Abstract
Background
Endometrial cancer (EC) is a hormone-related disease, showing highly diverse features of ER/PR/HER2 status-based molecular subtypes. Long noncoding RNA (lncRNA) HOX antisense intergenic RNA (HOTAIR) has recently emerged as a key molecule in many cancers, triggering epithelial-mesenchymal transition (EMT)–mediated cancer stem cell (CSC) formation, but little is known about its significance in EC. Thus, we aimed to investigate the clinical significance of HOTAIR itself in different molecular subtypes of EC and possible links between HOTAIR, EMT and CSC-related markers.
Methods
The study group included 156 consecutive, stage I-IV EC patients treated between 2000 and 2010. ER, PR and HER2 protein expression were examined by immunohistochemistry (IHC) on tissue microarrays. RT-qPCR was used to analyze the expression levels of HOTAIR, EMT-related genes – SNAIL and SLUG – and the CSCs marker CD133.
Results
Molecular subtypes, defined as ER/PR+HER2+, ER/PR+HER2-, ER-PR-HER2+ and ER-PR-HER2-, occurred in 40.2%, 52.3%, 4.7% and 1.9% of cases, respectively. The expression of HOTAIR did not differ between the subtypes, but high HOTAIR expression correlated with shorter overall survival (p = 0.04) in the entire group. The expression levels of HOTAIR, SNAIL, SLUG and CD133 were similar in defined EC molecular subtypes.
Conclusions
Our data do not confirm the role of HOTAIR in EMT-mediated CSC formation in EC. Neither does the diversity of EC molecular subtypes influence these processes. But HOTAIR expression could serve as an independent prognostic factor in EC. The clinical importance of the above discoveries requires further studies.
Introduction
Endometrial cancer (EC) is the most common gynecological malignancy in developed countries, with 10-20 per 100,000 women diagnosed annually with the disease (1). Its progression can be stimulated by estrogens, with simultaneous progestogen deficiency. Recent studies have proven the crucial importance of EC molecular subtype classification, distinguishing ER/PR+HER2- patients as a group with the most favorable outcome and the ER-PR-HER2+ group as having the poorest outcomes. The revealed diversity has an impact on prognosis and clinicopathological and molecular characteristics (2).
The epithelial-mesenchymal transition (EMT) is the process of converting tightly connected epithelial cells into highly mobile mesenchymal cells (3). It has been shown to play a key role in metastasis formation, providing tumor cells with the molecular features of an invasive and motile phenotype, such as cell polarity loss, a decrease in epithelial marker expression (e.g., E-cadherin), an increase in mesenchymal marker expression (e.g., N-cadherin) and secretion of extracellular matrix metalloproteinases (3). EMT is partially achieved through activation of transcription factors, such as SNAIL (SNAI1) or SLUG (SNAI2) (4).
Cancer stem cells (CSCs) are a subpopulation of cells within the tumor able to initiate and sustain tumor growth. They have self-renewal and differentiation potential, giving rise to a wide spectrum of primary tumor cells (5). Growing evidence indicates that EMT induces stem cell activity (5-6-7). The dependence of CSCs formation on EMT has been confirmed in many cancers, yet data about this relationship in EC are scarce. The only study conducted on primary tumor samples did not show a correlation between EMT and CSC markers (4), yet the reliability of CD133 as a marker for CSCs in EC is currently under discussion (1, 4).
Long noncoding RNAs (lncRNAs) are transcripts longer than 200 nucleotides, lacking protein coding potential. They are engaged in numerous processes, from morphological development control, to cell function regulation (8). HOX antisense intergenic RNA (HOTAIR) is a lncRNA regulating HOX genes. It can retarget the polycomb repressive complex 2 (PRC2) protein to a pattern resembling embryonic fibroblast occupancy. PRC2 catalyzes trimethylation of histone H3 at lysine 27, leading to epigenetic silencing of metastasis suppressor genes (9). HOTAIR is overexpressed in many cancers (10, 11). Colorectal cancer and breast cancer (BC) cell line studies show that HOTAIR triggers EMT, thus causing CSC formation (12). HOTAIR overexpression increases expression of SNAIL. PRC2 represses E-cadherin expression (10). HOTAIR and PRC2-mediated regulation may promote chromatin global state favoring EMT and CSC formation (12).
HOTAIR's role in EMT and CSC formation in EC remains unknown. Although the possible link between EMT and CSC markers was not found in EC, the phenomenon itself might be affected by any meaningful molecular subtype diversity. Thus we aimed to investigate the possible links between HOTAIR, EMT and CSC-related markers in different molecular subtypes of EC, and relate them to clinicopathological data and patients’ outcomes.
Materials and methods
Clinical Samples
The study group included 156 consecutive, stage I-IV EC patients (Tab. I), operated on in the Department of Gynaecology, Gynaecological Oncology and Gynaecological Endocrinology (Medical University of Gdańsk) between 2000 and 2010. Patients were treated surgically, with the potential option of radiotherapy or chemotherapy. The inclusion criteria were operable EC confirmed histologically and a signed consent form. The study was accepted by the ethics committee of the Medical University of Gdańsk.
Patient characteristics by tumor subtype
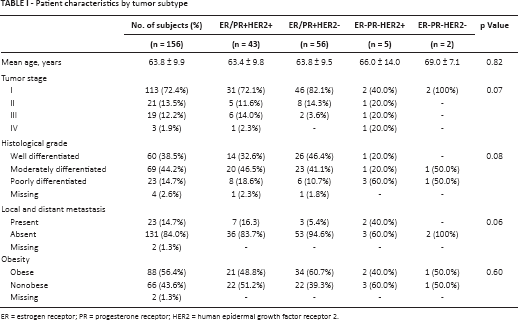
ER = estrogen receptor; PR = progesterone receptor; HER2 = human epidermal growth factor receptor 2.
The samples for molecular analysis were obtained by surgical excision from the tumor core before any systemic treatment. The samples were frozen immediately after collection and stored in -80°C. They were kept on ice during transport. The samples for immunohistochemistry (IHC) analysis were formalin fixed and paraffin embedded.
The study group included patients with stages from IA to IVB, according to International Federation of Gynecology and Obstetrics (FIGO) criteria (13). The median age was 63 (range 30-87 years). The last follow-up data were collected in May 2013. Patients with body mass index higher than 30 were classified as obese (14).
Hormone receptors status and HER2 expression was previously determined by IHC for 107 patients (68.6%) (2). HOTAIR, SNAIL, SLUG and CD133 expression were measured for the entire group.
ER, PR and HER2 Expression Analysis
Estrogen receptor (ER), progesterone receptor (PR) and human epidermal growth factor receptor (HER2) status were determined by IHC on tissue microarrays using the following antibodies: ER with SP1 clone (Roche); PR with 1E2 clone (Roche); and HER2 with 4B5 clone (Roche). PR and ER evaluation of the nuclear staining was based on Allred score (positivity cutoff: ≥4), while HER2 status was determined based on the HercepTest™ criteria (positivity cutoff: ≥2), as reported earlier (2, 15).
HOTAIR, SNAIL, SLUG and CD133 Expression Analysis
Rna Isolation
For RNA isolation, tissue samples (25 mg per sample) were homogenized (1 minute, 6,000 rpm) using a MagNALyser (Roche). RNA was isolated with an All Prep DNA/RNA Mini Kit (Qiagen), according to the manufacturer's protocol. After the isolation, the RNA concentration was measured with an Agilent 2100 Bioanalyzer (Agilent Technologies). RNA samples were stored in -80°C. RNA was reverse transcribed to cDNA with a Transcriptor First Strand cDNA Synthesis Kit (Roche) according to the manufacturer's protocol, using random hexamer primers. The total amount of RNA per reaction was 1,000 ng.
Genes and Starters
SNAIL, SLUG and CD133 expression analyses had been performed previously (4).
HOTAIR transcript variant 1 expression analyses were done using Gene Expression Assay (Hs03296630_m1, RefSeq NR_047517.1, 64-bp amplicon, exons 1 and 2 boundary; Applied Biosystems). The following TaqMan® Gene Expression Assays were chosen as reference genes: ACTB (Hs99999903_m1, RefSeq NM_001101.3, 171-bp amplicon at exon 1; Applied Biosystems) and GAPDH transcript variant 1 (Hs99999905_m1, RefSeq NM_002046.4, 122-bp amplicon at exon 3, Applied Biosystems).
Quantitative Reverse Transcription - Quantitative Polymerase Chain Reaction Analysis
SNAIL, SLUG and CD133 measurements were performed as reported previously (4). HOTAIR analysis was performed using a CFX96 thermocycler (Bio-Rad) in 96-well plates in a reaction volume of 20 µL. The reaction mixture included 100 ng of cDNA, 2x TaqMan® Universal PCR Master Mix (Applied Biosystems) and 20x TaqMan® Gene Expression Assay (Applied Biosystems). The thermal profile of quantitative polymerase chain reaction (qPCR) was as follows: (i) 50.0°C for 2 minutes; (ii) 95.0°C for 10 minutes; (iii) 95.0°C for 15 seconds; (iv) 60.0°C for 1 minute; with steps 3 and 4 repeated 45 times. Negative control (no reverse transcriptase) and a calibrator (cDNA derived from healthy endometrium) were always included. Each qPCR reaction was performed twice. Gene expression analysis was done using qbase+ software (Biogazelle NV).
Statistical Analysis
All calculations were done using Statistica 10.0 software (Statsoft). Normality of the data was checked by Shapiro-Wilk test. Comparison of the tumor subtypes with clinicopathological data of the patients was done using crosstabs statistics and Pearson's chi-square test and Kruskal-Wallis and Mann-Whitney tests. Genes’ expression correlations were calculated using Spearman correlation. For CD133, SNAIL, SLUG and HOTAIR, median expression values were used as a cutoffs, as previously described (4, 16, 17). Survival analysis was done with the Kaplan-Meier estimator and verified with the F-Cox test. The end point for the study was overall survival. Statistical significance was assumed with a p value ≤0.05.
Results
Hormone Receptor Status and Investigated Transcripts
Positive status of ER, PR and HER2 occurred in 90.6%, 84.9% and 44.9% of cases, respectively. The number of patients with high HOTAIR, SNAIL, SLUG and CD133 expression was 35.9%, 50%, 50% and 50%, respectively.
Molecular Characteristics of the Elucidated Subtypes
Molecular subtypes, defined as ER/PR+HER2+, ER/PR+HER2-, ER-PR-HER2+ and ER-PR-HER2-, occurred in 40.2%, 52.3%, 4.7% and 1.9% of cases, respectively.
Average age of the patients was lowest in the ER/PR+ groups. The ER-PR-HER2+ group had the highest percentage of stage III and IV cases. Similarly, histological examination revealed the poorest differentiation occurring in the ER-PR-HER2+ group. Local and distant metastasis status analysis showed the highest metastasis percentage in the ER-PR-HER2+ group. The ER-PR-HER2+ group was the least obese. In contrast, tumor stage, grade and metastasis status were the most favorable for the ER/PR+HER2- group (Tab. I).
Overall Survival by HOTAIR Status
Analysis of overall survival by HOTAIR status did not reveal any significant differences between molecular subtypes. Hence, survival probability according to HOTAIR status was determined for the entire group of patients (Fig. 1). Patients with high HOTAIR expression had shorter overall survival when compared with patients exhibiting low HOTAIR expression level (p = 0.04).
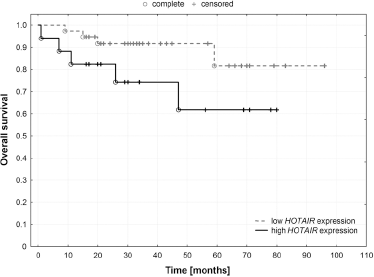
Overall survival by HOTAIR status (p = 0.04).
HOTAIR Status in Relation to Clinicopathological Characteristics
No significant correlations between HOTAIR status and clinicopathological characteristics were found (Tab. II).
HOTAIR status in relation to patient characteristics
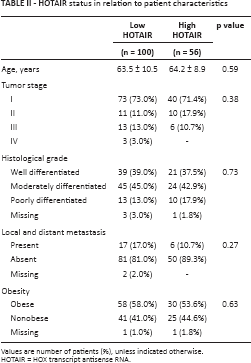
Values are number of patients (%), unless indicated otherwise.
HOTAIR = HOX transcript antisense RNA.
Molecular Characteristics by Tumor Subtype
Analysis of HOTAIR, SNAIL, SLUG and CD133 gene expression correlations revealed strong associations between SNAIL and SLUG in the ER/PR+ groups. No other correlations were found (Tab. III).
Gene expression correlations within molecular subtypes
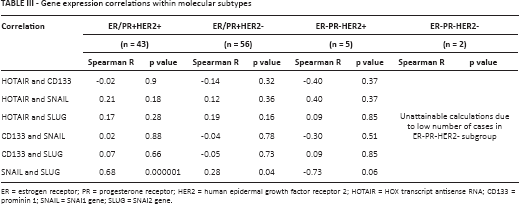
ER = estrogen receptor; PR = progesterone receptor; HER2 = human epidermal growth factor receptor 2; HOTAIR = HOX transcript antisense RNA; CD133 = prominin 1; SNAIL = SNAI1 gene; SLUG = SNAI2 gene.
Analysis of the relationship between investigated gene expression status (based on cutoff values) and EC molecular subtypes did not reveal any statistically significant differences (Fig. 2).
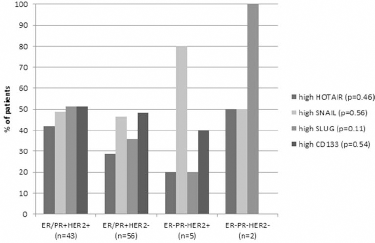
Molecular characteristics by tumor subtype.
Discussion
LncRNA HOTAIR, overexpressed in many cancers (10, 11) and crucial for EMT-mediated CSC formation in breast and colon cancer (12), remains poorly analyzed in EC. The first study concerning the EMT dependency of CSCs in EC did not reveal any relationship (4), yet the issue of CSC marker selection is still discussed. Furthermore, ER/PR/HER2 status might affect the possible links. We have shown recently that 4 molecular subtypes based on hormone receptor expression may be distinguished in EC patients. The subtypes were associated with different clinicopathological characteristics and patient outcomes, with ER-PR-HER2+ exhibiting the least favorable features (2).
The survival analysis by HOTAIR status did not reveal any significant differences between molecular subtypes, suggesting the lack of the dependencies between HOTAIR and ER/PR/HER2 status. However, correlations may be masked by their complex nature. BC cell line studies have shown that ERs bind HOTAIR promoter in an estradiol-dependent manner, stimulating its expression (18). PRC2 subunit EZH2 correlated inversely with ER and PR status (19). Therefore, the presence of ER and PR was associated with lower PRC2 accessibility for HOTAIR. Analysis of stomach cancer clinical samples has revealed that HOTAIR binding to microRNA repressors of HER2 might increase its expression (20). The limitation of our study was the low number of patients in the 2 subgroups.
HOTAIR expression deregulation was shown for the first time in BC, where its level was higher in primary tumors than in close nontumorigenic tissues (9). Since its emergence in BC, elevated expression of HOTAIR has been reported in 16 types of malignancies (21). The spectrum ranges from carcinomas of epithelial origin to gastrointestinal stromal tumors of stromal origin. Increased HOTAIR expression has also been reported in prostate cancer cell lines. HOTAIR expression knockdown caused a reduction of prostate cancer cell proliferation in vitro and in vivo (22). HOTAIR's carcinogenic nature has also been proven for colorectal, liver, pancreatic and other cancers (10, 23). Survival analysis of HOTAIR status conducted independently of molecular subtype disclosed worse prognosis for patients with high HOTAIR expression, which is in accord with the work of He et al (11), who also showed a correlation of high HOTAIR expression with poor overall survival in EC patients. A recent meta-analysis aimed at assessment of the prognostic role of HOTAIR in estrogen-dependent malignancies found that HOTAIR expression was associated with poor overall survival, especially in non-BC patients (i.e., EC and cervical and ovarian cancer) (24). The study of He et al reported that HOTAIR expression was higher in cancer samples when compared with normal tissue, and HOTAIR level correlated with tumor grade and lymph node metastasis (11). In contrast, our work did not reveal any significant correlations with occurrence of metastasis and other clinicopathological data. This discrepancy might be partially explained by the different populations studied (Asian in the study of He et al, and white in our study), as it was recently postulated that HOTAIR may be regarded as a risk factor in Asian populations. A significant association between HOTAIR expression and overall survival in Asian cancer patients was found but not in whites (24). Moreover, both studies included a relatively small number of samples. To confirm the significance of HOTAIR in EC, a study including a larger number of samples is required.
As HOTAIR regulates the methylation of a large set of genes, it was considered a key factor controlling the signaling pathway associated with EMT and stem cell phenotype in colon and breast cancer cell lines (12). It was confirmed in vitro that elevated HOTAIR expression can induce SNAIL expression in BC (9). Furthermore, HOTAIR decreases E-cadherin expression, facilitating breakage of intercellular connections and CSC formation (12). In BC, coexpression of ER and PR led to the phosphorylation of a transcription factor, STAT3, inducing the expression of EMT and CSC markers. It was also proven that STAT3 induced EMT-associated invasive cancer phenotype and treatment resistance in lung and other cancers. Considering features acquired by cells during EMT, this process is perceived as crucial in CSC formation (25). Despite this, our study did not reveal any correlations between HOTAIR expression level and EMT and CSC markers for any of the EC molecular subtypes or for entire group of patients either (data not shown). The expression levels of these genes were not significantly different for any patient group. These results suggest that HOTAIR does not play an important role in EMT triggering and CSC formation in any of the EC molecular subtypes. Perhaps the described phenomenon could be reanalyzed with different markers.
Altogether, our data show for the first time that HOTAIR does not play any important role in EMT-mediated CSC formation in EC. Nevertheless, HOTAIR expression alone could serve as a prognostic factor in EC. The EC molecular subtypes apparently do not influence these processes. The research performed here did not show any particular correlations between HOTAIR status and molecular subtypes in EC. The clinical importance of the above discoveries requires further studies.
Footnotes
Financial support: This research was financed by the Polish Ministry of Science and Higher Education (grant N407571538). Research was part of the project The role of non-coding RNA in chromatin methylation and invasiveness of prostate cancer – participation in [the] Undergraduate Student Caucus and Poster Competition and prestigious conference American Association for Cancer Research annual meeting, initiated by the European Union as part of the work supported by the European Regional Development Fund.
Conflict of interest: The authors declare that they have no conflicts of interest.
Meeting presentation: This work was presented to the Undergraduate Student Caucus and Poster Competition at the American Association for Cancer Research annual meeting, San Diego, California, 5 April 2015.
