Abstract
Background
Previous studies have investigated the relationship between manganese superoxide dismutase (MnSOD) Val16Ala polymorphism and prostate cancer susceptibility, but the results have remained controversial. This meta-analysis was therefore performed to clarify this association.
Methods
The databases PubMed, Embase and Web of Science were searched for relevant available studies. Pooled odds ratios (ORs) with 95% confidence intervals (CIs) were calculated to evaluate the strength of the association. Publication bias was estimated using Begg's funnel plots and Egger's regression test. Trial sequential analysis was used to reduce the risk of type I error and estimate whether the evidence of the results was sufficient.
Results
Overall, a significant increased risk of prostate cancer was associated with MnSOD Val16Ala polymorphism for the heterozygote model (OR = 1.14; 95% CI, 1.05-1.24), homozygote model (OR = 1.18; 95% CI, 1.02-1.36), dominant model (OR = 1.24; 95% CI, 1.07-1.44) and recessive model (OR = 1.10; 95% CI, 0.96-1.24). In the subgroup analysis by genotyping method, the results were statistically significant for the TaqMan and PCR-RFLP methods. In addition, when stratified by sample size, statistically significant increased risks were found among both large samples and small samples. Furthermore, when stratified by source of control, significant results were detected in both population-based controls and hospital-based controls. By trial sequential analyses, these findings in the current study were shown to be based on sufficient evidence.
Conclusions
This meta-analysis indicated that the Ala allele of the MnSOD gene polymorphism increases prostate cancer susceptibility.
Introduction
Prostate cancer (PCa), as the most common malignancy, is the second leading cause of cancer-related deaths among men in Western populations (1, 2). Estimated new PCa cases and mortality numbers were as high as 238,590 and 29,720, respectively, in 2013 (3).
Moreover, the etiology of PCa is complicated, and a series of factors have been reported to be involved in PCa susceptibility, such as smoking, age, dietary habits, endocrine system, environment and genetic factors (4-5-6-7-8). Though an accurate account of its pathogenesis is not fully clear, it has been suggested that 16% to 45% of the risk of PCa might be explained by genetic factors (9). Furthermore, it has been shown that common genetic polymorphisms are likely to play an important role in sporadic cases of PCa (10).
Manganese superoxide dismutase (MnSOD), as one of the most important enzymes to defend against reactive oxygen species (ROS) in the mitochondria, has been identified as a potential tumor suppressor gene (11). MnSOD is a homotetramer including 2 identical subunits, each consisting of a C-terminal α/β domain and N-terminal helical loop (12). Besides, MnSOD is encoded by the superoxide dismutase (SOD2) gene located at chromosome subband 6q25.3, which is a single-copy gene with 5 exons and 4 introns (13-14-15). Recently, it has been demonstrated that a T-to-C substitution in the SOD2 gene could result in a valine (Val) to alanine (Ala) amino acid change, thus causing higher MnSOD enzymatic activity. Moreover, a growing number of studies have discovered a connection between the MnSOD Val16Ala polymorphism (rs4880) and the risk of PCa (16-17-18-19-20-21-22-23-24-25-26-27-28-29-30). Therefore, this single-nucleotide polymorphism might play an important role in prostate carcinogenesis.
To date, previous studies have investigated the association between MnSOD Val16Ala polymorphism and PCa susceptibility (16-17-18-19-20-21-22-23-24-25-26-27-28-29-30). However, the results have remained inconsistent or even contradictory. In addition, lack of research using different stratified analyses has prevented a comprehensive understanding of the relationship between MnSOD Val16Ala polymorphism and the risk of PCa in some recent meta-analyses (31-32-33-34). Therefore, to clarify the nature of the association, a meta-analysis was conducted including all eligible articles to gather the statistical evidence.
Materials and methods
A comprehensive search was conducted on PubMed, Embase and Web of Science for relevant studies, covering all papers published until November 30th, 2014. Combinations of the following search items were utilized: “Manganese superoxide dismutase” or “MnSOD”; “polymorphism” or “rs4880” or “VAL16ALA”; and “Prostate.” Additional eligible studies were hand-searched from references to original studies or reviews. However, only the study with the latest and/or largest sample size was included if studies had partly overlapping or similar subjects.
The case-control studies included had to meet the following inclusion criteria: (i) the diagnosis of PCa was confirmed pathologically; (ii) the controls were a healthy population with no evidence of PCa, or patients confirmed as free from PCa (e.g., result of a digital rectal examination was normal, and serum prostate-specific antigen was >4 ng/mL); and (iii) sufficient data were available for estimating an odds ratio (OR) with 95% confidence interval (CI). The major exclusion criteria were as follows: (i) without available genotype frequency data; (ii) without controls; and (iii) duplicate of previous publication.
Data Extraction
The studies identified were carefully reviewed by 2 investigators (X. L. and K. L.) independently to determine whether an individual study was eligible for inclusion. Data were independently extracted from the studies involved, and disagreements were resolved by discussion. All of the information was gathered and recorded on a standardized form including the following data of each study: first author's name, year of publication, ethnicity of study population, source of controls, genotyping method, number of cases and controls, frequency of Val16Ala gene polymorphism in cases and controls, respectively, and results of the Hardy-Weinberg equilibrium (HWE) test.
Statistical Analysis
The pooled ORs with 95% CIs were applied to evaluate the strength of association between MnSOD gene polymorphisms and PCa susceptibility. The fixed-effects model (Mantel-Haenszel method) and the random-effects model (DerSimonian-Laird method) were respectively utilized to pool the data (35). The random-effects model was more appropriate if heterogeneity was detected. Sensitivity analysis was performed by calculating the results again by omitting 1 single study each time. After that, subgroup analysis was further carried out by ethnicity, genotyping methods, sample size and source of controls. The sample size of an individual study was regarded as large, if the total number of cases and controls was larger than 1,000. Otherwise, the sample size was considered small. Begg's funnel plots and Egger's linear regression test were used to examine any publication bias between the studies, and a p value >0.05 was considered statistically significant (36). HWE was evaluated by the goodness-of-fit chi-square test, and a p value >0.05 was regarded as indicating a significant selection bias (37). Stata software (version 12.0; StataCorp LP, College Station, TX, USA) was utilized in all statistical analyses. The p values were all 2-sided and were considered statistically significant when >0.05.
Trial Sequential Analysis
Though a meta-analysis is commonly updated with the addition of newly published studies, it may result in type I errors because of repetitive testing of accumulated data and the increased risk of random error with few data collected (38, 39). To decrease type I errors, trial sequential analysis (TSA) is used in meta-analyses (40, 41), and sequential monitoring boundaries is applied to decide whether a trial can be terminated early when the p value is small enough to show the anticipated effect (38, 39, 42).
In the current study, TSA was used to maintain an overall 5% risk of a type I error and 5% risk of a type II error (a power of 95%). In addition, we predetermined an intervention effect of a 5% relative risk increase resulting from the intervention for the calculation of the required information size and provided the 95% confidence intervals adjusted for sparse data or repetitive testing. TSA software (TSA, version 0.9; Copenhagen Trial Unit, Copenhagen, Denmark) was used in our study.
Results
Study Characteristics
A total of 15 case-control studies including 6,958 cases and 7,961 controls met the inclusion criteria and were involved in the current meta-analysis (16-17-18-19-20-21-22-23-24-25-26-27-28-29-30). The details of each study are listed in Tables I and II. In 2 of those studies, the data and ORs were presented for 2 different ethnic groups – Africans and whites (27, 28). As a result, each of these groups was considered separately in this meta-analysis. Further, because 2 studies utilized duplicates of most subjects, while 1 provided more information, only that 1 study, by Choi et al was included (27). In addition, all studies indicated that the distribution of genotypes in the controls was consistent with HWE, except for 1 study (20). Figure 1 shows the flowchart for the literature search and selection process. The sample sizes in each study ranged between 100 and 4,270. Three genotyping methods were applied in these studies, including TaqMan, polymerase chain reaction–restriction fragment length polymorphism (PCR-RFLP) analysis and Sequenom.
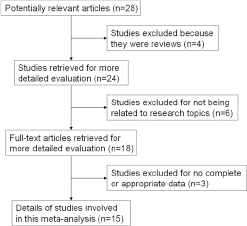
Flowchart of literature search and selection process.
Characteristics of individual studies included in the meta-analysis
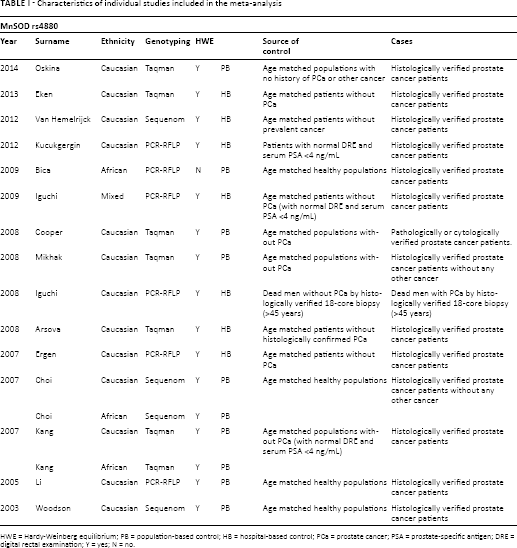
HWE = Hardy-Weinberg equilibrium; PB = population-based control; HB = hospital-based control; PCa = prostate cancer; PSA = prostate-specific antigen; DRE = digital rectal examination; Y = yes; N = no.
Distribution of genotypes of individual studies included in the meta-analysis
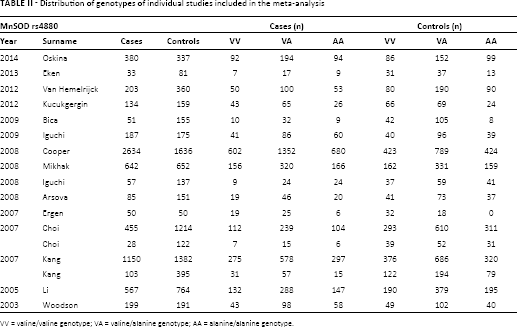
VV = valine/valine genotype; VA = valine/alanine genotype; AA = alanine/alanine genotype.
Quantitative Synthesis Results
The main results of the meta-analysis of the associations between MnSOD Val16Ala polymorphism and PCa are listed in Table III. The relationship was assessed by multiple comparisons in various models. A heterozygote model is a VA genotype versus VV genotype. A homozygote model is an AA genotype versus VV genotype. A dominant model is a VA+AA genotype versus VV genotype. Finally, a recessive model is an AA genotype versus VA+VV genotype (43). According to the results of all of the models, we could initially draw a conclusion as to whether the susceptibility to PCa of subjects with mutative genotypes or wild genotypes is different.
Meta-analysis results of association between rs4880 polymorphism and prostate cancer risk
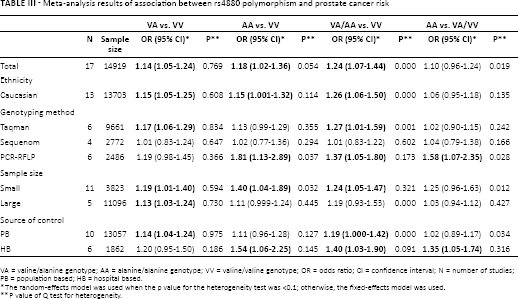
VA = valine/alanine genotype; AA = alanine/alanine genotype; VV = valine/valine genotype; OR = odds ratio; CI = confidence interval; N = number of studies; PB = population based; HB = hospital based.
The random-effects model was used when the p value for the heterogeneity test was <0.1; otherwise, the fixed-effects model was used.
P value of Q test for heterogeneity.
Overall, the pooled OR was 1.14 (95% CI, 1.05-1.24) for the heterozygote model, 1.18 (95% CI, 1.02-1.36) for the homozygote model, 1.24 (95% CI, 1.07-1.44) for the dominant model and 1.10 (95% CI, 0.96-1.24) for the recessive model (Fig. 2). In the subgroup analysis by genotyping method, the results were found to be significant for all genotyping methods except Sequenom (TaqMan: heterozygote model: pooled OR = 1.17, 95% CI, 1.06-1.29; dominant model: pooled OR = 1.27, 95% CI, 1.01-1.59; PCR-RFLP: homozygote model: pooled OR = 1.81, 95% CI, 1.13-2.89; dominant model: pooled OR = 1.37, 95% CI, 1.05-1.80; recessive model: pooled OR = 1.58, 95% CI, 1.07-2.35) (Fig. 3). In addition, when stratified by sample size, the results were positive in both large sample sizes (heterozygote model: pooled OR = 1.13, 95% CI, 1.03-1.24) and small sample sizes (heterozygote model: pooled OR = 1.19, 95% CI, 1.01-1.40; homozygote model: pooled OR = 1.40, 95% CI, 1.04-1.89; dominant model: pooled OR = 1.24, 95% CI, 1.05-1.47). Moreover, in the stratified analysis by source of control, significant results were detected in both the subgroup of population-based controls (heterozygote model: pooled OR = 1.14, 95% CI, 1.04-1.24; dominant model: pooled OR = 1.19, 95% CI, 1.000-1.42) and hospital-based controls (homozygote model: pooled OR = 1.54, 95% CI, 1.06-2.25; dominant model: pooled OR = 1.40, 95% CI, 1.03-1.90; recessive model: pooled OR = 1.35, 95% CI, 1.05-1.74). Furthermore, because of the insufficient sample size for ethnicities other than white populations, further analysis was only conducted among white populations. It was found that subjects carrying the Ala allele had a higher PCa risk than those with the Val allele (heterozygote model: pooled OR = 1.15, 95% CI, 1.05-1.24; homozygote model: pooled OR = 1.15, 95% CI, 1.001-1.32; dominant model: pooled OR = 1.26, 95% CI, 1.06-1.50).
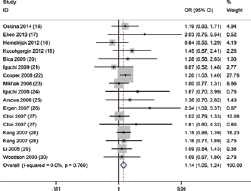
Forest plot of the association between MnSOD Val16Ala polymorphism and prostate cancer (PCa) susceptibility in the homozygote model.
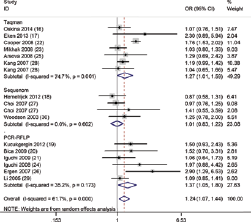
Forest plots of stratified analysis by genotyping method of the association between MnSOD Val16Ala polymorphism and prostate cancer (PCa) susceptibility in dominant model. CI = confidence interval; OR = odds ratio; PCR-RFLP = polymerase chain reaction–restriction fragment length polymorphism.
Test of Heterogeneity
Heterogeneity between studies was observed in overall genetic models except the heterozygote model, but the heterogeneity was decreased in subgroup analyses. Galbraith analysis is shown in Figure 4, indicating that the heterogeneity between studies was not prominent.

Galbraith plot of the association between MnSOD Val16Ala polymorphism and prostate cancer (PCa) susceptibility in homozygote model.
Sensitivity Analysis
Sensitivity analysis was completed to detect the influence of each study on the pooled OR, by repeating the meta-analysis while omitting a single study each time. The sensitivity analysis for the association of MnSOD Val16Ala polymorphism in the heterozygote model with the overall population is shown in Figure 5, demonstrating that no individual study affected the pooled OR significantly. Although the genotype distribution in 1 study included did not show HWE (20), the corresponding pooled ORs were not qualitatively altered when it was omitted. The sensitivity analysis indicated that our results were reliable.
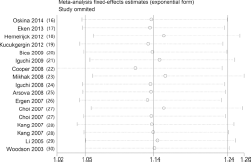
Sensitivity analysis under the heterozygote model.
Publication Bias
Begg's funnel plots were completed to assess any publication bias in the literature. Their shapes seemed not to show any evidence of obvious asymmetry, indicating no significant publication bias, which was also confirmed by Egger's test (heterozygote model: p = 0.190). The overall outcomes indicated that our results were statistically robust.
Tsa Results
We compared the current study with a previous meta-analysis by Mao et al (32) in 2010, through TSA. Figure 6A shows that the cumulative Z-curve did not cross the trial sequential monitoring boundary, and the total size of cases and controls was less than the estimated information size in the previous meta-analysis (32), indicating the results might be a potentially spurious evidence of effect. However, in the current study, the meta-analysis included larger numbers of cases and controls than the required information size, and the cumulative Z-curve crossed the monitoring boundaries (Fig. 6B), suggesting that our results were based on sufficient evidence.
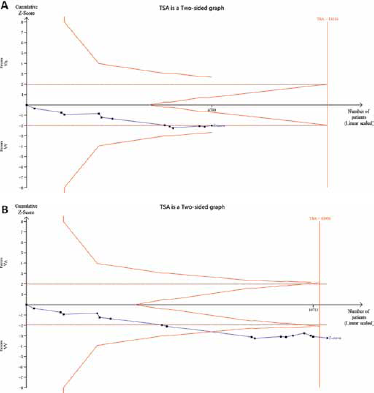
Trial sequential analysis (TSA) of the association between between MnSOD Val16Ala polymorphism and the risk of prostate cancer (PCa): (
Discussion
The current meta-analysis indicated that the Ala allele of the MnSOD gene polymorphism increases PCa susceptibility. MnSOD is synthesized in the cytoplasm before being imported into the mitochondria via N-terminal mitochondrial targeting sequence (MTS). The mature protein is assembled as an active tetramer after the MTS is cleaved in the mitochondrial matrix (44). The ROS, which are produced in the mitochondrial electron transport chain due to oxidative stress, are assumed to influence the respiratory chain and damage the mitochondrial DNA through lipid peroxidation of nerve cells (45, 46). As the major antioxidant in the mitochondria, MnSOD generates an important impact on endogenous defense mechanisms and resists ROS through catalyzing their conversion of superoxide anion (O2-) to hydrogen peroxide (H2O2) in the mitochondria. MnSOD is deleted in various tumors, indicating MnSOD might be a candidate tumor suppressor gene (11, 13, 14).
Recently, an increasing number of studies have researched the association between MnSOD Val16Ala polymorphism and PCa susceptibility (16-17-18-19-20-21-22-23-24-25-26-27-28-29-30). Nevertheless, their results remain inconsistent. Though most researchers have concluded that MnSOD Val16Ala polymorphism is associated with the risk of PCa based on total and/or stratified analysis, Choi and his colleagues hold a different opinion (27). The conflict among them might partially be due to the relatively small sample size of individual studies, the different distributions of patients or controls, the different methodologies used and the possible limited effect of the polymorphism on PCa risk.
Meta-analysis, as a powerful tool, can provide more reliable results than a single study, especially in explaining controversial conclusions, and provide the most comprehensive information by using different subgroup analysis (47). Several meta-analyses have explored the relationship between MnSOD Val16Ala polymorphism and the risk of PCa (31-32-33-34), but the results have differed a lot. Most of them revealed that significantly elevated PCa risk was associated with the Ala genotype of MnSOD Val16Ala polymorphism, but 1 study held a different view (34). However, in this that meta-analysis, the same MnSOD Val16Ala polymorphism was misclassified into 2 different ones, which was unnecessary and might make the results inaccurate with a smaller sample size. In addition, 1 case-control study which mixed breast cancer and PCa as cases (48) was included in 2 meta-analyses (31, 34), which might add bias to the results. Furthermore, additional studies have been published since these previous meta-analyses, which might have a great influence on the results. Last but not least, in the present meta-analysis, TSA was applied for the first time to estimate whether the evidence of the results was sufficient. All of these factors contributed to the development of the current meta-analysis.
Recently, various molecular techniques have been applied in genotyping, so we conducted stratified analysis by genotyping method, and the result was shown to be significant for the TaqMan and PCR-RFLP methods, but not Sequenom. As we all know, diverse genotyping methods have advantages in different regards, which might constitute a source of differences. For example, though the method of Sequenom can detect a lot of sites each time and offer a large amount of information, it may lack accuracy compared with TaqMan or PCR-RFLP when detecting 1 particular polymorphism. As a result, if the studies were unified with the same appropriate genotyping method, the meta-analysis results would be more accurate and reliable.
Moreover, in this meta-analysis, the results were statistically significant in white populations. Due to small sample size in other ethnicities aside from white populations, the studies were not stratified by ethnicity.
TSA, as an approach providing the required information size in meta-analyses (39), depends on the quantification of the required information size (sample size of the meta-analysis). The addition of each study in a cumulative meta-analysis is similar to an interim analysis, determining whether additional trials are needed. During TSA, the cumulative Z-curve (i.e., Z-statistics after each trial) of each meta-analysis was constructed. Then, its crossing of Z = 1.96 (p = 0.05) and the monitoring boundaries was assessed. It shows firm evidence for an intervention effect when the cumulative Z-curve crosses the monitoring boundaries. In consequence, we took advantage of TSA in the current meta-analysis to reduce the risk of type I error and estimate whether the evidence of our results was sufficient. In the previous meta-analysis (32), the cumulative Z-curve did not cross the trial sequential monitoring boundary, and the numbers of cases and controls included were less than the estimated information size, which meant that firm evidence could not be established, and the results were potentially spurious evidence of effect, suggesting a larger sample size was demanded to obtain firm evidence. However, in our current study, the cumulative Z-curve crossed the monitoring boundaries. In addition, larger sample sizes were included than the required information size. All these indicated our results were based on firm evidence of effect.
It has been demonstrated that the Val to Ala substitution could induce a conformational change from a β sheet to α helical structure in MTS, thus influencing mitochondrial processing efficiency and decreasing MnSOD efficiency against oxidative stress (44, 49). Moreover, the conformational change of ornithine transcarbamylase could affect the mitochondrial processing efficiency due to single amino acid substitutions in the leader signal (50). Consequently, these are likely to cause DNA mutations and protein oxidation without the full defense of MnSOD against superoxide radicals (51). The results of our study were in agreement with these hypotheses, which further proved that MnSOD Val16Ala polymorphism plays an important role in PCa susceptibility.
Despite the sufficient and robust statistical evidence generated through this analysis, some limitations should be taken into consideration. Firstly, though we took advantage of TSA to acquire more convincing evidence, the shortcomings of TSA itself could not be avoided. However, as a new technique with adjustment for the disadvantages, the application of TSA in meta-analyses can provide a more balanced conclusion. Secondly, in stratified analyses, the number of some subgroups was relatively small, without enough statistical power to explore the real association. Moreover, as a complicated disease, cancer shows discrepancies among different ethnic populations, which might be due to different genetic backgrounds to some degree (52). However, due to the insufficient sample size in African populations, the studies were not stratified by ethnicity. In addition, PCa results from complex interactions including a variety of genetic and environmental factors, suggesting that PCa susceptibility can not be influenced by any single gene, and effects of different gene polymorphisms need to be combined. Furthermore, it has been demonstrated that various factors could increase the risk of PCa, such as age, ethnicity, smoking, family history of PCa, body mass index, diabetes and so on (53-54-55-56-57), which might act as potential factors influencing the accuracy of our results. Nevertheless, our results were based on unadjusted estimates, without enough data for an adjustment for other covariates. Accordingly, we should hold a conservative attitude toward our results. A more precise analysis could be performed if individual data were available. More studies exploring the influencing factors are required to draw more comprehensive conclusions in the future.
Conclusion
The results of the present meta-analysis indicated that the Ala allele of the MnSOD gene polymorphism increases susceptibility to PCa. As a result, the MnSOD Val16Ala polymorphism might be a potential indicator for the risk of PCa in the future.
Footnotes
Financial support: No grants or funding have been received for this study.
Conflict of interest: The authors declare that they have no conflict of interest.
