Abstract
Background
Triple-negative breast cancer (TNBC) is characterized by the absence of estrogen receptor, progesterone receptor and HER2. TNBCs are a diverse subgroup, but one promising marker and therapeutic target of this breast cancer is the androgen receptor (AR). Previously we demonstrated that AR and cognate intracrine pathways are associated with decreased proliferation in invasive ductal carcinoma with their decrease also detected between organ-confined and invasive diseases. Therefore, in this study, we examined the status of AR and androgen-producing enzymes during the process of metastasis to lymph nodes and cancer recurrence.
Materials and Methods
We studied 2 series of patients with TNBC, one from Kumamoto University Hospital composed of 16 matched cases of primary and locally or distal recurrences and the other from Tohoku University Hospital examining 46 lymph node metastasis from 23 patients. In addition to studying concordance in AR expression, we also examined the interactions between AR and Ki-67 labeling index and AR and site of distal metastasis.
Results
In both series, AR status was concordant between primary and recurrent/metastatic disease, but coordinated expression of AR and androgenic enzymes was lost during the process. The inverse association between AR and Ki-67, previously reported in invasive ductal carcinoma (IDC), was markedly potentiated in both lymph node and recurrent cancers. In addition, AR expression appeared to have little effect on visceral metastasis but was associated directly with bone metastasis and inversely with brain metastasis.
Conclusions
The results of our present study demonstrated that AR remained in the majority of metastatic samples from AR-positive primary TNBCs and that AR manipulation could be exploited in the metastatic settings of TNBC.
Introduction
Triple-negative breast cancer (TNBC) is defined by the absence of estrogen receptor (ER), progesterone receptor (PR) and HER2 proteins. There are currently no targeted therapeutic options available, and a subset of current breast cancer research has been devoted to exploring novel therapeutic targets for these patients. One target suggested is that of the androgen receptor (AR) and manipulation of this pathway could be a novel therapeutic approach in at least a subset of TNBC patients (1).
However, to date it is undetermined whether to stimulate or inhibit androgen actions to exert therapeutic effects in TNBC patients. AR appears to function as an oncogene (1, 2) in the great majority of cell culture studies. The underlying biology of this finding has been reported to be at least partially driven by AR, in the absence of ER, enacting an ER-related gene signature stimulating cell proliferation (2). The paradox and hence confusion comes from the divergence between these molecular biology studies and the results of of immunohistochemical (3) and a handful of cell culture studies (4). The latter two suggest that the presence of AR in carcinoma cells may even provide a survival advantage to the patient through the inhibition rather than stimulation of cellular proliferation (4).
In an attempt to address this question, we have previously examined the correlation between AR and androgen synthesis enzymes in primary invasive ductal carcinoma (IDC) and in organ-confined cancers, to further explore this latter interaction between AR expression and outcome in triple-negative cancers (5, 6). In these studies, we reported that AR expression alone or in tandem with enzyme expression was significantly associated with a lower Ki67 labeling index (LI) in primary invasive tumors (6) and that progression within ductal carcinoma in situ (DCIS) grades and between DCIS and IDC was also associated with the loss of AR immunoreactivity of carcinoma cells (5). These results suggested that AR could have suppressive properties, especially in tandem with its synthetic enzymes. Therefore, we hypothesized that the loss or disruption of this system could be a nonobligate step in further metastasis of TNBC cells. In addition, a secondary goal of this particular study was to examine the prevalence of AR and androgen-producing enzymes in TNBC metastasis to examine any potential that androgens could have as a therapeutic target in advanced disease stages or recurring diseases.
Material and Methods
Cases of TNBC
Lymph node metastasis specimens were retrieved from a wider cohort of IDC cases from the surgical pathology files of Tohoku University as we previously described (5) and selected for the presence of lymph node metastasis and the availability of archival paraffin-embedded tissue. The recurrent cases were a TNBC subset of samples retrieved from the surgical pathology files of Kumamoto City Hospital, and their detailed clinicopathological features have previously been published (7). Ethical approval for this particular project was obtained from the institutional review boards of both Tohoku University Hospital and Kumamoto City Hospital. In neither of the cohorts described in this paper had AR or androgenic enzymes been examined previously in the metastatic specimens. The only redundancy in this data is the use of mean values from the historical IDC cohort to compare the status of AR and enzymes between primary and lymph metastatic sites.
Immunohistochemistry
Immunohistochemistry of AR, 5-alpha-reductase type 1 (5αR1), 17-beta-hydroxysteroid dehydrogenase type 5 (17βHSD5) and Ki67 was performed as previously described (5, 6). In brief, 10% formalin-fixed paraffin-embedded tissue specimens were dewaxed and hydrated by a xylene-ethanol gradient. Antigen retrieval (Autoclave, Ph6) was performed for all antibodies, with the exception of 5αR1. Primary antibodies used were as follows: AR (AR441, 1:50; DAKO), 5αR1 (ab79058, 1:1,000; Abcam), 17βHSD5 (NP6.G6.A6, 1:200; Sigma) and Ki-67 (Clone:MIB1, 1:100; DAKO). Visualization of the primary antibody was performed by the streptavidin-biotin method. Quantification was performed as previously reported by our group, with AR being quantified by the gold standard H score (0-300, measure of intensity and prevalence), enzymes quantified in a semiquantitative manner (Score 0 was equivalent to 0% carcinoma cells stained, Score 1 was 1-50% of carcinoma cells stained, Score 2 was 51-100% of carcinoma cell stained) and Ki67 being determined by the LI (% positive cells) across the 5 areas of greatest immunointensity (hot spot method).
Statistical Analysis
Statistical analysis was performed using JMP V10.0.2. The correlations between AR status in primary and recurrent/lymph metastasis specimens were tested using correlation analysis, due to their continuous nature, while those between enzyme expression between primary and recurrent/lymph metastasis were tested using the chi-square test due to their categorical nature. In dichotomizing, enzyme-positive cases were defined as those with greater than 50% immunoreactivity throughout the carcinoma, while those with less than 50% were considered negative. When it was necessary to dichotomize AR into a positive or negative variable, we used a cutoff point of 10% LI based on the prevalence component of the H score assessment.
Results
Prevalence and Localization of AR, 5αR1 and 17βHSD5 in Lymph Node Metastasis of Primary and in Recurring TNBC
All 3 proteins above were immunolocalized primarily to the epithelial layer of breast cancer specimens with AR retaining its nuclear localization and the 2 enzymes being localized to the cytoplasm of tumor cells (Figs. 1 and 2). There were 16 recurrent TNBC cases and 46 lymph node metastasis from 23 patients available for analysis in this study. In lymph node metastasis cases that had multiple blocks available in each patient (n = 14), the correlation between AR and enzyme status was similar among all of the blocks examined, suggesting no heterogeneity of AR expression in the great majority of lymph metastasis. The subsequent comparison in these cases revealed that the lowest AR-expressing case was used as a point of comparison between primary and metastatic site, but results obtained in the data did not change if the highest AR-expressing case was used in the same fashion.
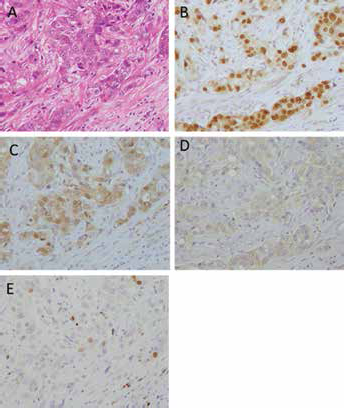
Representative immunohistochemical staining of lymph node metastasis. A) hematoxylin and eosin stained section; B) androgen receptor (AR) staining; C) 17βHSD5 staining; D) 5αR1 staining and E) Ki-67 staining.
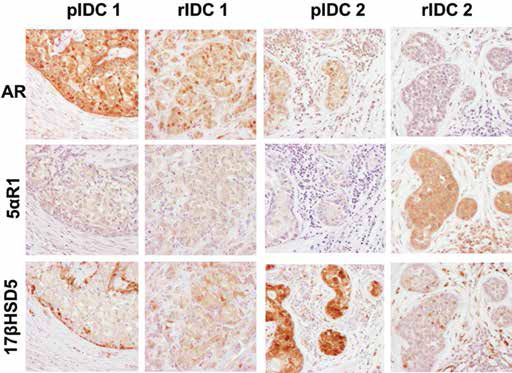
Patterns of androgen receptor (AR) and androgenic enzyme immunolocalization in matched pairs of primary and recurrent TNBCs. Representative illustrations of hematoxylin and eosin staining and immunohistochemistry (IHC) of AR, 5αR1 and 17βHSD5 of matched primary invasive ductal carcinoma (pIDC) and recurrent invasive ductal carcinoma (rIDC). Immunoreactivity was more heterogeneous in these cases, and patterns of AR immunoreactivity were maintained between pIDC and rIDC but not those of androgenic enzymes.
In the recurrent cases (Kumamoto cohort), there was a trend toward the association between AR and 5αR1 expression in the primary cancers (Fisher's exact test, p = 0.056) and a weak tendency toward the association between AR and enzyme status in the recurrent specimens. In the matched primary and lymph node specimens from the Tohoku series, no association was detected between AR and enzyme status in the lymph node metastasis specimens (data not shown).
Changes in AR and Enzyme between Primary and Lymph Node Metastasis/Recurrences
In recurrent diseases, the status of AR LI between pIDC and rIDC was significantly correlated (p<0.001, R2 = 0.79; Fig. 3A), but the levels in recurrent cases were not significantly different from those in historical IDC cases (data not shown). A gain of AR was not detected in any of the recurrence cases if AR was not detected in the primary cancer (Fig. 3A). Enzyme status was not correlated between primary and recurrent cancers (Fig. 3B, C). In lymph node specimens, AR status was similar between the primary tumor and the metastasis (Fig. 3D); the exception to this were 3 specimens that lost AR expression and 1 specimen that gained a low level of AR expression (H Score 40) between the primary and lymph metastasis. There was no significant correlation between the status of enzymes between primary and metastatic specimens (Fig. 3E, F)
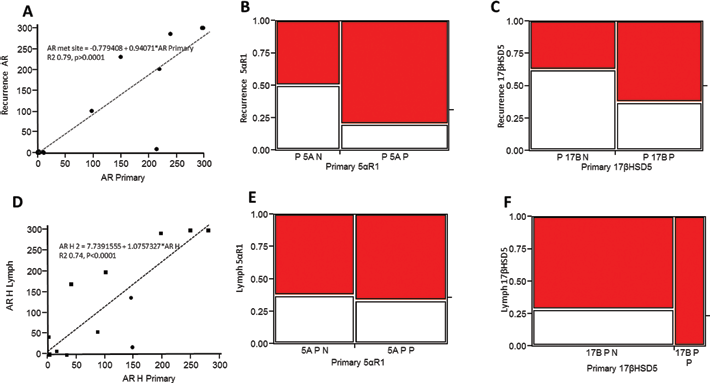
Androgen receptor (AR) and enzyme expression in recurrent compared with primary disease. Comparison of AR, 5αR1 and 17βHSD5 expression between primary cancer and recurrent metastasis (A-C) or primary and lymph node carcinoma cells (D-F). In rIDC and lymph node metastasis, AR expression was preserved at similar levels between primary cancers and recurrent (A) or lymph specimens (D); however, no significant associations were detected between enzyme status in pIDC and rIDC or in lymph metastasis. There is an apparent trend in rIDC (B, C); however, the power of the analysis was limited by the relatively small number of samples (n = 16).
AR Regulation of Ki67 in Lymph Metastasis and in Primary/Recurrent Cancers
In both the recurrent (Fig. 4A–D) and lymph node metastasis cohorts (Fig. 4E–H), the inverse correlation between AR expression and Ki-67 seen in their IDC counterparts was preserved, and for both situations, this correlation was more pronounced at the sites of metastasis or recurrence than in the primary cancer. This was evidenced by more marked R squared value for the Ki-67 with AR association (Fig. 4A vs. C, E vs. F) as well as the greater significance of the association between AR and Ki-67 detected in the secondary/recurring rather than the primary sites (Fig. 4B vs. D, F vs. G).
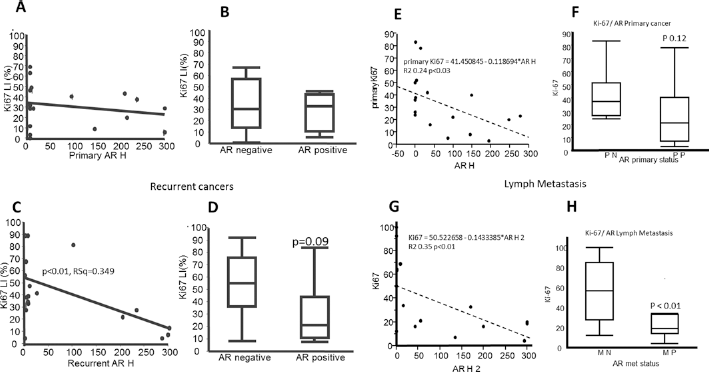
Associations between androgen receptor (AR) and Ki67 expression in lymph and recurrent samples. In both recurrent (A-D) samples and concurrent lymph metastasis samples (E-H), the correlation between Ki-67 and AR was stronger in the recurrent/metastatic sample (C, G) compared with the primary tumor (A, E). This same trend was also observed when AR was dichotomized into positive and negative groupings with an increase trend toward significance in the recurrent samples (B vs. D) and significance in the lymph metastasis samples (F vs. G). LI = labeling index.
Correlation between AR Status and Disease-Free Interval or Distal Metastasis Sites
In the recurrent specimens, we also examined whether there was any significant correlation between the sites of distal metastasis and AR status. No significant correlations were detected for visceral metastases such as those to the lung or liver, but there was an apparent trend for AR-positive recurrent cancer cases to metastasize to the bone, and inversely for AR-negative recurrent cancers to metastasize to the brain of the patients. This finding was, however, of limited statistical power due the relatively small number of patients who had distal metastasis at recurrence (n = 12) available for this study (Fig. 5). A similar trend was also detected in the limited number of available distal metastatic specimens from the Tohoku series (bone, n = 1; skin, n = 2; lung, n = 1; respiratory tract n = 1), with bone metastasis being the only specimen with AR expression (Fig. 5). The presence of AR in the recurrent disease and particularly in the lymph metastasis suggested a longer disease-free interval (data not shown) for the patients, but the correlation did not reach statistical significance.
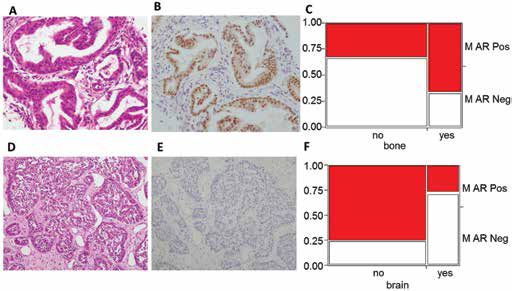
Association between androgen receptor (AR) expression and site of distal metastasis. A, B) Immunohistochemical staining of AR expression in bone metastasis; C) AR expression was associated with increased rates of metastasis to bone; D, E) example of non-bone metastasis (respiratory tract). F) Lack of AR was associated with metastasis to brain.
Discussion
In this study, we assessed if AR could also serve as a prognostic or biomarker in the recurrent and metastatic settings of TNBC patients and if so, what roles it might possibly play in these circumstances. We found that AR status tended to be preserved and its association with lower Ki-67 LI of carcinoma cells was particularly potentiated in both metastatic and recurrent tumors. However, the androgenic enzyme status did not appear to as stably preserved in either lymph metastasis or cancer recurrence.
A secondary question raised in this study was what roles AR expression plays at the sites of distal metastasis in TNBC patients. The preservation of AR in carcinoma cells between primary and metastatic/recurrent sites has previously been reported by other investigators (8–10); however, the association between AR or androgenic actions and metastatic properties or tendencies of breast cancer patients specifically in TNBC has not been examined. Previous reports found that in non-TNBC-specific breast cancers, AR expression was preserved between surgically resected primary tumors and metastases but lost in end-stage of metastases (8). The results of our present study did not contradict this and suggested that AR-positive TNBC cases are less likely to be associated with distant metastasis as previously postulated (9) and that AR status was preserved between primary tumor and lymph metastasis, and any changes in AR status between primary tumor and lymph metastasis were unidirectional, which is in accord with previous studies (i.e., a loss of AR in metastasis) (10). The association between lack or loss of AR with increasing disease aggression is consistent with the results of our previous studies (5, 6), suggesting that the presence of AR is associated with a less aggressive phenotype of TNBC.
The association between AR expression and proliferation is another area of uncertainty in relation to TNBC. The results of our present study demonstrated a potentiation of the inverse association between AR expression and cellular proliferation in metastatic and recurrent diseases compared with their cognate primary cancers in TNBC. The results of our previous studies also demonstrated a similar correlation between AR and proliferation in triple-negative IDC (6) and in organ-confined disease (DCIS) (5). The potentiation or augmentation of this correlation in the setting of recurrent disease also suggests a potential novel androgen manipulation in these patients, but this awaits further investigation for clarification.
The issue of how primary carcinoma phenotype relates to metastatic behavior and location is another area relevant to both basic biology and clinical practice. The results of our present study demonstrated that AR status in carcinoma cells appeared to be increased in bone metastasis and decreased in brain metastasis, suggesting an association between AR status and the modes of metastasis in TNBC patients. This association is of marked interest, especially given the propensity of TNBCs toward brain metastasis (see, e.g., (11, 12)). In line with these results it was previously shown in a Korean population that there were no AR-expressing primary tumors associated with brain metastasis (13), suggesting an association between AR expression and brain metastasis. Previous studies have shown brain metastasis to be more frequent in the patients with basal-like cancer (14, 15), and basal-like TNBCs were also reported to be associated with reduced rates of AR status in carcinoma cells (16), suggesting associations between AR, metastasis and differentiation. The correlation between AR expression of carcinoma cells and bone metastasis has been little studied. TNBCs show a lower propensity to bone metastasis compared with other breast cancer types (11). Koo et al examined AR expression and bone metastasis in a series of 6 patients and reported that only 1 of these 6 patients had AR in carcinoma cells (13). Given the very small number of cases in our study, and in those of others, it is difficult to draw any definite conclusions, but this interesting concept of roles of androgenic action in involving the sites of possible metastasis is certainly worth pursuing in further studies.
While we have previously reported that the expression of androgen producing or metabolizing enzymes as well as their association with AR was lost between DCIS and IDC of TNBC phenotypes (5) – as also reported in other subtypes of breast cancer patients (17) – this pattern did not clearly emerge with regard to metastases. Before this current study, the status of the androgen producing or metabolizing enzymes in primary and cognate metastatic TNBC cases has not been reported in the literature; however, there seems to be little coordination or dysregulation beyond that seen in IDC. While this does not refute the suggestion that dysregulation of androgenic intracrine pathways could be a causative factor in breast cancer progression, it does suggest they may not be an important factor in subsequent metastasis.
In conclusion, this study demonstrated that AR status was preserved between primary tumors, lymph metastasis and recurrent tumors in TNBC patients. In addition, in any of the settings above, the presence of AR in carcinoma cells was significantly associated with a lower Ki-67 LI, suggesting a protective role for, or at least a better biological fundament associated with, AR expression in these cancers regardless of the primary tumor or metastasis setting.
Footnotes
Financial support: No grants or funding have been received for this study.
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
