and give written consent before the collection of samples for research (9, 10). A first version of the informed consent policy was approved by the CRO ethics committee in November 2007. The original consent form contained relevant information describing the purposes of the CRO-Biobank, and clearly illustrated the implications, advantages, drawbacks and risks of donating samples to the Biobank. It also included statements on donors’ rights to be informed about their sample use, and their ability to withdraw their consent at any time, followed by the destruction of their remaining samples. The patients are not given any compensation or other benefits for their donation of biological materials. The form was subsequently revised:
We developed a single form to be used at different therapeutic stages for multiple collections; this avoids having a physician available at each collection time to sign the forms, which can be difficult to handle in clinical practice, often leading to noncollection of numerous samples;
We also added a phone number for patients who need additional information;
We gave the patients the possibility to know the return of results (i.e. to know the results of the researches performed using their samples) by contacting the Biobank at the same phone number; their call is then forwarded to the project manager of the Biobank. This enables us to avoid needing to call each patient every time a study based on their samples produces some result. We can thus save time and trouble (e.g., if a patient changes phone number) for the Biobank staff and discomfort for patients (a call from the cancer center where they have been receiving treatment may initially alarm them and then remind them of their illness). Our records show that only about 1 patient out of 300 (1:285) has phoned to us to obtain results. Considering that each call takes an average of 15 minutes, this has allowed our organization to save many working hours;
Based on our previous experience (e.g., compilation errors, patients’ and operators’ feedback), we simplified the language and the form-filling procedures to retain the completeness and accuracy of the information on a single page. We assessed the vocabulary using the EULOGOS Censor (an online tool which measures the readability and usage of basic vocabulary of the Italian language) and evaluating the comprehension of a group of volunteer patients. To ensure that conscious decisions can be made, we implemented an easy-to-apply multisource informed consent procedure allowing a high rate of consensus, as more than 95% of patients understand and comply with it (11).
While the possibility of calling us and getting information is straightforward for the expected results of a project, problems can arise with unexpected results (incidental findings) when they may be of clinical relevance for the donors or their families. This situation is currently more frequent than in the past due to biotechnological development – for instance, with a Genome-Wide Association Study (GWAS) or whole genome sequencing (e.g., if an unexpected germline BRAC1 mutation appeared in a GWAS analysis). This topic is currently controversial and represents a hot issue under discussion. Current ethical standards, EU directives and some domestic laws indicate that it is the Biobank that is in charge of the active information, unless the donors have signed a “right not to know“ declaration. In this undefined scenario, we are currently evaluating whether to ask researchers to promptly inform the Biobank when an incidental finding of clinical relevance occurs, and whether to include a clause to this effect in the Material Transfer Agreement (MTA). CRO-Biobank would then send the report to the patient's physician, who could explain to the patient the possible implications related to the findings; alternatively, the patients and their families could prefer to receive the report directly. These multiple options (including the right not to know) will be listed on the upcoming informed consent form.
Researchers
Researchers who request samples or data are required to indicate the reference of the approval of the project by their ethics committee (if any) or of their institutional review board (IRB).
Incidental findings are managed like all other findings. The CRO-Biobank MTA states that whenever the investigation results in a discovery, invention, new use or product, the researcher agrees to promptly disclose such information on a confidential basis. Moreover, to allow both parties to identify all potentially patentable inventions, the recipient also agrees to provide a yearly written report including any results deriving from the use of the biological material until the expiration or termination of the agreement. This also allows the CRO-Biobank to ensure that the biological material is used under the terms of the agreement (i.e., complying with the aims stated in the informed consent, etc.).
Process Overview
Patients are recruited by physicians, either at the time of diagnosis or upon notification by the Biobank team based on hospitalization lists (Fig. 2). The Institutional IT system is organized to prepare labels for all biological samples, both for routine examinations and for Biobank collection tubes.
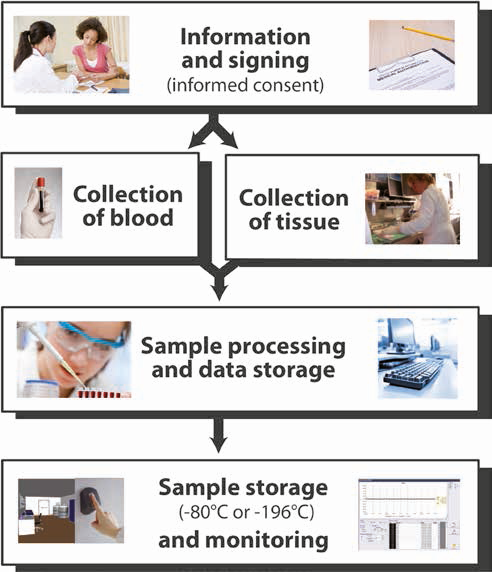
Biobanking workflow overview. On the left: blood and derivatives samples; on the right: tissue samples.
Each patient's signed informed consent is checked both before sample collection and at the time of delivery of samples to the labs. Immediately after collection, whole blood tubes are kept at +4°C and then are transported to the clinical pathology lab in special bags containing ice packs. The tubes are centrifuged at room temperature for 10 minutes at 2,608g, then serum, plasma and buffy coat are transferred into 2-dimensional (2D) barcoded tubes and then stored at -80°C (within 3 hours of collection).
Tissue specimens are immediately delivered to the pathology lab after collection, then examined and sampled by the pathologist. Samples are stored in tubes or in Cryomolds. Frozen tissue samples are embedded in OCT compound and snap frozen in liquid nitrogen. Moreover, all formalin-fixed paraffin-embedded (FFPE) tissues archived in the CRO Pathology Division may be available. Any requests by researchers for FFPE samples, when approved, are passed to the pathology division for evaluation of the feasibility of tissue sample retrieval.
Blood and tissue samples are stored at -80°C in 2D barcoded tubes within 3 hours and 30 minutes, respectively, to prevent degradation of biospecimen components, while viable cells are stored at -196°C. Any nonconformity to procedures, such as hemolysis, freezing delay, etc., are registered in the database as required by the quality management system.
To completely characterize the samples, clinical data are obtained from medical charts and patient interviews performed by the Biobank nurse using a questionnaire.
Toward Harmonization
At presently biobanked samples in different biorepositories are subject to a number of different preanalytical steps (concerning collection, processing, storage) which influence experimental outcomes and the ability to reproduce scientific results. In addition, this issue prevents researchers from asking for samples from different biobanks (for the same study) because of their inhomogeneity. For these reasons, many efforts are being made toward harmonization of biobanking procedures and sample data sets (for information about CRO-Biobank procedures and guidelines see the “Quality aspects” section). In the absence of the adoption of unique guidelines, it is critical that the information regarding biospecimens be reported in a thorough, accurate and standardized manner. For these reasons, the US National Institutes of Health (NIH) have developed the Biospecimen Reporting for Improved Study Quality (BRISQ) standards, which are often required for publication (12). BRISQ recommendations consist of a list of data elements which includes general information for consistent documentation of classes of biospecimens (solid tissues and body fluids) and factors that may influence integrity, quality and molecular composition of samples. Reporting the details enumerated in this list does not fully guarantee the quality of samples and should not represent a substitute for empirical quality evaluations, but it supplies researchers with more consistent and standardized information to better evaluate, interpret, compare and reproduce the experimental results (12).
The clinical data set used to characterize CRO-Biobank samples adheres to the BRISQ standards. Although these requirements were published in 2011 (12), we were able to classify all biobanked samples (including those collected prior to that date); in fact when we planned the Biobank, we carefully took into account the need to strictly characterize the samples.
Quality Aspects
Both the facilities and instruments of the CRO-Biobank are certified (UNI EN ISO 9001) or accredited (Accreditation Canada) by the current regulations for safety and quality, as required by our regional government. Standard operating procedures and protocols for collection, processing and storage of samples and data are optimized according to the best practice guidelines of the International Society for Biological and Environmental Repositories (ISBER) (13) and the Organisation for Economic Co-operation and Development (OECD) best practices (14) in order to guarantee homogeneity and long-term stability of samples. In this context, respect for timing, preservation of cold-chain from sample collection to storage, optimal tissue freezing, prevention of hot and cold tissue ischemia and accuracy in the OCT embedding process, are all essential and mandatory technical issues and represent a prerequisite for the accuracy and reproducibility of the subsequent research analyses
Well-defined tissue control methods are used: histocytology (frozen sections, FFPE-mirrored samples and cytological imprint) and molecular pathology. Sections obtained from the frozen sample at -20°C represent the gold standard for morphological control. However this procedure leads to a consumption of the material, so it may be problematic in cases of small samples. Mirrored FFPE sections of tissue adjacent to the frozen sample give a good representation of the morphology, but they do not truly correspond to the frozen tissue sample. Cytological imprint is a cost-effective rapid method that assures the preservation of the frozen tissue. Molecular analyses are randomly performed for DNA, RNA and protein quality checks, using appropriate positive and negative controls.
Tumor or healthy frozen tissues are also evaluated to establish the percentage of necrotic/fibrotic areas using histological sections. Many cryostatic sections can be obtained from embedded frozen tissue for different research analyses and projects. The process of aliquoting our stored material has been checked for quality and consistency of biomolecular analyses in tissues sections with optimal results (unpublished observations).
Importantly, the Biobank storage room is equipped with 2 independent alarm devices (mutual backup): one of them is a cabled system alerting the receptionist of the institute (EY3600; Sauter), and the other one is a computer system alerting the managers by e-mail (Spylog Plus; AHSI). The Sauter EY3600 is an integrated control system of technological structures, specifically concerning the climatization of the entire institute (room and freezer temperatures, humidity, air conditioning etc.). It remotely connects all alarms to a centralized point manned 24 hours a day, which manages all alarms in the Institute and notifies the personnel on call. For a description of the additional Biobank alarm system see also the section “IT systems and privacy solutions.”
IT systems and Privacy Solutions
The CRO-Biobank processes are designed to ensure the traceability and safety of samples both on paper (in key-protected archives) and on electronic devices (password-protected double-coded databases).
Traceability is guaranteed by the Matrix system based on password-protected software (Easytrack 2D ver. 2.0; Twinhelix) for the management of sample data, including their physical location. Each sample is uniquely identified using tubes marked with a 2D code which is associated with a range of information on the sample, including clinical and storage data. This strategy allows the Biobank to protect the privacy of the data, as the system encodes any sensitive data of the patient (according to the ISBER's best practice guidelines for biobanking) (12). Furthermore, to maximize the privacy protection, sensitive data are collected on a different database, hosted and managed by a certified company on a server farm (2). A backup of the databases is performed daily.
A temperature-monitoring software system (Spylog Plus) allows the Biobank to control and trace all daily recorded freezer temperatures (sampled every 15 minutes) over years. It is based on designated temperature probes which communicate with a PC through wireless transmitters–receivers. It is programmed to activate audible alarms and automatically send e-mail alerts to the managers in case of emergency. Audible alarms and pop-ups appear on all computers of the Institute on which the software is installed, not only in the storage room (where operators are not present all of the time). The alert system is also activated in case of breakdown (e.g., offline transmitter–receiver).
Sample Delivery
A first pilot project for a sample delivery system was set up in 2012 and proved to work appropriately. At present, internal and external researchers can request samples and data by completing a request form. These requests are then monthly evaluated by the Biobank steering committee, whose decisions are based on the scientific value and feasibility of the submitted projects, the absence of overlapping research areas/goals, and adherence to rules and regulations (mainly as it pertains to patients’ informed consent). Upon obtaining approval from the committee, applicants must sign a specific MTA (Fig. 3).
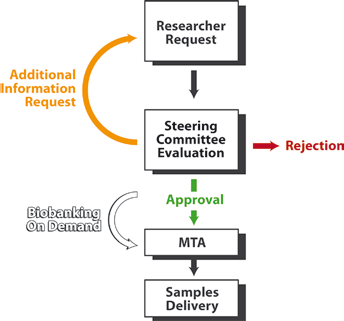
Flowchart of transfers from biobanked samples: from the request to the delivery. MTA = Material Transfer Agreement.
Before the delivery of tumor tissues, a pathologist checks the quality of each single sample by hematoxylin and eosin staining.
An accurate protocol for shipping the biological material has also been developed to ensure the safety and reliability of shipped samples. The scientific contribution derived from the use of Biobanked samples is constantly monitored: researchers are required to send an annual report and are asked to quote the source of the biospecimens in all scientific publications.
Facilities
The following is a list of the facilities in the CRO-Biobank.
Rooms
surgical room (for tissue sample and derivatives preparation);
lab room (for blood sample and derivatives preparation);
dedicated storage room (more than 100 m2);
blood collection/interview room.
Instruments
biological safety cabinets;
lab centrifuges;
cryostat microtome;
fast temperature abatement system for tissue samples;
N°4 -80°C horizontal freezers (including a backup freezer). Each freezer can contain up to 60,000 blood aliquots (stored in 500-μL vials, 96-sample boxes);
N°4 -80°C vertical freezers (including 3 backup freezer) for tissue sample preservation;
N°3 liquid nitrogen freezing systems (including 1 backup system) for viable cell preservation.
Safety Systems
Fire alarm systems in each room;
Oxygen depletion alarm systems and emergency aid systems;
Wireless freezer temperature monitoring systems (to safeguard sample quality) with remote alarms (audible alarms, manager e-mail alerts and technical service warnings);
Storage room temperature monitoring system (to allow the proper functioning of freezers);
Fingerprint identification system (to protect access to the storage room).
Sustainability Considerations
The institute (CRO) where the CRO-Biobank is located receives an annual grant from the Italian Ministry of Health for cancer research implementation. There are also institutional funds to be allocated in case of necessity, to guarantee the continuity of the Biobank. Moreover, the system is supported by the financial contributions of individual CRO researchers who are beneficiaries of research funds for projects involving banked biological material. (Researchers can include this item as “Biobanking costs” when submitting a grant application.) CRO-Biobank does not sell biological material; it only requests the reimbursement of transportation costs and the payment of a nominal fee for covering storage and processing costs. We require no additional fee for services, and we do not discriminate between intramural and extramural researchers; only in case of duplicate projects do we give priority to intramural requests.
The Scientific Directorate encourages intramural researchers to benefit from the CRO-Biobank for sample collection. If all institutional projects become part of the Biobank, there are no cumulative costs for each individual structure, researcher or project for sample retrieval, management and storage. Researchers can thus easily obtain high-quality homogeneous samples without using their own resources (i.e., time, staff, sample retrieval organization, management, patient enrollment etc.) and at the same time support the Biobank.
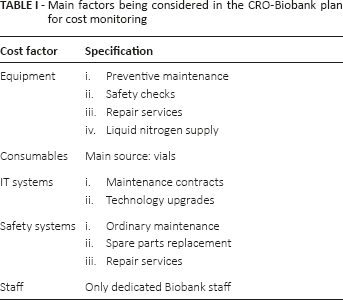
We developed a plan to monitor in real time the costs of the system and the continuity of Biobank activities. We also created an institutional database for research (DBR) to report and manage all funding obtained from institutional grants. Recently we implemented a section dedicated to Biobank cost recovery, so we will be able to evaluate the financial impact of the recovery over time. The main factors being considered in this plan are summarized in Table I.
Main factors being considered in the CRO-Biobank plan for cost monitoring
Network Participation
The CRO-Biobank is a member of the Italian Network of Cancer Biobanks (RIBBO). It is also participating in the setting up of a European network as a founding member of the Biobanking and Biomolecular Resources Research Infrastructure – European Research Infrastructure Consortium (BBMRI-ERIC). BBMRI-ERIC shall establish, operate and develop a pan-European distributed research infrastructure of biobanks and biomolecular resources to increase efficacy and excellence of biomedical research by facilitating access to quality-defined human health/disease-relevant biological resources through:
The inclusion of associated data in an efficient and ethically and legally compliant manner;
Reducing the fragmentation of the biomedical research landscape through harmonization of procedures, implementation of common standards and fostering of high-level collaboration;
Capacity building in countries with less developed biobanking communities, thereby contributing to Europe“s cohesion policy.
Results
The collection of samples started on November 2007 (pilot study) after more than 1 year of planning, and following the approval of the CRO ethics committee. We recruit more than 1,000 patients with cancer and other related pathologies and about 300 healthy individuals per year. The sample collection now comprises more than 20,000 samples (>60,000 aliquots), including serum, plasma (ethylenediaminetetraacetic acid [EDTA] or citrate), buffy coat and frozen tissue (OCT embedded). Moreover, all FFPE tissues archived in the CRO Pathology Division may be available for study. More than 6,000 samples (>15,000 aliquots), half of which were affected by gynecological pathologies, have been obtained from patients before surgery. We have also included about 200 patients whose blood was collected at different times after autologous stem cell transplantation.
In our experience, none of the donors have withdrawn their consent, and no related legal issues have occurred in the 8-years period of biobanking activity.
During a pilot study aimed at setting up the sample delivery protocols, the steering committee was convened 5 times and received 16 sample requests from researchers, 13 of which were accepted, 2 rejected and 1 partially accepted (a first quota of samples was assigned, while the remaining were to be decided upon presentation of preliminary results). Based on specific researchers’ requests, we also started 3 sample collections “on demand”, meaning we started to collect new types of samples, reserving an aliquot for the researcher (and enriching the Biobank collection with the remaining aliquots). At present, the procedural path has been consolidated, and the steering committee meets monthly.
Discussion
When planning to start a biobank, it is crucial to spend time conceiving its management prior to beginning the collection of the samples (15). This is a real investment to prevent unrecoverable errors that could compromise the future use of the samples (see “Toward harmonization” section). Biobanks act as guarantors for stakeholders on 3 levels:
for researchers (assuring quality of biospecimens and related information);
for patients (protecting privacy);
for the operators/staff (ensuring safety).
First, it is fundamental to decide (i) what types and quantities of biological material are to be stored, (ii) for which purpose, (iii) with which procedures must they be treated and (iv) under what conditions the materials are to be stored (15).
To answer these and similar issues, the CRO-Biobank was supported by 3 groups (SC, OG and EG) to benefit from the contributions, opinions and experiences of experts in different fields. Since the CRO-Biobank is located within a cancer institute, the consented use of the different samples is for research only in the field of cancer or other related topics. Compliance with this principle is guaranteed by the SC, which also acts as a guarantor of impartial access to the samples by researchers and ensures accuracy in the evaluation of the scientific feasibility and reliability of research projects.
The different professional backgrounds of the OG members initially allowed the identification of the most suitable specimen types to be collected to meet the different experimental/researcher needs. Based on their suggestions (and in relation to cost analysis), we initially decided to collect:
plasma obtained with 2 different coagulants to avoid the possibility that one of them may interfere with later analysis;
serum;
buffy coat to facilitate subsequent nucleic acid extraction (blood is much less stable);
frozen tissues.
Preparation and storage methods to ensure long-term stability of samples were established with the support of the EG. These procedures were chosen on the basis of the experience of the clinical cancer pathology and the pathology labs, and by following international guidelines (13, 14). CRO was one of the lead participants in the TubaFrost European Project, for the virtual sharing of tissue collections mainly in the field of pathological oncology. This opportunity gave us particular expertise, in the current pathologist's role in the organization of the CRO-Biobank. From the beginning, all procedures were implemented in compliance with quality standards (we benefited from having available certified/accredited labs and equipment), since the quality of samples and related information is a prerequisite to reduce experimental bias and to improve the authentication of research results. We decided to start the collection (and to implement any subsequent phase of the process) using pilot studies to adjust the organization step by step and consequently minimize the effect of possible errors.
Quality standards also require implementation of traceability and privacy protection systems. Deidentification and anonymization are strategies used to remove patient identifiers in electronic health record data. The use of these strategies is of primary importance, given the need to share electronic health record data across multiple institutions while safeguarding patient privacy (16). For these reasons, all CRO-Biobank samples are coded, and to respect patients’ privacy, only authorized Biobank staff can associate personal registry data for the samples. We decided to use 2D barcode systems and tubes to also minimize handwriting errors.
The safety protection of the operators represents the third aspect that has to be guaranteed by a biobank. In addition to the usual preventive measures nowadays implemented in all laboratories, particular attention was paid to the storage room. We settled oxygen depletion alarm systems and emergency aid systems to prevent asphyxiation due to the presence of liquid nitrogen vapors. We planned the acquisition of a closed-circuit camera and of a fall detector. This latter device must be tied to the belt before entering the room, and in case of sickness (e.g., fainting), when the person is at ground level, it triggers an alarm.
The CRO-Biobank organization led to a better rationalization of resources. The interfacing between specialized Biobank staff and other human resources working in the Institute, led to amortized costs while assuring high quality of samples. Since the CRO is an IRCCS, part of the institutional staff activity has to be dedicated to research. This has several advantages: (i) increased number of collected samples (e.g., patients’ enrollment requests by physicians are higher; better cooperation with administrative staff); (ii) quality of samples (e.g., the respecting of timing and conditions from the collection site to lab delivery, especially thanks to the cooperation of institutional nurses and technicians); (iii) quality of information (e.g., the contribution of physicians to clinical information updating); and (iv) cost savings (i.e., human resources mentioned in the previous points).
Our slogan “No waste time, just research” has a double meaning. First, with the support of biobanks, researchers do not spend time and resources (including financial resources) to organize the prospective collection of high-quality homogeneous samples. Second, they can rapidly receive a large number of samples that have been retrospectively collected, instead of waiting (often for years) to collect samples with specific characteristics. This system has allowed an effective acceleration of research and the use of retrospectively collected samples that otherwise often remain unutilized in freezers of individual researchers – thus avoiding an enormous waste of resources.
We are currently implementing the collection of samples (e.g., other types of cancer pathologies and patients during follow-up) through the involvement of other departments. At researchers’ request, we are evaluating procedures and protocols to collect, prepare and store other sample types, such as viable cells, ascites and sebum.
We are also planning to set up a high-throughput nucleic acid extractor facility to systematically prepare nucleic acid samples from buffy coat and tissues.
We are in the process of updating the CRO-Biobank webpage with a sample archive, an explanation of the mechanisms for accessing CRO-Biobank samples and information, and links to forms for requesting samples, in order to expand the possibility of delivering samples to external researchers or institutions.
In conclusion, the CRO-Biobank collection relies on a wide range of tumor and normal biospecimens, including rare tumors, allowing it to supply the research community. The biobanking process is implemented to guarantee the safety of the operators, the privacy of the patients and the traceability and quality of samples. Here we have described the organization of the CRO-Biobank, mainly focusing on ethical, legal, IT, sustainability and procedural aspects, and have offered new insights and help with solving practical issues that arise in developing and operating biobanks.
Footnotes
Acknowledgements
We thank the patients who participated in this project, and the physicians, nurses and other health care professionals who contribute to the activity of the CRO-Biobank.
Experiments on human subjects: The procedures followed were approved by the CRO ethics committee and were in accordance with the Helsinki Declaration.
Financial support: This project was entirely funded by the CRO Aviano National Cancer Institute.
Conflict of interest: No authors have any proprietary interest in this study.
