Abstract
Purpose
This study aimed to evaluate the associations between metabolic syndrome (MS) and its components at initial diagnosis and outcomes of breast cancer including triple-negative breast cancer (TNBC) and non-TNBC.
Methods
A cohort of 1,391 patients was reviewed between January 2004 and July 2008 (including 394 TNBC and 855 non-TNBC cases). MS and its components including body mass index (BMI), serum high-density lipoprotein (HDL) and triglycerides (TG) and their relationships with clinical outcomes were analyzed and then compared between groups.
Results
The incidences of MS and its components including BMI, the levels of HDL and TG were not differently distributed between the 2 groups (all p's >0.05). However, more TNBC than non-TNBC patients presented with hypertension and elevated serum glucose (20.3% vs. 14.9% and 16.0% vs. 10.8%, p = 0.018 and p = 0.012, respectively). TNBC patients had poorer 5-year relapse-free survival (RFS) than non-TNBC patients (72.8% vs. 84.2%, p<0.0001). Only in the TNBC group, patients with low high-density lipoprotein (HDL) demonstrated worse RFS and overall survival (OS; p<0.0001). Multivariate analysis identified that low HDL was an independent worse prognostic factor for both RFS (hazard ratio [HR] = 3.266, 95% confidence interval [95%CI], 2.087-5.112, p<0.0001) and OS (HR = 3.071, 95%CI, 1.732-5.445, p<0.0001) in TNBC patients.
Conclusions
Decreased level of HDL may predict worse outcomes both in terms of RFS and OS for TNBC patients but not for non-TNBC patients. Further investigations are warranted to detect the underlying mechanisms.
Introduction
Metabolic syndrome (MS), also referred to as insulin resistance or syndrome X, is a group of metabolic disorders (1). MS comprises central obesity, high body mass index (BMI), insulin resistance, hypertension, hypertriglyceridemia (HTG) and low high-density lipoprotein (HDL) cholesterol. Both MS (2, 3) and each component of MS, such as low HDL (2, 4–6), high triglycerides (TG) (2, 4–7), elevated glucose or type 2 diabetes mellitus (DM) (2, 8–12), obesity (7, 10, 11, 13, 14) or insulin resistance (9, 13, 15, 16) have been demonstrated to increase the risk of breast cancer (BC) and tumor recurrence irrespective of race (2–16).
However, the clinical significance of these findings on triple-negative breast cancer (TNBC) remains very limited and inconsistent (17–19–20). For example, a 2008 study examining 620 predominantly white women in rural Appalachia, 117 of whom had TNBC, reported a significant association between obesity and incidence of TNBC (18). However, the Carolina Breast Cancer Study reported no elevated prevalence of type 2 DM in TNBC compared with other BC subtypes (17). In addition, studies about the potential influence of MS and its components on recurrence and mortality of TNBC has not been explicitly performed, But as we know now, TNBCs display highly aggressive behavior and have a worse prognosis with higher visceral relapse rates compared with non-TNBCs (21, 22). No optimal clinical prognostic factors have been identified yet for this special subtype. MS components have been implicated in survival of TNBC based on some reports in the literature. Certain drugs, which alter components of MS, such as metformin and beta-blockers, have been shown to change the biologic feature of TNBC tumors (23–25). One study of a large series of cases suggests that beta-blocker use can improve relapse-free survival (RFS) of TNBC patients (24). The other preclinical study showed that metformin has a unique anticancer activity against TNBC through inhibiting cell proliferation and inducing apoptosis, both in vitro and in vivo (25). So it is supposed that the components of MS, such as low HDL or hypertriglyceridemia, also may alter the prognosis of the disease and contribute to inferior outcomes in TNBC patients.
To our knowledge, the roles of components of MS, especially HDL and TG, on BCs by estrogen/progesterone receptor status are not yet well studied. Therefore, we investigated the possible correlation of BC outcomes, especially for TNBC, with components of MS, such as HDL and TG, in a large series of Chinese BC patients.
Patients and Methods
Patient Selection
A consecutive cohort of 1,391 BC patients (394 TNBC and 855 non-TNBC) diagnosed between 1 January 2004 and 30 July 2008 at the Cancer Hospital and Institute, Chinese Academy of Medical Sciences, were retrospectively reviewed. In addition, 142 patients were excluded from the study for exclusion criteria. The exclusion criteria were as follows: (a) pure in situ carcinoma (n = 3); (b) without detailed information of MS (n = 57); (c) with another malignant tumor concurrently, except basal cell carcinoma (n = 7); (d) bilateral BC (n = 7); (e) metastatic disease at primary diagnosis (n = 4); or (f) loss to follow-up (n = 64).
Pathology Confirmation
Tumors with ≥1% nuclear-stained cells were considered positive for estrogen receptor (ER) and progesterone receptor (PR) according to the American Society of Clinical Oncology–College of American Pathologists (ASCO/CAP) guidelines (26). Human epidermal growth factor receptor 2 (HER2) status was evaluated by immunohistochemistry (IHC) or by FISH (27). HER2-negative was defined as negative receptor expression on IHC staining (membranous staining in <10% of cells) or negative gene amplification found on FISH. A gene copy to CEP-17 ratio less than 2.0 was considered negative. Staging of primary tumors was based on the TNM system of the sixth American Joint Committee on Cancer criteria (28). Grading of tumors and histological classification were based on the modified Black's nuclear grading system (29) and World Health Organization (WHO) criteria (30), respectively.
Data Collection
Data on MS components – namely, BMI, hypertension, blood glucose, HDL and TG levels at initial diagnosis were collected. For each patient, information was also obtained on medication for hypertension, elevated blood glucose and dyslipidemia. Patients’ information, including demographics, clinicopathological data, treatment modalities and details of clinical outcomes were also recorded. All patients were treated with a multidisciplinary approach and then followed up every 3-6 months.
The diagnosis of MS was ascertained in patients based on the AACE 2003 definitions. The diagnosis of MS by the AACE definition required the presence of BMI ≥25 (calculated as kg/m2) in addition to at least 2 of the following criteria: (i) fasting serum glucose ≥110 mg/dL, (ii) fasting HDL cholesterol <50 mg/dL, (iii) fasting TGs ≥150 mg/dL, (iv) resting blood pressure ≥130/85 mm Hg. RFS was measured from the date of diagnosis to the date of first documented local or distant recurrence or last follow-up. Overall survival (OS) was defined as the time from the date of diagnosis to the date of death or last follow-up.
This study was approved by the Medical Center Institutional Review Board of the Cancer Hospital and Institute, Chinese Academy of Medical Sciences.
Statistical Analysis
Statistical analyses were carried out using SPSS 19.0 (SPSS Inc., Chicago, IL, USA). The differences between the groups were evaluated by the chi-square test. Continuous variables were compared using the 2-sample t-test. The Kaplan-Meier product limit method was used to estimate the survival outcomes of all patients; groups were compared using the log-rank test. A multivariate Cox regression model was used to identify the variables that were independently associated with survival. A p value <0.05 was considered to be significant for all comparisons.
Results
Demographics
The entire cohort comprised 394 TNBC and 855 non-TNBC patients. The median follow-up time of this study was 79.0 months (range 4.8-145.0 months). The age at diagnosis ranged from 22 to 91 years in the entire cohort, with the same median age of 49 years for both groups. Tumor characteristics were comparable between the 2 groups, such as TNM stage, histology type and percentage of lymphovascular invasion, except tumor grade since TNBC tumors tend to have higher histology grade than non-TNBC tumors (p<0.0001). As expected, due to the aggressiveness of TNBC tumors, more patients in the TNBC group received adjuvant chemotherapy (90.1%) and radiation therapy (40.6%) compared with the non-TNBC group, 85.0% and 32.5%, both p's <0.05. Moreover, compared with the non-TNBC group, more cases in TNBC group received Taxane-based adjuvant chemotherapy, 57.4% vs. 41.9%, p<0.0001 (Tab. I).
Clinicopathological characteristics of TNBC and non-TNBC group
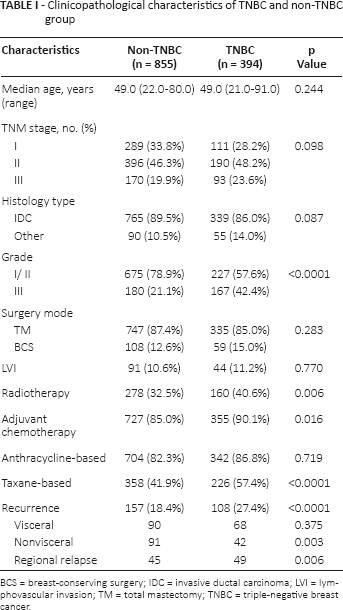
BCS = breast-conserving surgery; IDC = invasive ductal carcinoma; LVI = lymphovascular invasion; TM = total mastectomy; TNBC = triple-negative breast cancer.
Distribution of MS Components between TNBC and non-TNBC Groups
Based on AACE 2003 definitions, the frequencies of MS and its components including index of BMI, the level of serum HDL and TG were distributed evenly between the 2 groups (all p's >0.05). For example, 126 TNBC patients (32.0%) had low HDL and 77 TNBC cases (19.0%) high TG; similarly 274 non-TNBC patients (32.0%) had low-HDL, and 179 non-TNBC cases (20.9%) high TG. However, significantly more TNBCs than non-TNBCs presented with high blood pressure (BP ≥130/85 mm Hg) and elevated serum glucose (>110 mg/dl) (20.3% vs. 14.9% and 16.0% vs. 10.8%, p = 0.018 and p = 0.012, respectively). With regard to medication, there were no significant differences in medication use for MS components between TNBC and non-TNBC groups (Tab. II).
Distribution of MS components in TNBC and non-TNBC patients
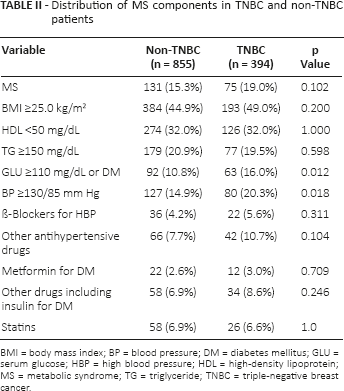
BMI = body mass index; BP = blood pressure; DM = diabetes mellitus; GLU = serum glucose; HBP = high blood pressure; HDL = high-density lipoprotein; MS = metabolic syndrome; TG = triglyceride; TNBC = triple-negative breast cancer.
Recurrence and Survivals
In total, 265 cases developed a recurrence, 108 cases (27.4%) from TNBC patients and 157 cases (18.4%) from non-TNBC patients (p<0.0001). Metastatic TNBC tumors were prone to have distant organ metastasis, regional relapse, but not bone metastasis. Only 5 cases in the non-TNBC group died of cardiovascular and cerebrovascular events, and all of the other 146 deaths were attributed to cancer-specific causes. The 5-year RFS rate of TNBCs and non-TNBCs was 72.8% and 84.2%, respectively (p<0.0001). Similarly, compared with non-TNBC cases, TNBC patients had a worse 5-year OS rate (83.5% vs. 93.3%, p<0.0001).
MS Variables and Survivals for TNBC and non-TNBC Patients
In the TNBC group, patients with decreased serum HDL levels showed both worse RFS and OS (p's<0.0001) (Fig. 1). Elevated fasting glucose was correlated with higher risk of tumor recurrence (p = 0.022). It also demonstrated a trend to more death events, with borderline significance (p = 0.092). It was observed that patients with MS had a trend to more tumor recurrence than patients without MS (p = 0.058). No significant correlations were found for other MS variables with survival rates in TNBC patients (Tab. III). For non-TNBC patients, the presence of MS and high blood pressure increased the risk of all-cause death significantly (p = 0.046 and p = 0.014 respectively); no positive results were suggested in relationships between other components of MS and non-TNBC survival rates (Tab. III).
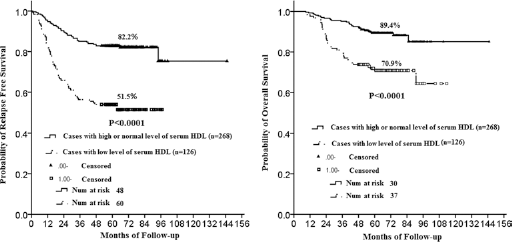
Kaplan-Meier survival curves for relapse-free survival (RFS) and overall survival (OS) in triple-negative breast cancer (TNBC) patients based on serum high-density lipoprotein (HDL) levels. Patients with low HDL had a significantly worse prognosis than patients with normal or high HDL, both p's<0.0001 (low HDL vs. normal or high HDL = RFS hazard ratio [HR] = 3.346; 95%CI, 2.287-4.895; OS HR = 2.986; 95%CI, 1.843-4.837).
Relationships of MS and MS variables with survival rates in TNBC and non-TNBC patients
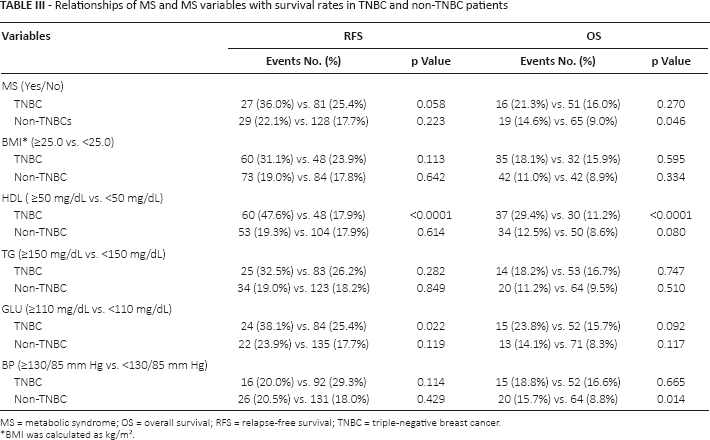
MS = metabolic syndrome; OS = overall survival; RFS = relapse-free survival; TNBC = triple-negative breast cancer.
BMI was calculated as kg/m2.
In the Cox proportional hazards regression analyses adjusting for possible confounding factors, only elevated HDL was identified as an adverse prognostic factor for both RFS and OS in TNBC patients (hazard ratio [HR] = 3.266, 95% confidence interval [95% CI], 2.087-5.112, p<0.0001; and HR = 3.071, 95% CI, 1.732-5.445, p<0.0001). Besides that, larger tumor size (>2 cm), positive lymph node and younger age were independent poor prognostic factors for both RFS and OS (all p's<0.005) (Tab. IV).
Multivariate analysis for RFS and OS for TNBC cases
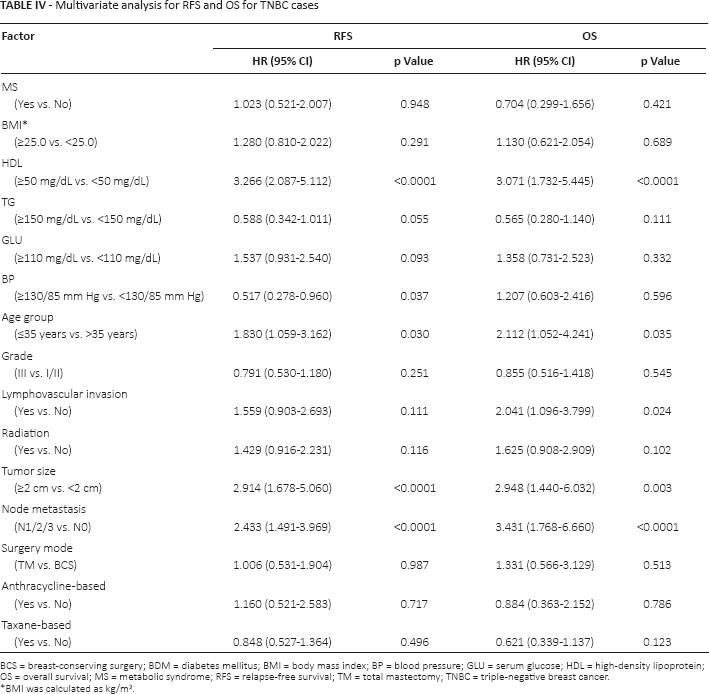
BCS = breast-conserving surgery; BDM = diabetes mellitus; BMI = body mass index; BP = blood pressure; GLU = serum glucose; HDL = high-density lipoprotein; OS = overall survival; MS = metabolic syndrome; RFS = relapse-free survival; TM = total mastectomy; TNBC = triple-negative breast cancer.
BMI was calculated as kg/m2.
But in the Cox proportional hazards regression analyses adjusting for possible confounding factors in the non-TNBC cases, none of the MS variables was identified as a significant prognostic factor for survival, for either RFS or OS (Tab. V).
Multivariate analysis for RFS and OS for non-TNBC cases
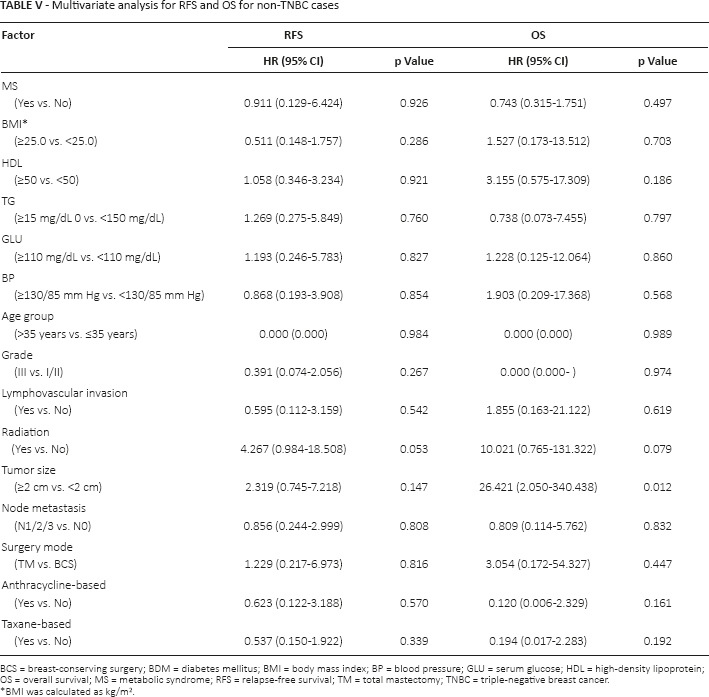
BCS = breast-conserving surgery; BDM = diabetes mellitus; BMI = body mass index; BP = blood pressure; GLU = serum glucose; HDL = high-density lipoprotein; OS = overall survival; MS = metabolic syndrome; RFS = relapse-free survival; TM = total mastectomy; TNBC = triple-negative breast cancer.
BMI was calculated as kg/m2.
Discussion
Despite the growing amount of evidence that both MS and components of MS, such as obesity, insulin resistance, elevated TG, elevated glucose or type 2 DM predict a higher risk of BC (2–16), questions remain to be answered: Does MS have an association with the outcomes for TNBC? Is there any difference between TNBC and non-TNBC patients in terms of their relationships with MS. Therefore, the present study was conducted to find clues to these questions.
The present study first revealed that HDL, TG or MS was not associated with TNBC susceptibility in a Chinese population – that is, no differences were observed in the frequencies of these variables between the TNBC and non-TNBC cohorts, which is inconsistent with previous studies reported in the literature (16, 18). In a case-control study of 2,070 cases in Korea, a significant negative association was observed between HDL and BC, the risk was evident in ER-/PR- cases compared with ER+/PR+ cases, with odds ratios (ORs) of 0.49 (95% CI, 0.32-0.76) and 0.59 (95% CI, 0.41-0.85), respectively (6). In another study of 176 patients from the United States, HDL levels also showed a significant independent inverse association with TNBC (p = 0.003) (20). The difference may be attributed to different race or different sample size.
We further demonstrated that only low HDL level was correlated with worse survivals of TNBC tumors. Compared with TNBC patients with HDL ≥50 mg/dL, those with HDL <50 mg/dL had a statistically significant about threefold greater risk of both cancer recurrence and all-cause mortality during a median 79 months of follow-up. The association of low HDL with worse survival was independent of age, BMI, tumor stage, tumor grade, lymph nodes and other confounding factors. However, such was not the case for non-TNBC patients.
Why HDL levels play an important role in TNBC cases could be in part explained by the following mechanism. HDL particles play an important role in removing cholesterol from peripheral tissues, thus the level of serum cholesterol could be decreased. While cholesterol could contribute to BC progression and tumor metastasis via various pathways, such as CD44, AKT, EGFR etc. (31), so it is assumed that HDL acts as a protective mediator from cancer progression and development. Another possible mechanism is that HDL cholesterol correlates negatively with ANG II, but ANG II is positively associated with VEGF in TNBC, while the VEGF signaling pathway plays a pivotal role in TNBC (2, 32, 33); therefore, low HDL may lead to more aggressive behavior of TNBC. Besides that, a positive correlation was found between adiponectin and HDL (34, 35), and in a Korean cohort study, serum adiponectin concentrations showed a significant inverse association with ER/PR-negative BC recurrence (p = 0.009) (9). These are just assumptions regarding the underlying mechanisms for the influence of HDL on TNBCs, however, and this is still far from explaining why low HDL acts differently between TNBC and non-TNBC patients.
When it comes to other MS factors, only elevated serum glucose was negatively correlated with tumor recurrence in the TNBC cases. However, it was not associated with recurrence or death after adjustments for tumor variables, HDL level and other possible prognostic factors. In fact, previous studies have indicated that diabetes was associated with both higher risk of tumor recurrence and BC all-cause mortality (10–12). A possible reason for the discrepancy is that elevated fasting serum glucose (defined as ≥110 mg/dL) does not equate to DM.
In addition, it was noted that increased TG did not show any significant association with tumor recurrence, irrespective of hormone receptor status. The results were in agreement with 2 other cohort studies (9, 36). They reported that TG was not associated with BC recurrence or death before or after adjustment for age, tumor-related variables, BMI or fasting insulin levels (9, 36).
Of course, the current study had several limitations. First, we used BMI rather than waist circumference to define obesity. Second, the present study was a retrospective study, and life styles, endocrine drugs and chemotherapy drugs may affect lipid profiles. Thus, definitive conclusions on the associations of HDL with TNBC survival rates cannot be drawn in the presence of the decreased power due to the above confounding factors.
Conclusion
Although HDL did not contribute as a prerequisite factor for the occurrence of TNBCs, it has prognostic significance in TNBC tumor recurrence and cancer-specific mortality, and it may protect against recurrence in TNBC patients. Thus, it may be especially important for monitoring and normalizing the HDL level in this subtype of BC. As these findings were not observed in the case of non-TNBC patients, further investigations are warranted to detect the underlying mechanisms.
Footnotes
Financial support: This study was supported in part by a grant from the National Natural Science Foundation of China for Young Scholars (No. 81202108).
Conflict of interest: The authors declare that there are no conflicts of interest.
