Abstract
Long noncoding RNA (LncRNA) homeotic genes (HOX) transcript antisense RNA (HOTAIR) has been reported to play a vital role in various cancers. It has been found that HOTAIR was upregulated in non–small cell lung cancer (NSCLC) and involved in cell invasion and metastasis. The aberrant expression of HOTAIR is expected to serve as a potential biomarker for patients with NSCLC. Our aim in this study was to detect the plasma levels of HOTAIR and further evaluate its diagnostic value for NSCLC. The levels of HOTAIR were measured in 105 patients with NSCLC and 80 healthy controls by quantitative real-time polymerase chain reaction. The results indicated that plasma HOTAIR levels were higher in NSCLC than in healthy controls. Besides, plasma HOTAIR levels were associated with histology subtype (P = .039) and tumor-node-metastasis stage (P = .022). The ROC curves showed that plasma HOTAIR has high diagnostic accuracy for NSCLC, and the area under curve (AUC) for NSCLC versus healthy was 0.791 (95% CI: 0.727-0.855) which was higher than carcinoembryonic antigen (CEA) (AUC = 0.737, 95% CI: 0.666-0.808). Moreover, the combination of HOTAIR and CEA could provide a more accurate diagnosis than HOTAIR or CEA alone (AUC = 0.841, 95% CI: 0.783-0.898). Plasma HOTAIR levels were significantly lower in postoperative samples than in preoperative samples. Plasma HOTAIR could serve as a promising biomarker for diagnosing and monitoring NSCLC.
Introduction
Cancer statistics in China reported that China had an estimated 4 292 000 new cancer cases and 2 814 000 cancer deaths in 2015. 1 The leading causes of cancer death was lung cancer in those aged 40 years or older. 2 Non–small cell lung cancer (NSCLC) is the main type of lung cancer, which included squamous cell carcinoma, adenocarcinoma, large cell carcinoma, and several other types. 3 Despite impressive development in treatment of patients with NSCLC, the 5-year survival rate is still unfavorable. 2 The screen strategy of lung cancer has been based on chest radiography and traditional biomarkers, but the sensitivity is far from satisfaction. 4,5 Therefore, it is important to identify more sensitive biomarkers to complement and improve the current NSCLC screening methods.
Long noncoding RNA (LncRNA) is a type of RNA molecule that is longer than 200 nt without protein-coding capacity. 6 LncRNAs could regulate many biological processes at transcriptional, posttranscriptional, and epigenetic levels. 7 Accumulating studies indicated that LncRNAs play a vital role in the initiation and progression of NSCLC. 8 –10 For instance, LncRNA metastasis-associated lung adenocarcinoma transcript 1 (MALAT1) could promote tumor growth and metastasis and associated with unfavorable prognosis in NSCLC. 11 It has been reported that circulating LncRNAs were derived from tumor cells and could reflect the pathological and physiological change of patients with cancer. 12 Moreover, multiple studies confirmed that LncRNA was stable in plasma. 13 –15 Therefore, a number of circulating LncRNAs were identified as reliable biomarkers for patients with cancer. 16,17 For instance, plasma POU class 3 homeobox 3 (POU3F3) could serve as an ideal biomarker for the diagnosis of esophageal squamous cell carcinoma. 12 However, few studies investigated circulating LncRNAs for the diagnosis of patients with NSCLC.
HOX transcript antisense RNA (HOTAIR) is located within the Homeobox C (HOXC) gene cluster on chromosome 12 and is coexpressed with HOXC genes. It was originally identified as an LncRNA that interacted with polycomb repressive complex 2 (PRC2) through its 5′ domains to repress transcription of homeobox gene D cluster (HOXD) genes. 18 It has been found that HOTAIR was dysregulated in various cancers, such as breast cancer, 19 gastric cancer, 20 and hepatocellular carcinoma. 21 Nakagawa et al. discovered that HOTAIR was upregulated in NSCLC tissue and correlated with worse prognosis. 22 Besides, circulating HOTAIR has been characterized as a diagnostic biomarker in breast cancer and colorectal cancer. 23,24 These results indicated that circulating HOTAIR might serve as a potential biomarker for NSCLC.
In this study, we investigated the expression pattern of HOTAIR in NSCLC and analyzed the correlation between HOTAIR and clinicopathological characteristics. Then, we further evaluate the diagnostic efficiency of circulating HOTAIR for patients with NSCLC.
Materials and Methods
Sample Collection
Whole blood samples of 105 patients with NSCLC (80 men and 25 women; mean age 61 ± 8) and 80 healthy controls were included before any anticancer treatment between January 2016 and November 2016. All patients were included according to medical or pathology reports. Then we collected 46 paired postoperative whole blood samples after approximately 1 month. Plasma was isolated from whole blood samples with a 2-step centrifugation protocol (2000g for 5 minutes at 4°C, 12 000g for 5 minutes at 4°C). All plasma samples were stored at −80°C until RNA extraction.
Ethical Approval
All patients signed their informed consent before they participated in the study. The study was conducted according to the Declaration of Helsinki, and the study was approved by the ethics committee of Zhongnan Hospital of Wuhan University (Ethical Approval No. 2013059).
Data Extraction
The stage of patients with NSCLC was determined according to the eighth edition of tumor node metastasis (TNM) classification for lung cancer (UICC). Clinical data were collected, including gender, age, histology subtype, tumor size, lymph node metastasis, histological grade and traditional biomarkers data.
RNA Extraction and Reverse Transcription
The total RNA was extracted from 300 μL plasma samples by using blood/liquid sample total RNA Rapid Extraction kit (BioTeKe, Beijing, China). RNA was then reverse transcribed to complementary DNA (cDNA) by using PrimeScript RT reagent kit (Takara: RR036A). Reverse transcription steps were as follow: 37°C for 15 minutes and 85°C for 5 seconds. All cDNA samples were stored at −80°C before real-time PCR analysis.
Quantitative Real-Time PCR Analysis
Quantitative real-time polymerase chain reaction (qRT-PCR) was conducted to detect the levels of HOTAIR transcripts using SYBR-Green I Premix Ex Taq in a 20-µL reaction volume, which contained 10 µL of SYBR-Green master PCR mix, 0.8 µL each of forward and reverse primers, 2 µL of diluted cDNA template, and appropriate amounts of sterile distilled water. The cycling conditions were initial denaturation at 95°C for 5 minutes; 40 cycles of denaturation at 95°C for 30 seconds, annealing at 62.5°C for 30 seconds, and elongation at 72°C for 30 seconds. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was chosen as internal reference gene to normalize HOTAIR expression. There was no significant difference between GAPDH expression in patients with NSCLC patients and in healthy controls (P = .340). The sequence of primers were as follows: HOTAIR (forward: 5′-GTGATGTCCCCAGTGATCCA-3′ and reverse: 5′-CCTTCGCTTCCTTGTAATTC-3′); GAPDH (forward: 5′-GGTCTCCTCTGACTTC-AACA-3′ and reverse: 5′-GTGAGGGTCTCTCTCTTCCT-3′). The cycle threshold (Ct) is defined as the number of cycles required for the fluorescent signal to cross the threshold in qRT-PCR. The relative expression was calculated using the comparative cycle threshold (Ct) method (2− ΔΔCt). ΔCt = Ct HOTAIR − Ct GAPDH. All samples were analyzed in duplicate with no template controls included.
Statistical Analysis
All statistical analyses were performed using SPSS software package version 19.0 (SPSS, Chicago, Illinois, USA). All figures were drawn by GraphPad Prism 5.0 software (GraphPad Software, La Jolla, California). The paired-sample t test was used to compare differences in HOTAIR expression in paired tissues and paired plasma samples before and after surgery. The independent t test was used for the comparison between NSCLC plasma samples and healthy control plasma samples. Correlations between the expression level of plasma and tissue were analyzed using the Spearman correlation. Receiver–operating characteristic (ROC) curves and the area under the curve (AUC) were applied to evaluate the diagnostic value. P < .05 was considered to be statistically significant.
Results
Validity of Detection Method
Intra-assay and inter-assay coefficient of variation (CV) of Ct value were used to evaluate the repeatability and precision of the qRT-PCR results. The intra-assay CV and inter-assay CV of HOTAIR and GAPDH were all <5% (Table 1).
Intra-assay and Inter-assay Coefficient of Variation (CV) of HOTAIR and GAPDH in the Two Groups.
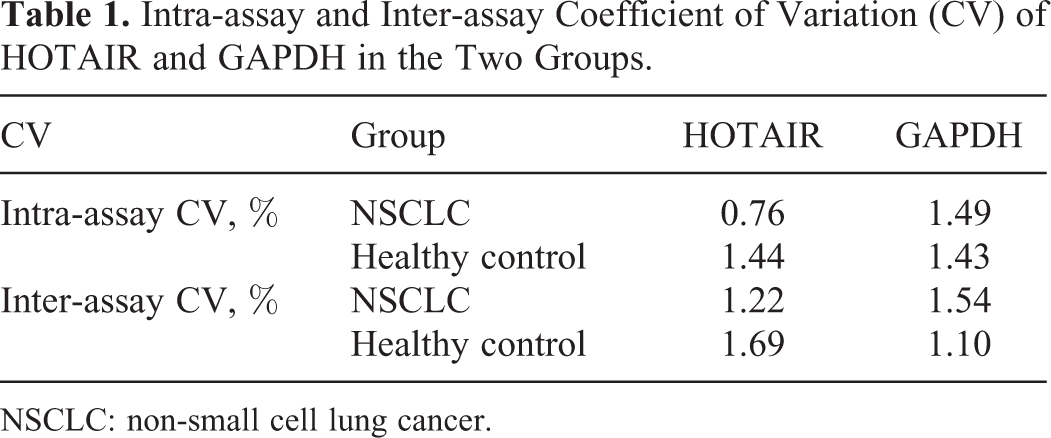
NSCLC: non-small cell lung cancer.
Test-Scale Analysis of HOTAIR Expression Levels in Tissues and Plasma
Several studies found that HOTAIR expression levels were upregulated in NSCLC. 25,26 To validate the expression pattern of HOTAIR in NSCLC, we used qRT-PCR to detect the expression levels of HOTAIR in 20 paired NSCLC tissues and corresponding adjacent normal tissues. The result showed HOTAIR was significantly higher in NSCLC tissues than in the corresponding adjacent normal tissues (P = .007; Figure 1A). Then, we investigated whether HOTAIR could be detected in plasma. Twenty-four paired NSCLC patients and healthy controls plasma were measured by qRT-PCR. Elevated HOTAIR levels were observed in NSCLC plasma (P = .011; Figure 1B).
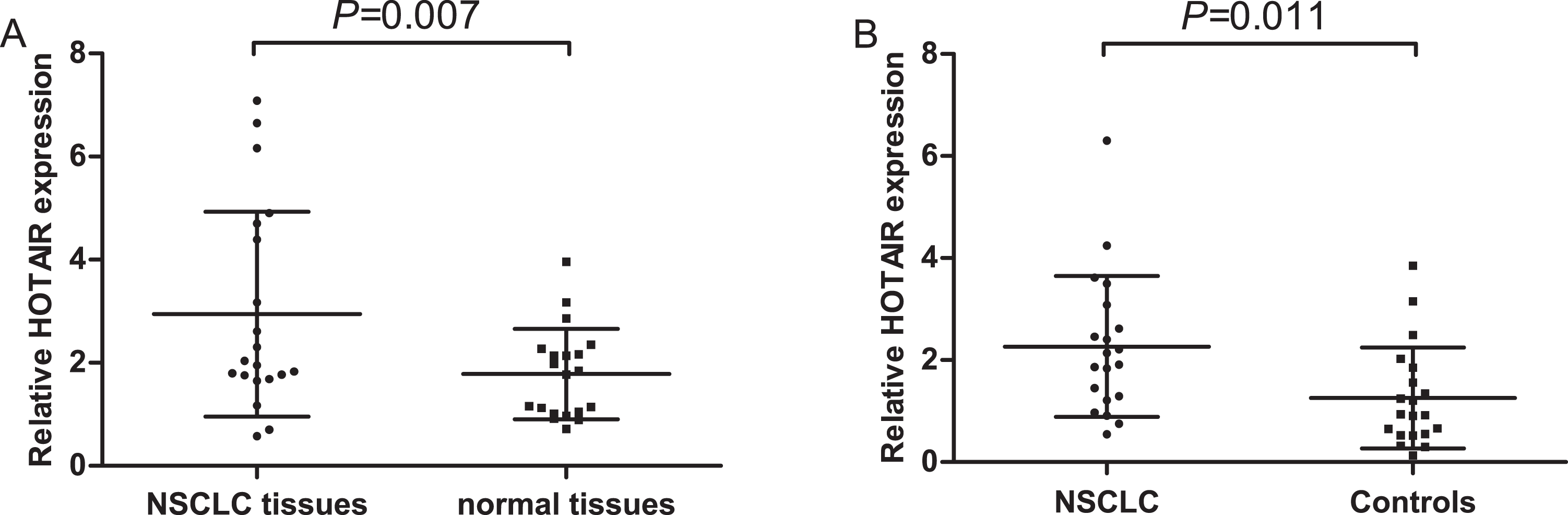
Test-scale analysis of HOTAIR expression levels in tissues (A) and plasma (B).
Analysis of Stability of HOTAIR in Plasma
To analysis the stability of circulating HOTAIR, four healthy plasma samples were left under harsh conditions such as incubation at room temperature for 0, 6, 12, and 24 hours or repeated freeze–thaw cycles. However, no obvious alterations were observed in plasma HOTAIR levels under both of the above conditions (P>0.05, Figure 2A, B). These results demonstrated that HOTAIR remained relatively stable in plasma, which could laid the foundation for identification of circulating HOTAIR as a reliable biomarker.
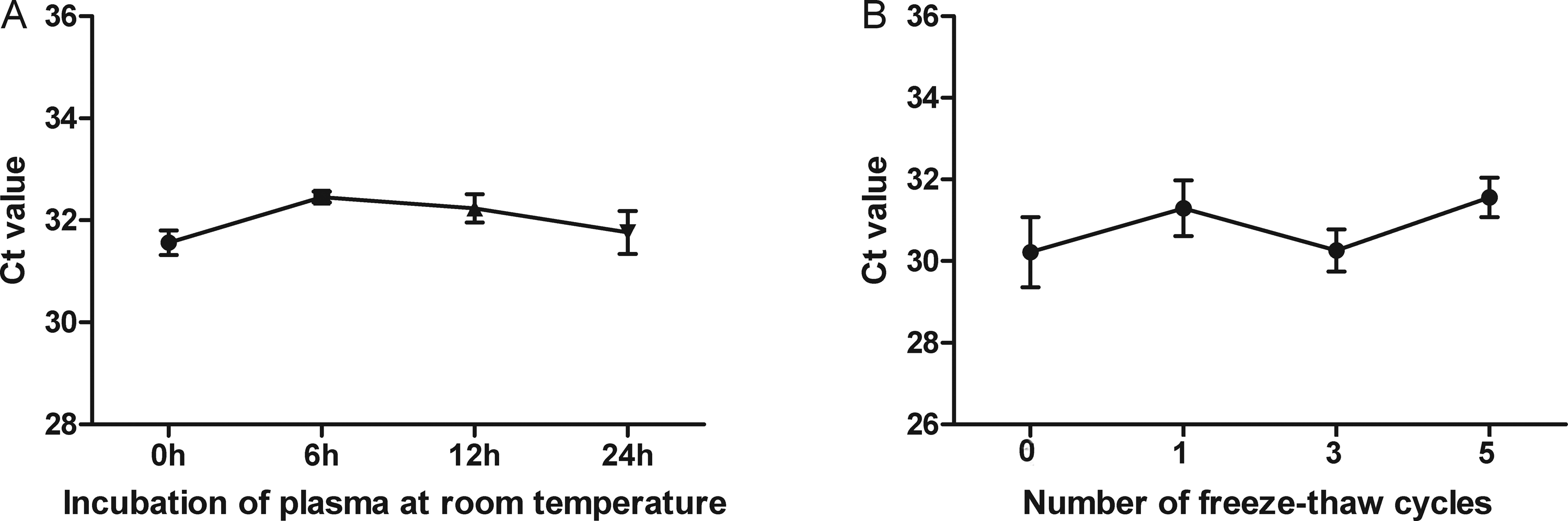
The stability of HOTAIR plasma in harsh environments. Incubation of plasma at room temperature (A) and the freeze–thawing processes (B).
Large-scale Analysis of HOTAIR Expression Levels in NSCLC Patients and Healthy Controls Plasma
To evaluate whether HOTAIR could serve as a circulating biomarker for NSCLC patients, we detected the levels of HOTAIR in 105 NSCLC patients plasma and 80 healthy controls plasma. As shown in Figure 3, plasma HOTAIR levels were significantly increased in NSCLC patients (P<0.001). We further analyzed the correlation between plasma HOTAIR levels and clinicopathologic features (Table 2). Plasma HOTAIR levels were strongly correlated with histology subtype (P=0.039) and TNM stage (P = .022). Plasma HOTAIR levels was significantly higher in lung squamous cell cancer (SCC) than adenocarcinoma (ADC). However, there was no significant association between plasma HOTAIR levels and clinicopathologic features.

Large-scale analysis of HOTAIR expression levels in NSCLC patients and healthy controls plasma
Correlation Between Clinicopathologic Features and HOTAIR Levels in NSCLC Patients.

Abbreviation: TNM, tumor-node-metastasis.
Diagnostic Value of Plasma HOTAIR for NSCLC
To assess the potentiality of clinical application of plasma HOTAIR, ROC curves were employed to evaluate the diagnostic value of HOTAIR and CEA. The AUC of HOTAIR was 0.806 (95% CI: 0.745-0.868, Figure 4A) for distinguishing NSCLC patients from healthy controls, and the optimal sensitivity and specificity were 76.2% and 71.9%, respectively. The ROC curves indicated that there was strong separation between SCC patients and healthy controls, and the AUC of HOTAIR was 0.827 (95% CI: 0.745-0.909, Figure 4B). While the AUC of HOTAIR for ADC patients was 0.795 (95% CI: 0.717-0.873, Figure 4C). To further understand the potential value of HOTAIR as a clinical biomarker for diagnosing NSCLC. We compared the diagnostic value of HOTAIR to CEA which is one of common NSCLC biomarkers. 4 The results suggested plasma HOTAIR had a higher diagnostic efficiency than CEA (AUC=0.737, 95% CI: 0.666-0.808). Moreover, the combination of HOTAIR and CEA could provide a more powerful diagnostic efficiency than HOTAIR or CEA alone (AUC=0.841, 95% CI: 0.783-0.898, Figure 4D). These results showed that HOTAIR could serve as a potential biomarker for diagnosing NSCLC.

Diagnostic value of plasma HOTAIR for NSCLC. (A)Comparison of all NSCLC patients with healthy controls; (B)Comparison of lung squamous cell cancer patients with healthy controls; (C)Comparison of lung adenocarcinoma patients with healthy controls; (D)Combination of HOTAIR and CEA.
Evaluation of the Use of HOTAIR for Monitoring NSCLC Patients Dynamics
We performed two step to investigate whether plasma HOTAIR levels could monitor NSCLC patients dynamics. Firstly, qRT-PCR was used to measure the expression levels in 24 paired NSCLC tumor tissues and plasma samples which were from the same patients. The association of HOTAIR levels between the two groups was analyzed. Plasma HOTAIR levels has a significant association with tissue levels (r=0.552, P=0.01, Figure 5A).
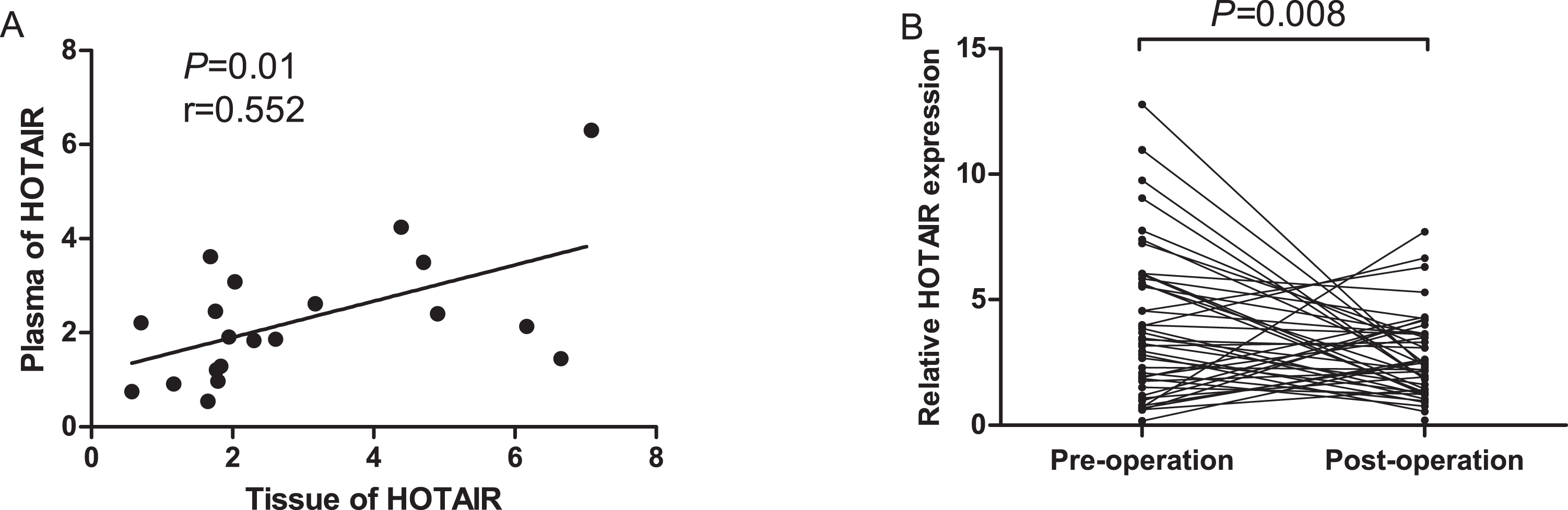
Dynamic monitoring of plasma HOTAIR levels in NSCLC patients. (A)Correlation of HOTAIR expression levels in tumor and plasma samples; (B)Comparison of plasma HOTAIR levels between pre- and post-operative samples.
Then, the levels of HOTAIR were analyzed in 45 paired pre- and postoperative plasma samples from NSCLC patients. The results showed that plasma HOTAIR levels were significantly lower in post-operative samples than in pre-operative samples (P=0.008, Figure 5B). These results demonstrated that plasma HOTAIR levels may reflect tumor dynamics of NSCLC patients.
Discussion
The LncRNAs are a class of RNA molecule longer than 200 nucleotides in length which are not translated into proteins. 27 LncRNA can regulate gene expression in different levels, including genomic imprinting, chromatin modification, transcription and post-transcriptional processes. 28 Multiple lines of study have demonstrated that LncRNAs play a vital role in many biological processes, especially in tumor biology. 29 Several circulating lncRNAs have been characterized as potential tumor markers for cancers. 13,30,31 However, there are still few studies reported circulating lncRNAs in diagnosis of NSCLC patients. In this study, we set out to investigate whether circulating HOTAIR could serve as a potential biomarker for NSCLC.
Nakagawa et al. found HOTAIR was associated patients with advanced stage, lymph node metastasis and worse disease free survival. Moreover, brain metastases samples showed strongly higher HOTAIR expression compared to primary cancer tissues. 22 It has reported that HOTAIR is a direct target of hypoxia-inducible factor 1 alpha (HIF-1α) through interaction with putative hypoxia response elements (HREs) in the upstream region of HOTAIR in NSCLC cells. Under hypoxic conditions, HOTAIR could enhance cancer cell proliferation, migration and invasion in NSCLC cells. 32 Recently, Zhai et al. found a regulatory role of p53 in HOTAIR expression and HOTAIR could regulate the expression of p53 via affecting the modification of H3K27me3 with a negative feedback loop. 33 Based on the above studies, the aberrant expression of HOTAIR is expected to serve as a diagnostic biomarker for NSCLC patients.
In this study, our aim was to detect the plasma levels of HOTAIR and further evaluate its diagnostic value for NSCLC. First, we confirmed the repeatability and precision of the qRT-PCR method by the intra-assay CV and inter-assay CV. Then, qRT-PCR was used to measure the levels of HOTAIR in NSCLC patients and healthy controls. We found plasma HOTAIR levels were significantly higher in NSCLC patients than healthy controls and its levels were correlated with histology subtype and TNM stage. Our results were in accordance with previous studies, which suggested that HOTAIR may play an oncogenic role in NSCLC. Furthermore, SCC patients has a higher levels of HOTAIR in plasma, which indicated circulating HOTAIR may has a high specificity for diagnosing SCC patients.
We further investigated diagnostic value of plasma HOTAIR. ROC curves showed HOTAIR has a high diagnostic power for diagnosis of NSCLC patients (AUC=0.806, 95% CI: 0.745-0.868), which was higher than CEA. Furthermore, the combination of HOTAIR and CEA could provide a more powerful diagnostic efficiency than HOTAIR or CEA alone. Previously, circulating microRNAs (miRNAs) have been shown to be stable and could serve as reliable biomarkers for cancers. One study reported that circulating miR-182, miR-183, miR-210 and miR-126 were useful markers for NSCLC diagnosis, and the AUC of miRNAs were 0.734, 0.626, 0.616 and 0.793, respectively. 34 Compared to these miRNAs, HOTAIR could provide more powerful diagnostic efficiency. These results suggested circulating HOTAIR may serve as promising biomarker for NSCLC patients.
Due to the high expression of HOTAIR in SCC patients, we investigated the diagnostic value of HOTAIR for different type of lung cancer. There was strong separation between SCC patients and healthy controls, and the AUC of HOTAIR was 0.827 (95% CI: 0.745-0.909). While the AUC of HOTAIR for ADC patients was 0.795 (95% CI: 0.717-0.873). Plasma HOTAIR may serve as a potential histologic type-specific biomarker.
Ultimately, we conducted two analyses to evaluate whether plasma HOTAIR levels could monitor NSCLC dynamics. First, the association of HOTAIR levels between plasma and tissue samples was analyzed. There was a significant correlation between HOTAIR levels of tissue and plasma. Second, the levels of HOTAIR were analyzed in 45 paired pre- and postoperative plasma samples. Plasma HOTAIR levels were significantly decreased after operation. The possible explanation was that circulating lncRNAs were mainly derived from tumor cells, and it would presumably back to normal after removing of tumor. These results indicated plasma HOTAIR levels may reflect tumor dynamics of NSCLC patients.
We investigated circulating HOTAIR in NSCLC for the first time. Plasma HOTAIR levels were significantly higher in NSCLC patients than healthy controls and its levels were correlated with histology subtype and TNM stage. In conclusion, circulating HOTAIR could serve as a promising biomarker for diagnosing and monitoring NSCLC patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Basic Research Program of China (973 Program) (2012CB720605).
