Abstract
Background
Several studies have recently investigated the association between mucin 1 (MUC1) rs4072037 polymorphism and gastric cancer (GC) risk, but with conflicting results. The aim of this meta-analysis was to evaluate the association between MUC1 rs4072037 polymorphism and GC risk.
Methods
A comprehensive database search of PubMed, Elsevier, Embase and China National Knowledge Infrastructure (CNKI) databases was performed to identify relevant studies. Pooled odds ratios (ORs) and 95% confidence intervals (CIs) were used to assess the strength of any association.
Results
A total of 12 papers containing 18 studies were included in this meta-analysis, involving 12,373 cases and 15,008 controls. Our data suggested that rs4072037 polymorphism was associated with a decreased risk of GC. Stratification analyses of ethnicity indicated that rs4072037 decreased the risk of GC among white populations, but no significant relationship was observed among Asian populations. No significant associations were observed in subgroups of Lauren classification (intestinal or diffuse) and anatomical classification (cardia or non-cardia).
Conclusions
In conclusion, this meta-analysis suggested that rs4245739 polymorphism in the MUC1 gene may play a pivotal role in the pathogenesis of GC, especially for white populations.
Introduction
Gastric cancer (GC) is the fourth most prevalent cancer and the second leading cause of cancer-related death worldwide (1). GC causes more than 700,000 deaths each year around the world. It has been a heavy burden in developing areas like East Asia, Eastern Europe and South America. The etiology of GC remains unclear. Helicobacter pylori infection has been recognized as a widely accepted risk factor for GC (2). Research shows that almost half of the population in the world is infected with H. pylori. However, only a minority of those infected individuals eventually develop GC, indicating that hereditary and environmental factors may also play important roles in the pathogenesis of GC (3).
Mucin 1 (MUC1) is a heavily polymorphically glycosylated mucin that is expressed on the surfaces of epithelia containing gastric mucosa (4). MUC1 protein protects gastric epithelial surfaces against environmental insults that may cause inflammation and carcinogenesis (5). Baldus and Hanisch detected high levels of MUC1 mRNA in GCs (6). Studies have demonstrated that the MUC1 gene plays an important role in forming protective mucous barriers on epithelial surfaces of the stomach (7). All of this cumulative evidence indicates a possible role for the genetic variation of MUC1 in the pathogenesis of GC.
MUC1 rs4072037 polymorphism is located in the second exon of MUC1 at chromosome band 1q22. This single nucleotide polymorphism (SNP) can alter transcriptional regulation, and determines splice variants in MUC1. The association between MUC1 rs4072037 polymorphism and the risk of GC has been explored in many studies (4, 8-18), but with conflicting and inconclusive results. The clinical heterogeneity and small sample sizes of these studies, and different ethnic populations may contribute to these disparities. To overcome these limitations, several research groups have conducted meta-analyses on this topic (19-22). However, these meta-analyses had some unavoidable defects, such as omitting studies that were in accordance with the inclusion criteria or including studies that did not conform to Hardy-Weinberg equilibrium (HWE). Additionally, their findings were not exactly the same. Furthermore, several studies have been reported in recent years since these meta-analyses. Consequently, to better understand the roles of MUC1 rs4072037 polymorphism in the development of GC, we explored the association between MUC1 rs4072037 polymorphism and GC risk by performing a comprehensive meta-analysis.
Methods
Literature search
We systematically searched the PubMed, Elsevier, Embase and China National Knowledge Infrastructure (CNKI) databases to identify studies through June 20, 2016. We used the following search terms: “Gastric Neoplasm,” “Stomach Cancer,” “Gastric Cancer,” “Gastric Carcinoma,” “Gastric Adenocarcinoma,” “Mucin-1,” “MUC1,” “MUC-1” “polymorphism,” “SNP” and “polymorphisms”. Two independent investigators performed the literature search. No restrictions were placed on the search. Additional initially omitted studies were searched manually.
Inclusion and exclusion criteria
Studies were included according to the following criteria: (i) they evaluated the association between GC risk and MUC1 rs4072037 polymorphism, (ii) they were studies of humans, (iii) they provided sufficient data to calculate the odds ratios (ORs) and 95% confidence intervals (CIs), (iv) they were case-control studies and (v) they conformed to the HWE. Exclusion criteria were (i) duplication of previous studies, (ii) they were case reports or review articles and (iii) they were studies without detailed genotype data.
Data extraction and quality assessment
The following information was extracted: name of first author, publication year, country of origin, ethnicity and genotypes of study population, and numbers of cases and controls. Two investigators conducted the extraction of data and assessed study quality independently based on the Newcastle-Ottawa scale (NOS) (23). All conflicting information was discussed and resolved by consensus.
Statistical analysis
Crude ORs and 95% CIs were used to investigate the strength of any association between GC risk and MUC1 rs4072037 polymorphism. Stratification analyses were carried out by ethnicity, Lauren classification (intestinal or diffuse) and anatomical classification (cardia or non-cardia). A p value <0.05 was considered statistically significant. Heterogeneity between studies was measured by chi-square-based Q test. A fixed-effects model was used when p was >0.1 or I 2 <50%. Otherwise, a random-effects model was applied (24). Pooled ORs were calculated for allele model, dominant model, recessive model, homozygous model and heterozygous model. We performed sensitivity analyses by omitting each study in turn to evaluate the stability of the overall results. HWE was assessed in the controls using Pearson's chi-square test. Potential publication bias was assessed by Begg's and Egger's linear regression tests (25); a p value <0.05 was considered to be statistically significant. All statistical analyses were performed using Stata 11.0 software (StataCorp, College Station, TX, USA). The power of this meta-analysis for MUC1 rs4072037 polymorphism was calculated according to the method recommended by Hedges and Pigott (26), with a significance value of 0.05.
Results
Characteristics of the publications included
Our search yielded 366 papers after an initial search. After removing duplicates and screening the titles and abstracts, 346 papers were excluded. Finally, 20 papers were selected for further full-text review. Eight papers were excluded due to the following reasons: 2 papers (27, 28) described other polymorphisms of MUC1, 2 (29, 30) did not provide detailed genotyping data, and 4 (21, 31-33) did not conform to HWE. Twelve eligible papers (4, 8-18) containing 18 studies (12,373 cases and 15,008 controls) were identified in this meta-analysis. The selection process for eligible papers is presented in Figure 1. The characteristics of the studies included are shown in Table I. The NOS scores of all studies included ranged from 5 to 7 stars, suggesting that they were studies of high methodological quality. Among the 18 studies included in the meta-analysis, 15 were carried out in Asian populations, and 3 in white populations. The research conducted by Abnet et al (18) consisted of 5 studies; the research conducted by Saeki et al (10) included 3 studies.
Characteristics of included studies
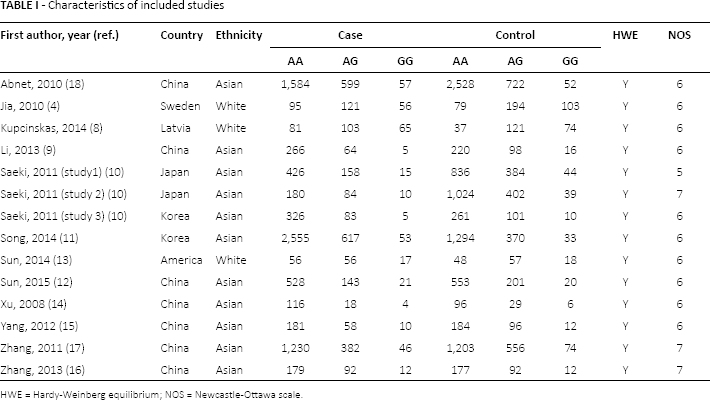
HWE = Hardy-Weinberg equilibrium; NOS = Newcastle-Ottawa scale.
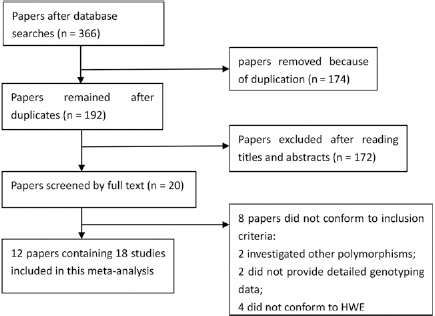
Selection of eligible papers to be included in this meta-analysis. HWE = Hardy-Weinberg equilibrium.
Meta-analysis of MUC1 rs4072037 polymorphism
As shown in Table II, the MUC1 rs4072037 polymorphism was significantly associated with a decreased risk of GC in all 3 models (GG + AG vs. AA: OR = 0.86; 95% CI, 0.75-0.98, p = 0.030; Fig. 2). Stratification analyses of ethnicity indicated that rs4072037 polymorphism was significantly associated with a decreased risk of GC among white populations (G vs. A: OR = 0.82; 95% CI, 0.72-0.94, p = 0.004; Fig. 3), but not among Asian populations (Tab. III). No significant associations were observed in subgroups of Lauren classification (intestinal or diffuse) or anatomical classification (cardia or non-cardia). All included studies conformed to HWE, indicating that their control subjects were representative of the general population.
Meta-analysis of association between MUC1 rs4072037 polymorphism and gastric cancer risk
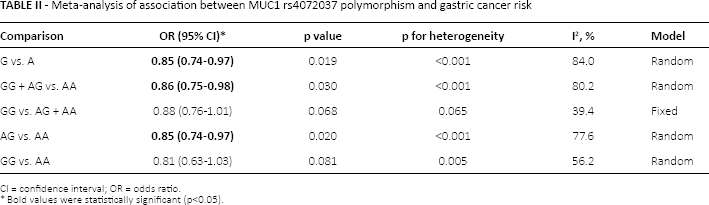
CI = confidence interval; OR = odds ratio.
Bold values were statistically significant (p<0.05).
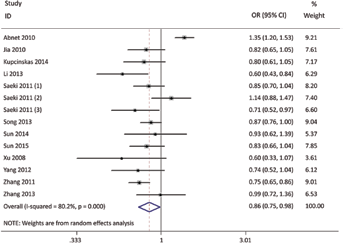
Forest plot for dominant model (GG + AG vs. AA).
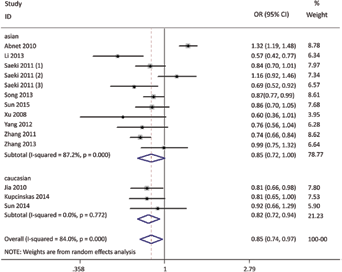
Stratification analyses of ethnicity in allele model (G vs. A).
Summary of subgroup analyses in this meta-analysis
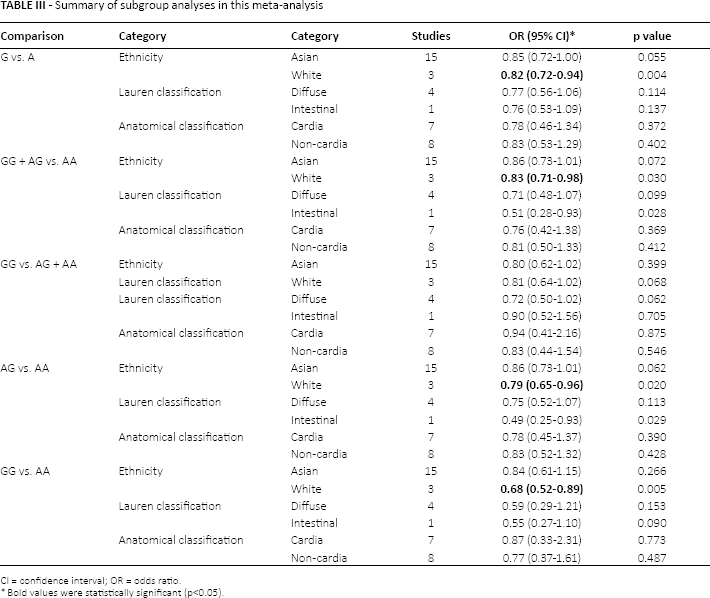
CI = confidence interval; OR = odds ratio.
Bold values were statistically significant (p<0.05).
Sensitivity analyses and publication bias
We conducted sensitivity analyses by omitting each study one at a time in every genetic model for rs4072037. Excluding every single study from the analysis did not affect the pooled results for the rs4072037 polymorphism, indicating that our data for this SNP were stable and trustworthy (AG vs. AA; Fig. 4). Both Egger's and Begg's tests (GG vs. AG + AA; Fig. 5) were used to evaluate the publication bias of this meta-analysis. Our data revealed that there was no obvious publication bias for the rs4072037 polymorphism.
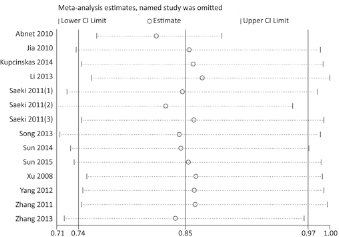
Sensitivity analysis in heterozygous model (AG vs. AA).
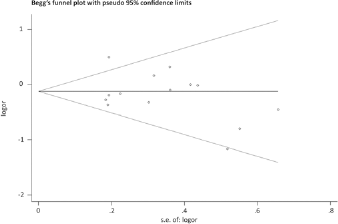
Funnel plot for publication bias in recessive model (GG vs. AG + AA).
Discussion
The present meta-analysis explored recent studies regarding the potential association between MUC1 rs4072037 polymorphism and susceptibility to GC. The data of this meta-analysis suggested that MUC1 rs4072037 polymorphism was associated with a decreased risk of GC. Stratification analyses of ethnicity indicated that rs4072037 polymorphism decreased the risk of GC among white populations, but no significant relationship was observed among Asian populations. In subgroup analysis, no significant interactions were observed between rs4072037 polymorphism and clinicopathological characteristics such as tumor site (cardia or non-cardia) or histological type (intestinal or diffuse).
MUC1, a membrane-bound protein, is highly expressed on the gastric mucosal surface (34). It acts as a barrier against exogenous insults in normal epithelial cells. The genetic variants in the MUC1 gene have been shown to play important roles in some types of cancers, including GC (35). The rs4072037 polymorphism is recognized to be a splicing acceptor site in the second exon of MUC1 (36). Studies demonstrated that rs4072037 polymorphism of MUC1 gene regulates alternative splicing of the second exon, and alters the transcriptional activity of the promoter (10). Risk allele A at the rs4072037 polymorphism has been reported to be associated with a decreased physiological function of MUC1 (33). Recently, many studies (4, 8-18, 21) have reported the association between MUC1 rs4072037 polymorphism and GC susceptibility, but their findings were conflicting. And these studies may have had inadequate statistical power due to relatively small samples, clinical heterogeneity and different ethnic populations. Several meta-analyses (19-22) investigated this topic, but they included some studies that did not conform to HWE, and their sample sizes were not very large. In addition, these previous meta-analyses had remarkable limitations that can not be ignored. For example, in a study by Zheng et al (22), we found they used the fixed-effects model mistakenly under the condition of I 2 = 70% and a p value of 0.07; when actually a random-effects model should have been adopted. And they did not include a Chinese study (15). As for 2 meta-analyses (19, 20) conducted by Liu et al and Duan et al, there were 2 studies (14, 16) that should have been included in these meta-analysis but were omitted. With regard to the recent meta-analysis by Qiu et al (21), it did not include 6 studies (10, 12, 13, 15, 16, 18) which actually conformed to the inclusion criteria. Furthermore, the p value for HWE (p<0.00001) in that study by Qiu et al was less than 0.05, indicating that their control subjects were not representative of the general population. As a result, the stability and reliability of their conclusions (21) should be interpreted with doubt.
Due to these limitations in the above meta-analyses, we performed this comprehensive meta-analysis to assess the relationship between MUC1 gene polymorphism rs4072037 and GC susceptibility. Our data suggested that rs4072037 polymorphism was weakly associated with a decreased risk of GC.
Previous meta-analyses (19-22) also explored the relationship between this SNP and GC susceptibility, but with inconsistent results. All of these meta-analyses concluded that the rs4072037 polymorphism was significantly associated with a decreased risk of GC. Furthermore, stratification analyses of ethnicity in these meta-analyses (19-22) indicated that rs4072037 polymorphism was related to the decreased risk of GC among white and Asian populations. However, our data revealed that this SNP decreased the risk of GC among white populations, while it was not associated with any risk for GC in Asians. The genetic background of GC may vary among different ethnicities. Compared with those of previous meta-analyses, our conclusions were more robust due to the larger sample sizes. We analyzed 15 studies including 11,987 cases and 14,340 controls among Asian populations. Furthermore, the power analysis showed that this meta-analysis had a power of 99% to detect the effects of rs4072037 polymorphism on Asian GC susceptibility, assuming an OR of 0.86, indicating that our data were trustworthy.
We further evaluated the association in subgroups of Lauren classification and anatomical classification. No significant associations were observed in these subgroups. MUC1 rs4245739 polymorphism was not associated with the risk of intestinal or diffuse GC, or in patients with cardia or non-cardia tumor. Approximately 90% of GCs are adenocarcinomas, which are divided into intestinal-type GC (IGC) and diffuse-type GC (DGC) (10). Saeki et al (10) indicated that this SNP rs4072037 in MUC1 increased the risk of DGC, while Kupcinskas et al (8) suggested the rs4072037 polymorphism decreased the risk of DGC. Subsequent meta-analyses (20, 22) demonstrated significant associations with both IGC and DGC. However, our data revealed this SNP was not associated with the risk of IGC or DGC. The different sample sizes and clinical heterogeneity of GC may partially explain the different results of our meta-analysis and those of Saeki et al and Kupcinskas et al. It is obvious that the sample sizes of these studies were not large enough to obtain a relative precise estimate for the strength of association. Further studies were needed to validate these findings.
This meta-analysis has several strengths when compared with previous meta-analyses. First, all studies included conformed to HWE. Testing for deviation from HWE is necessary for meta-analysis of case-control studies (37). Second, more studies were included in this meta-analysis, and the sample size of this meta-analysis including 12,373 cases and 15,008 controls was large enough. Third, sensitivity analysis suggested that the data of this meta-analysis were stable and robust regarding the rs4072037 polymorphism. Additionally, the power analysis showed that this meta-analysis had a power of 99% to detect the effects of rs4072037 polymorphism on GC susceptibility, assuming an OR of 0.86.
However, several potential limitations of this meta-analysis should be acknowledged. First, we could not conduct any analysis of the subgroups of age, sex, smoking, drinking and H. pylori infection, due to the lack of corresponding data. Second, our results were based on unadjusted estimates for confounding factors, which might have affected the final results. Third, we could not assess potential gene–gene and gene–environment interactions because of the lack of original data.
Conclusions
In conclusion, this meta-analysis indicates that the rs4072037 polymorphism of the MUC1 gene plays important roles in GC pathogenesis, especially among white populations. Further studies are necessary to explore the specific mechanism between MUC1 rs4072037 polymorphism and GC.
Footnotes
Abbreviations
Financial support: No grants or funding have been received for this study.
Conflict of interest: The authors declare they have no conflicts of interest.
