Abstract
Inversin, encoded by NPHP2, is one of the 10 NPHP proteins known to be involved in nephronophthisis (an autosomal recessive cystic kidney). Although the previous reports showed that inversin played an important role in embryonic development and renal diseases, its function in cancer was not revealed clearly so far. As measured by immunohistochemical staining, inversin was highly expressed in the cytoplasm of lung cancer samples (63.4%, 161/254) compared with adjacent normal lung tissues (22.0%, 11/50,
Introduction
Inversin, also known as Nephrocystin-2, is a 1065-amino acid protein encoded by NPHP2 (NPHP, nephronophthisis, an autosomal recessive cystic kidney) and a member of Nephrocystin protein family. It is expressed during presomite-stage embryos and persists in adulthood, which is essential for establishment of the left–right axis, ciliary functions, and normal renal development.1–3 Mice expressing inversin mutations are primarily generated by random insertional mutagenesis and result in the reversal of left/right polarity and cyst formation in the kidneys.4–6 Wnt signaling pathways play important roles in oriented and asymmetric cell division, ciliary polarization, renal development, and cystogenesis.7–10 The studies suggested that these above-mentioned roles of inversin were more or less involved in its interaction with dishevelled (DVL) and its effects on Wnt signaling, especially the non-canonical Wnt signaling pathway.11–13 Aberrant Wnt signaling is a key player in the initiation, maintenance, and development of many cancers.14,15 However, the expression pattern and biological role of inversin in human malignant tumors remains unknown by now.
In this study, we collected 50 cases of adjacent normal lung tissues and 254 cases of lung cancer specimens, including 129 cases of squamous cell carcinoma, 108 cases of adenocarcinoma, 17 cases of other histological types. Immunohistochemical staining was used to detect the expression of inversin in these organizations. A positive correlation between inversin expression and the clinicopathological features of lung cancers was found in the 254 examined specimens. Furthermore, we measured the effects of inversin expression on lung cancer cell proliferation and invasion. Our results could provide a preliminary theoretical and experimental basis for targeting inversin in the treatment of lung cancers.
Materials and methods
Patients and specimens
Ethical approval for this study was obtained from the local trials committee of the China Medical University. A total of 50 cases of adjacent normal lung tissues were collected. Primary tumor specimens were obtained from 254 patients (139 males and 115 females) who were diagnosed with lung cancers and underwent complete resection in
Immunohistochemistry
Surgically excised tumor specimens were fixed with 10% neutral-buffered formalin, embedded in paraffin, and cut into 4-µm-thick sections. Immunostaining was performed using the avidin–biotin–peroxidase complex method (Ultrasensitive; MaiXin, Fuzhou, China). The sections were deparaffinized in xylene, rehydrated with graded alcohol, and then boiled in 0.01 M citrate buffer (pH 6.0) for 2 min in an autoclave. The sections were incubated with normal goat serum to reduce non-specific binding. Tissue sections were incubated with anti-inversin rabbit polyclonal antibody (1:100, Abcam, Cambridge, MA, USA). Rabbit immunoglobulin (at the same concentration as that of the antigen-specific antibody) was used as an isotype control. Staining was performed overnight at 4°C. Biotinylated goat anti-mouse serum IgG was used as a secondary antibody. Following three rinses with phosphate-buffered saline (PBS), the sections were incubated with streptavidin–biotin conjugated with horseradish peroxidase, and the peroxidase reaction was developed with 3, 3′-diaminobenzidine tetrahydrochloride. The sections were counterstained with hematoxylin and dehydrated in ethanol before mounting. Two independent, blinded investigators examined all tumor slides, and 100 cells were observed and scored at 400× magnification. Both the proportion of positive cells and the staining intensity were taken into consideration due to the variation among lesions. The rate of positive cells was categorized as follows: 0, <5%; 1, 5%–25%; 2, 26%–50%; 3, 51%–75%; 4, >75%. The intensity was graded as follows: 0, negative; 1, weak; 2, moderate; 3, strong. The proportion and intensity scores were then multiplied to obtain a final score. Scores of 0–3 were defined as “negative expression” and scores of 4–12 as “positive expression.”
Cell culture
The human normal bronchial epithelial (HBE) cell line was obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). The A549, LTE-A-2 (LTE), H1299, H460, H292, and SPC-A-1 (SPC) cell lines were obtained from Shanghai Cell Bank (Shanghai, China). The LK2 cell line was a gift from Dr Hiroshi Kijima (Department of Pathology and Bioscience, Hirosaki University Graduate School of Medicine, Japan). All cells were cultured in RPMI 1640 (Invitrogen, Carlsbad, CA, USA) supplemented with 10% fetal calf serum (Invitrogen), 100 IU/mL penicillin (Sigma, St. Louis, MO, USA), and 100 µg/mL streptomycin (Sigma) and passaged every other day using 0.25% trypsin (Invitrogen).
Plasmid transfection and small interfering RNA treatment
The pCMV6-ddk-myc empty vector and pCMV6-ddk-myc-inversin were purchased from Origene (Rockville, MD, USA). SiRNAs for inversin (sc-106294, which consist of pools of three to five target-specific 19-25 nt siRNAs designed to knockdown gene expression) and non-targeting siRNAs (sc-37007) were purchased from Santa Cruz Biotechnology Inc. (CA, USA). A549 and H1299 cells were seeded at 1 × 105 cells per 35-mm well. After 24 h, cells were transfected with pcDNA or siRNA for Inversin, using Lipofectamine 3000 (Invitrogen). After transfection, cells were incubated for 48 h and subjected to Western blot analyses. Cells transfected with empty pcDNA plasmid or non-targeting siRNAs served as negative controls to ensure that the observed biological changes were caused by the expression of the target gene.
Western blot analysis
Total protein from cells was extracted in lysis buffer (Pierce, Rockford, IL, USA) and quantified using the Bradford method. In all, 50 µm of protein were separated by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). After transferring to polyvinylidene fluoride membranes (Millipore, Billerica, MA, USA), the membranes were incubated overnight at 4°C with rabbit polyclonal antibody against inversin (1:500, ab65187, Abcam), mouse monoclonal antibody against E-cadherin (1:1000, BD Biosciences, San Jose, CA, USA), mouse monoclonal antibody against N-cadherin (1:1000, BD Biosciences), mouse monoclonal antibody against Vimentin (1:500, BD Biosciences), mouse monoclonal antibody against matrix metalloproteinase-2 (MMP-2; 1:500, Abcam), rabbit monoclonal antibody against matrix metalloproteinase-7 (MMP-7; 1:200, Abcam), rabbit monoclonal antibody against matrix metalloproteinase-9 (MMP-9; 1:500, Abcam), and mouse monoclonal antibody against glyceraldehyde 3-phosphate dehydrogenase (GAPDH; 1:1000, Santa Cruz Biotechnology). After incubation with peroxidase-coupled anti-rabbit and anti-mouse IgG (Santa Cruz Biotechnology) at 37°C for 2 h, bound proteins were visualized by enhanced chemiluminescence (ECL; Pierce, Rockford, IL, USA) and detected by BioImaging System (UVP, Upland, CA, USA).
Real-time polymerase chain reaction
Quantitative real-time polymerase chain reaction (PCR) was performed using SYBR Premix Ex Taq II (TaKaRa, Dalian, China) with the 7900HT fast real-time PCR system (Applied Biosystems, Carlsbad, California, USA) as follows: 50°C for 2 min, 95°C for 10 min, and 40 cycles of 95°C for 15 s and 60°C for 60 s. β-actin was used as the reference gene. The relative levels of gene expression were represented as ΔCt = Ct, gene − Ct, reference, and the fold change of gene expression was calculated by the 2−ΔΔCt method. Experiments were done in triplicate and the following primers were used: inversin, Forward 5′-GCAGGGAACAAACTCCAGAAGG-3′ and Reverse 5′-CTGTCTCACCTCTGGACTTCTC-3′.
Immunofluorescence
Cells were fixed with 4% paraformaldehyde, blocked with 1% bovine serum albumin (BSA), and incubated with inversin rabbit polyclonal antibody (1:100, Abcam). Overnight at 4°C, followed by tetramethylrhodamine isothiocyanate (TRITC)-conjugated secondary antibodies, added at room temperature for 1 h. Primary antibodies were applied overnight at 4°C followed by incubation with secondary antibody conjugated to TRITC at room temperature for 1 h. Cells were counterstained with 4′,6-diamidino-2-phenylindole (DAPI). Epifluorescence microscopy was performed using an inverted Nikon TE300 microscope (Melville, NY, USA); confocal microscopy was carried out on a Radiance 2000 laser scanning confocal microscope (Carl Zeiss, Thornwood, NY, USA).
MTT
Cells were plated in 96-well plates in medium containing 10% fetal bovine serum (FBS) at a density of about 3000 cells per well. For quantitation of cell viability, cultures were subjected to MTT assay 4 days post-transfection. Briefly, 20 µL of 5 mg/mL Methylthiazolyldiphenyl-tetrazolium (MTT) bromide solution was added to each well and incubated for 4 h at 37°C. Then, media was removed from each well and the resultant MTT formazan was solubilized in 150 µL of dimethyl sulfoxide (DMSO). The results were quantified spectrophotometrically using a test wavelength of 570 nm.
Matrigel invasion
The cell invasion assay was performed using a 24-well transwell chamber with a pore size of 8 µm (Costar, Cambridge, MA, USA). The inserts were coated with 20- µL Matrigel (1:3 dilution, BD Biosciences). About 48 h after the transfection, cells were trypsinized, and 1 × 105 cells in 100 µL of serum-free medium were transferred to the upper Matrigel chamber and incubated for 18 h. A medium supplemented with 10% FBS was added to the lower chamber as the chemoattractant. After incubation, the cells that passed through the filter were fixed with 4% paraformaldehyde and stained with hematoxylin. The number of invaded cells was counted in 10 randomly selected high-power fields under the microscope.
Statistical analysis
SPSS version 13.0 for windows was used for all analyses (SPSS, Chicago, IL, USA). The Pearson chi-square test was used to examine possible correlations between inversin and clinicopathological factors.
Results
Inversin expression was elevated in non–small cell lung cancer tissues
First, we performed immunohistochemistry on 254 archived non–small cell lung cancer (NSCLC) specimens and adjacent noncancerous tissues. Inversin was not or very lowly expressed (Figure 1(a)–(c)) in normal tissues adjacent to carcinomas; meanwhile, inversin expression in the cell cytoplasm was elevated along with the poor differentiation of lung squamous cell carcinoma (Figure 1(d)–(f)) and lung adenocarcinoma (Figure 1(g)–(i)); in addition, the positive expression rate (63.4%, 161/254) in cancer specimens was significantly higher than that in normal lung tissues (22.0%, 11/50,
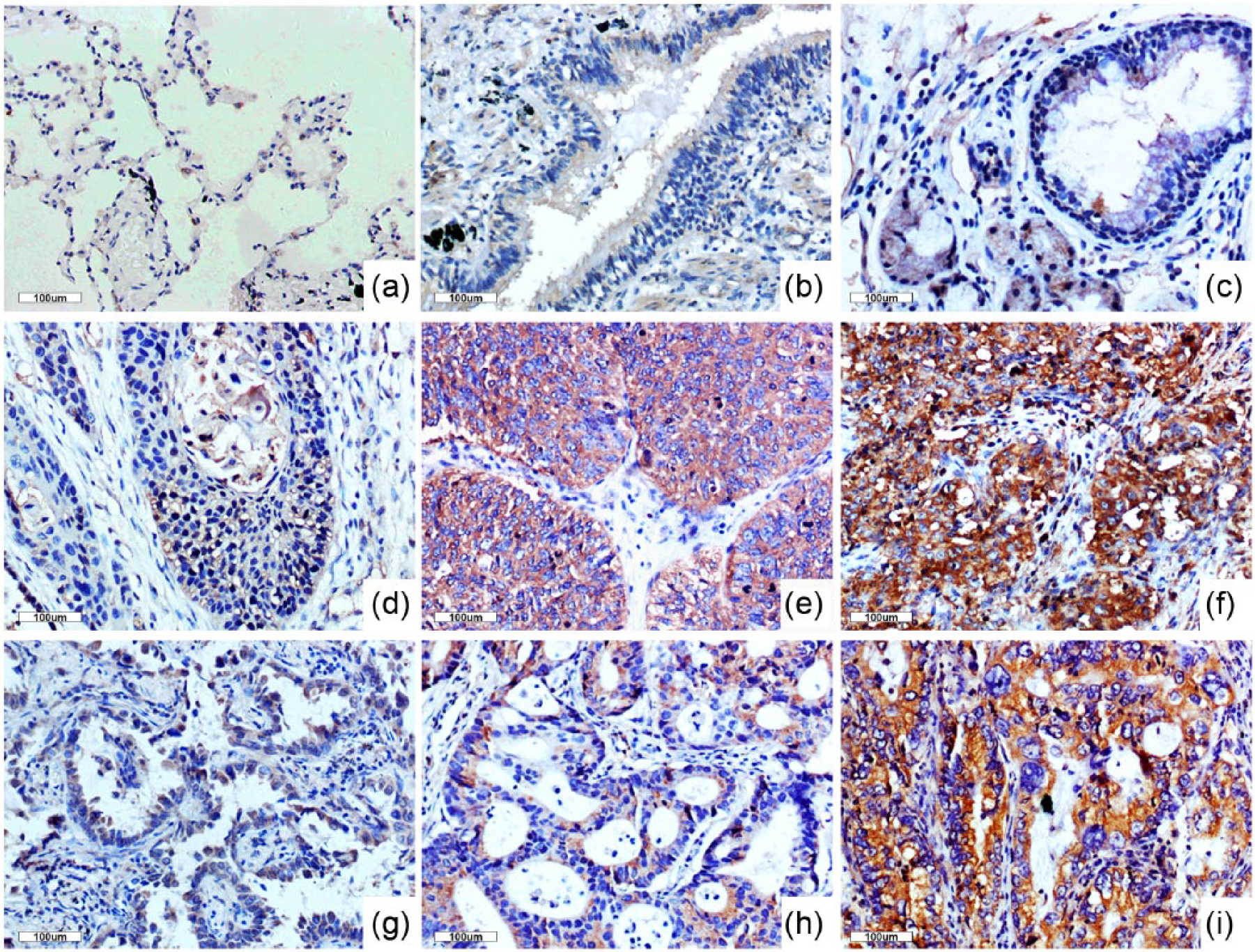
Immunohistochemical analysis of inversin in non–small cell lung carcinoma. The cytoplasm of (a) alveoli, (b) normal bronchial epithelium, and (c) glands was negative or weakly positive for inversin. Positive cytosolic inversin immunoreactivity was observed in lung squamous cell carcinoma (d–f) and adenocarcinoma (g–i); inversin was highly expressed in poorly differentiated squamous cell carcinoma (f) and adenocarcinoma (i); the moderately differentiated squamous cell carcinoma (e) and adenocarcinoma (h) showed moderate inversin cytosolic expression; weak cytosolic inversin immunoreactivity was observed in well-differentiated lung squamous cell carcinoma (d) and adenocarcinoma (g).
Inversin expression in adjacent non-tumor tissues and lung cancer.

Second, we evaluated inversin expression in clinical NSCLC samples using Western blotting. Inversin was expressed at the higher level in lung cancer tissues of 13/16 cases (81.25%) compared with the corresponding adjacent noncancerous tissues (Figure 2(a)). Image analysis of Western blot data yielded a normalized value of 0.6059 ± 0.1092 for inversin protein expression level in all the 16 cases of NSCLC specimens, which was significantly higher than that of the corresponding noncancerous tissues (0.3176 ± 0.07927;
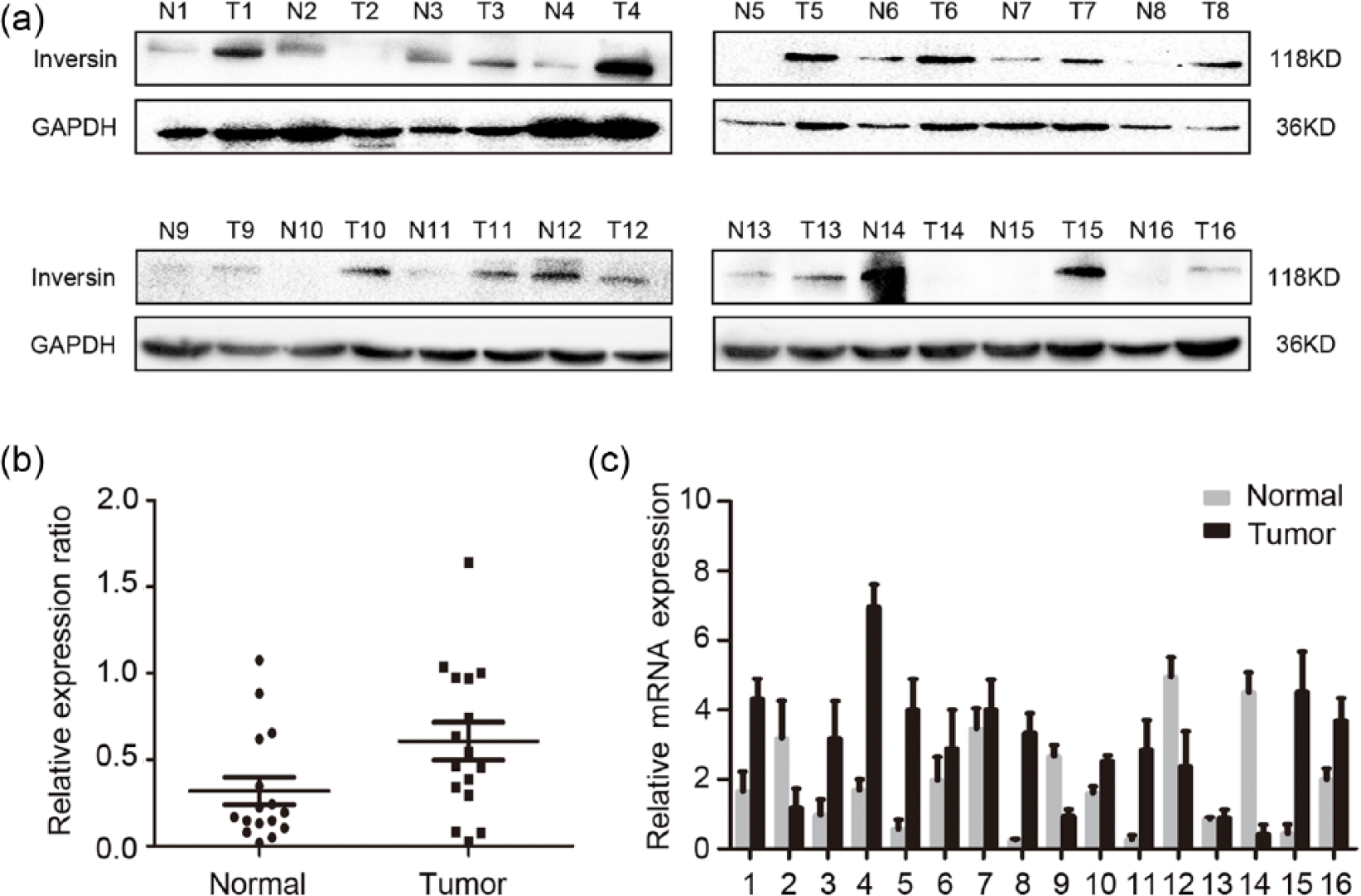
Western blot and real-time PCR analyses of inversin expression in non–small cell lung carcinoma. (a) In all, 13 cases (No. 1, 3, 4, 5, 6, 7, 8, 9, 10, 11, 13, 15, and 16) of the 16 tissue specimens examined had higher inversin expression levels in NSCLC compared with adjacent noncancerous tissues. (b) Image analysis of Western blots data yielded a normalized value for inversin protein expression level in NSCLC specimens and was significantly higher than in normal tissues. Values represent mean ± SE (bars) of three independent experiments. (c) The mRNA expression of inversin in NSCLC specimens was significantly higher than in normal tissues. Values represent mean ± SE (bars) of three independent experiments.
The correlation between inversin expression and clinicopathological features of NSCLC
We analyzed the correlation between inversin expression and the clinical pathological factors of NSCLC in 254 cases of NSCLC. As shown in Table 2, inversin expression had the significantly positive correlation with differentiation, lymph node metastasis, and TNM stage of NSCLC (
Summary of correlation of inversin expression with clinicopathological characteristics in NSCLC.

NSCLC: non–small cell lung cancer; TNM: tumor node metastasis.

Kaplan–Meier curves of overall survival of the 254 NSCLC patients stratified by inversin expression level. Inversin positive expression was correlated with overall survival (
Inversin enhances the invasion ability of NSCLC cell
We performed Western blot and real-time PCR to assess the protein and mRNA expression of inversin in six NSCLC cell lines and HBE cell line. The results showed that both inversin protein (Figure 4(a)) and mRNA (Figure 4(b)) levels in most NSCLC cell lines (5/6) were significantly higher than the levels obtained in HBE cells, except for in A549. Thus, NSCLC cell lines A549 and H1299, as the representative of low and high inversin expression, respectively, were selected for further testing. As shown in Supplementary Figure 1, inversin was mainly localized in the cytoplasm of HBE, A549, and H1299 cells by immunofluorescence, which was consistent with the results of immunohistochemistry.

Expression pattern of inversin in NSCLC cell lines. (a) Protein levels of inversin in seven lung cancer cell lines (A549, SPC, H292, H1299, LTE, LK2, and H460) and a human bronchial epithelium cell line (HBE). Data represent at least three independent experiments with similar results. (b) mRNA levels of inversin in seven lung cancer cell lines (A549, SPC, H292, H1299, LTE, LK2, and H460) and a human bronchial epithelium cell line (HBE). Values represent mean ± SE (bars) of three independent experiments.
The transfection efficiency was shown in Figure 5(a) and (b), after inversin overexpression and silencing in A549 and H1299 cells, respectively. The MTT assays showed that the effects of inversin were very limited on the proliferation abilities of A549 (

The roles of inversin in the regulation of lung cancer cell proliferation and invasion. (a) Western blot analyses of inversin protein levels after overexpression or silencing of inversin in A549 and H1299 cells, respectively. (b) Real-time PCR analyses of inversin mRNA levels after overexpression or silencing of inversin in A549 and H1299 cells, respectively. Values represent mean ± SE (bars) of three independent experiments, *
Inversin may modulate lung cancer cell invasion through the regulation of cadherins and MMPs
It has been reported that inversin could affect Wnt signaling, especially the non-canonical Wnt signaling pathway.11,12 To further explore the mechanisms how inversin modulated lung cancer cell invasion, we investigated its influence on cadherins and the invasion-related target genes of Wnt signaling pathways, such as E-cadherin, N-cadherin, Vimentin, MMP-2, MMP-7, and MMP-9. Our results suggested that in A549 cells transfected with inversin (Figure 6), expression of N-cadherin, Vimentin, MMP-2, and MMP-9 was significantly upregulated. In contrast, for H1299 cells with inversin knocked-down (Figure 6), expression of N-cadherin, Vimentin, MMP-2, and MMP-9 was significantly downregulated. However, E-cadherin and MMP-7 had no significant changes (Figure 6). These results implied that inversin might promote lung cancer cell invasion by regulating the expression of N-cadherin, Vimentin, MMP-2, and MMP-9.
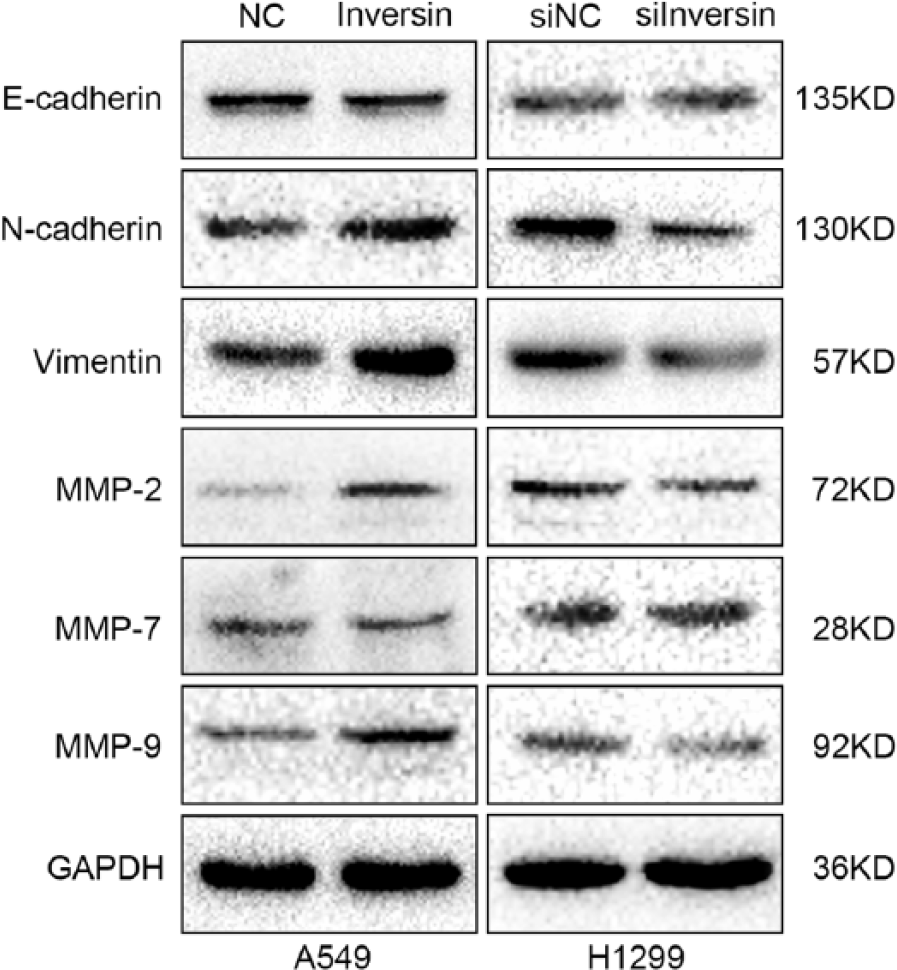
Inversin could regulate the expression of cadherins and MMPs. N-cadherin, Vimentin, MMP-2, and MMP-9 were significantly upregulated in A549 cells transfected with inversin; N-cadherin, Vimentin, MMP-2, and MMP-9 were significantly downregulated in H1299 cells knocked-down inversin. However, E-cadherin and MMP-7 had no significant changes.
Discussion
Inversin plays essential roles in establishment of left-right asymmetry, nephronophthisis type II (an cystic kidney disease), and the ciliary polarization and function.2–6 And it has been pointed out that inversin, as a key molecular switch of canonical and non-canonical Wnt signaling pathways, could interact with DVL and promote non-canonical Wnt/planar cell polarity (PCP) signaling.11–13 Furthermore, Wnt signaling pathways are closely related to cancers.14,15 Although the roles of inversin in mammalian embryonic development have been well described, its expressions and biological functions in tumors have not been explored.
We performed immunohistochemistry, Western blot, and real-time PCR in NSCLC specimens and lung cancer cell lines. The results showed that inversin was highly expressed in the cytoplasm of NSCLC cells, almost no nuclear expression was found in NSCLC specimens. Its expression was significantly correlated with the differentiation, TNM staging, and lymph node metastasis of NSCLC patients. Moreover, NSCLC patients with inversin positive expression showed obviously less survival time, which indicated that inversin might be associated with malignant phenotype and poor prognosis of NSCLC. In addition, we found that although without significant effects on proliferation, inversin could enhance the invasion of NSCLC cells. Furthermore, we confirmed that inversin could upregulate the expression of N-cadherin, Vimentin, MMP-2, and MMP-9, which are closely associated with tumor cell invasion ability. Although the detailed mechanism is yet unclear, all these above results indicated that inversin is highly expressed in NSCLC and has the capacity to promote the invasion of lung cancer cells.
In the process of epithelial–mesenchymal transition (EMT), the invasion and metastasis abilities of tumor cells are enhanced, accompanied by downregulation of epithelial marker E-cadherin and upregulation of mesenchymal markers N-cadherin and Vimentin. Although E-cadherin had no change in our results, N-cadherin and Vimentin were upregulated significantly, which implied that inversin could regulate the EMT of lung cancer cells. It has been reported that inversin could associate with N-cadherin and catenins, 16 which might contribute to the stability of N-cadherin, but the specific regulation mechanism remained to be clarified.
Matrix metalloproteinases (MMPs) play essential roles in the invasion and metastasis of cancer cells, 17 and we found overexpression of inversin could upregulate both MMP-2 and MMP-9, which were the downstream factors of non-canonical Wnt/PCP signaling.18–20 As a key molecular switch, inversin could activate non-canonical Wnt/PCP signaling.11–13 Additionally, the main domain of inversin was the same as the ankyrin repeat protein Diversin, 11 and we reported that Diversin was overexpressed in NSCLC and enhanced cancer cells invasion via promoting non-canonical Wnt (PCP/JNK) signaling pathway. 21 Therefore, inversin is likely to upregulate MMP-2 and MMP-9 by activating PCP/JNK signaling and then enhance the invasion of lung cancer cells. However, the assumption should be confirmed in our future studies.
To our knowledge, this is the first study to investigate the expression and function of inversin in NSCLC. Inversin is highly expressed in NSCLC and correlates with clinical pathological factors and poor prognosis, and its overexpression promotes invasion of NSCLC cells. Inversin may be useful as a valuable biomarker for the molecular diagnosis and a potential target for gene therapy of NSCLC.
Footnotes
Acknowledgements
We are grateful to Dr Hiroshi Kijima (Department of Pathology and Bioscience, Hirosaki University Graduate School of Medicine, Japan) for providing the LK2 cell line mentioned in this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (No. 81272606 to E.-H.W., No. 81401885 to X.-Y.L., and No. 81402369 to G.-Y.J.) and the Natural Science Foundation of Liaoning Province (No. 2013021049 to Y.Z.).
