Abstract
Background
Methods
Results
Conclusions
Introduction
Reliable determination of predictive factors for treatment outcomes in breast cancer is a prerequisite to the selection of optimal therapeutic strategy. Estrogen receptor (ER) and progesterone receptor (PR) are well-recognized prognostic markers in early-stage breast cancer, and prognostic markers for endocrine response guiding the routine use of endocrine therapy (ET) in clinical practice (1, 2). The use of adjuvant antiestrogen treatment reduces breast cancer mortality by more than 30% throughout the first 15 years (3) and approximately 50% during the second decade after diagnosis (4), but late recurrence is commonly observed in ER-positive breast cancer. Though late recurrence is not independently associated with hormone receptor (HR) status of the disease (5-6-7), management of ER-positive breast cancer with ET relies heavily on the HR status at time of diagnosis.
Neoadjuvant chemotherapy (NAC) was in the early years the standard treatment for patients with locally advanced breast cancer (LABC) and nowadays is also a widely accepted treatment option for operable breast cancer (8, 9) to increase the rate of breast conservation. Some authors have reported that ER and PR status may alter in the course of NAC (10-11-12-13-14-15). A meta-analysis showed that approximately 15%-18% of patients had an ER switch after NAC (14, 16). The positive-to-negative switch of HR status after NAC might predict a poorer disease-free survival (DFS) in patients receiving adjuvant ET (16), but this is yet to be further confirmed. This study aimed to investigate whether ET could yield any benefit in ER-positive breast cancer patients.
Materials and Methods
Patient Population
From December 2000 to November 2010, 97 out of 687 eligible primary breast cancer patients (14.1%) treated with NAC at the Comprehensive Breast Health Center of Ruijin Hospital in Shanghai were retrospectively reviewed. The eligibility criteria for inclusion in the study were women with unilateral breast cancer, treatment with a minimum of 3 and at most 6 cycles of NAC. The following clinical data were drawn from patients’ clinical reports: age, menopausal status, clinical tumor size and lymph node status, pathologic axillary lymph node involvement and follow-up information. Only patients with a positive-to-negative switch in HR status after NAC were included in the study. The patients were classified into 2 groups based on the administration of ET: 57 ET-administered patients and 40 ET-naïve patients. Permission was granted by the ethics committee of Shanghai Jiaotong University School of Medicine to conduct this retrospective study.
Treatment
All patients were given 1 of the following chemotherapy regimens: NE (vinorelbine 25 mg/m2 on day 1 and day 8 plus epirubicin 60 mg/m2 on day 1, every 21 days), CEF (cyclophosphamide 500 mg/m2 on day 1, epirubicin 75 mg/m2 on day 1 and fluorouracil 500 mg/m2 on day 1, every 21 days) and ET (epirubicin 75 mg/m2 on day 1 plus docetaxel 75 mg/m2 on day 1, every 21 days); other NAC regimens included PCb (paclitaxel and carboplatin) and TCb (docetaxel and carboplatin). All patients underwent definitive surgery after 3 to 6 courses of NAC. Adjuvant chemotherapy was administered after surgery to complete a total of 6 to 8 courses of chemotherapy according to the preference of the treating physician on the basis of clinical and pathologic findings after the operation. Radiotherapy was administered after the completion of adjuvant chemotherapy. The prescription of ET and the choice of specific drugs were also determined according to the physician's decision and/or the patient's preferences. Only 1 patient with human epidermal growth factor receptor 2 (HER2)-positive disease received trastuzumab.
Assessment of Response
The clinical assessment of response was based on clinical measurements in the longest diameter of tumor and lymph node according to Response Evaluation Criteria in Solid Tumors (RECIST) guideline version 1.1 (17), and the response was classified as follows: complete response (CR): the disappearance of the disease; partial response (PR): at least a 30% decrease; progressive disease (PD): at least a 20% increase in the sum of the longest diameter of target lesions or the appearance of new lesions; stable disease (SD): neither sufficient shrinkage to qualify for PR nor sufficient increase to qualify for PD. Pathological complete response (pCR) was defined as absence of invasive tumor in both breast and axillary lymph node. Patients with pCR after NAC were excluded from this analysis because the HR status after surgery could not be accurately evaluated.
Hormone Receptor and HER2 Status Determination
All patients underwent a 14-gauge core needle biopsy (CNB) before NAC. The ER, PR and HER2 status of CNB and surgical specimens was determined by immunohistochemistry (IHC). Positive staining for ER/PR was defined as nuclear staining in ≥10% of the tumor cells. HR was defined as positive if ER and/or PR was positive, and as negative if both ER and PR were negative. HER2 positivity was considered as HER2 3+ by IHC or positive as tested by FISH, whereas cases with 0 to 1+ or 2+ without FISH detection were regarded as negative. The following antibodies were used for the IHC test: ER: clone 1D5 (rabbit monoclonal, Gene); PR: clone PR636 (mouse monoclonal, Dako); HER2: c-erbB-2 (2000-2008, rabbit polyclonal, Dako) or 4B5 (2009-2010, rabbit monoclonal, Roche).
Statistical Analysis
The chi-square test was applied to evaluate the relationship between administration of ET and other parameters. Fisher's exact test was performed when necessary. DFS interval was defined as the time from the date of the first administration of NAC to the earliest occurrence of any local, regional or distant recurrence, any second cancers or contralateral breast cancers, or death. Overall survival (OS) was defined as the time from the date of the first administration of NAC to death regardless of whether it was breast cancer related or not. DFS and OS were estimated using the Kaplan-Meier analysis, and the survival curves were compared using the log-rank test. Multivariate Cox regression analysis with stepwise selection was used to estimate the hazard ratio (HR), 95% confidence interval (95% CI), and the effects of the clinical and pathological variables. All statistical tests were 2 sided and a p value <0.05 was considered significant. The software package SPSS 16.0 (SPSS Inc., Chicago, IL, USA) was used for analysis.
Results
Patient Characteristics and Treatment
A total of 97 eligible patients without pCR after NAC were identified to have a positive-to-negative change in HR status. The median age was 51 years (range 31-74), and 55.7% of these patients were premenopausal. Thirty patients (30.9%) had stage IIb disease, 37 patients (38.1%) exhibited stage IIIa disease and the rest of the patients (30.9%) stage IIIb to IIIc. After NAC, CR, PR or SD was observed in 20 patients (20.6%), 53 patients (54.6%) and 21 patients (21.6%), respectively. Only 3 patients (3.1%) had PD. After definitive surgery and appropriate postoperative chemotherapy and radiotherapy, 57 patients (58.8%) received ET (47 patients with tamoxifen only, 39 with aromatase inhibitors only, 11 with both) and 40 patients (41.2%) did not receive further treatment. The correlations between characteristics of patients and administration of ET are summarized in Table I. Patients treated with or without ET did not show any statistically significant difference in their clinical or pathologic characteristics.
PATIENT CHARACTERISTICS, STRATIFIED BY THE USE OF ADJUVANT ENDOCRINE THERAPY
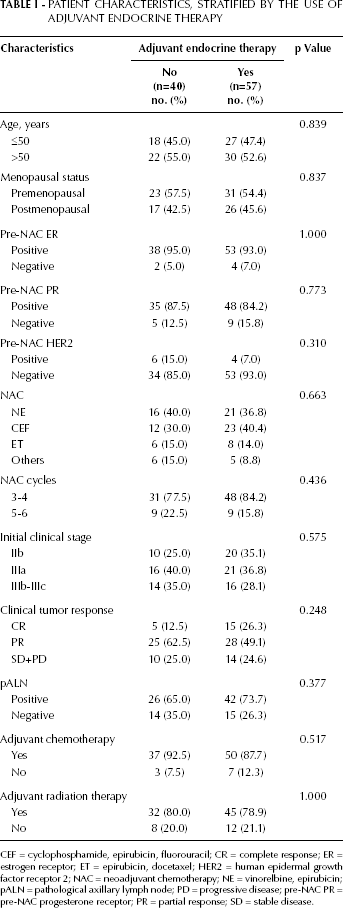
CEF = cyclophosphamide, epirubicin, fluorouracil; CR = complete response; ER = estrogen receptor; ET = epirubicin, docetaxel; HER2 = human epidermal growth factor receptor 2; NAC = neoadjuvant chemotherapy; NE = vinorelbine, epirubicin; pALN = pathological axillary lymph node; PD = progressive disease; pre-NAC PR = pre-NAC progesterone receptor; PR = partial response; SD = stable disease.
HR Status and HER2 Status
Seventy-seven patients (79.4%) had ER-positive/PR-positive tumors, 14 patients (14.4%) had ER-positive/PR-negative tumors and 6 patients (6.2%) displayed ER-negative/PR-positive tumors. A positive pre- and post-NAC HER2 status was observed in 10 patients (10.3%) and 11 patients (11.3%), respectively. Two patients had a positive-switch of HER2 status after NAC, and a negative-switch of HER2 status was observed in one patient. The pre-NAC ER/PR status and pre/post-NAC HER2 status are shown in Table II.
PATIENTS BY ER, PR AND HER2 STATUS BEFORE AND AFTER NEOADJUVANT CHEMOTHERAPY (N=97)

ER = estrogen receptor; HER2 = human epidermal growth factor receptor 2; NAC = neoadjuvant chemotherapy; PR = progesterone receptor.
DFS and OS
After a median follow-up of 68 months (range 14-103), the overall DFS rate was 61.9% and OS rate was 70.1%. Patients given adjuvant ET had a significantly longer DFS than patients not given ET (p=0.018, log-rank test) (Fig. 1). The 5-year DFS rates in ET-administered patients and ET-naïve patients were 77.0% and 55.5%, respectively. The use of adjuvant ET did not show OS benefit (p=0.053, log-rank test) (Fig. 2), but the 5-year OS rate for ET-administered patients was higher than that of ET-naïve patients (81.3% vs. 72.7%). In the exploratory subgroup analysis according to HER2 status, the administration of ET was also associated with better DFS in cases of HER2-positive (p=0.013), but not in HER2-negative disease (p=0.122) (Figs. 3 and 4). A univariate analysis summarized in Tab. III demonstrated that factors including pre-NAC HER2 positivity, higher clinical stage, positive lymph node involvement and omission of ET were significantly associated with worse DFS (p<0.05). Higher clinical stage at diagnosis and positive lymph node involvement were associated with worse OS (p<0.05).
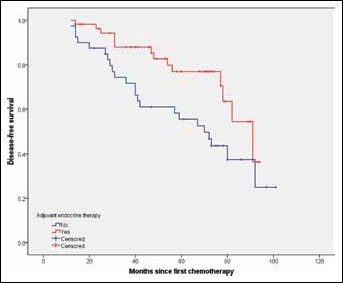
Comparison of DFS between patients given adjuvant ET and those not given adjuvant ET. DFS = disease-free survival; ET = endocrine therapy.
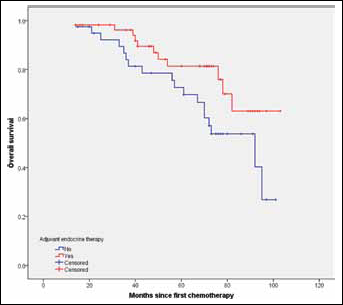
Comparison of OS between patients given adjuvant ET and those not given adjuvant ET. ET = endocrine therapy; OS = overall survival.
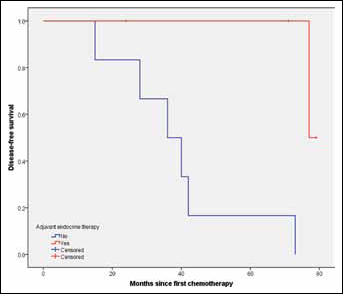
Comparison of DFS between patients given adjuvant ET and those not given adjuvant ET in HER2-positive subgroup. ET = endocrine therapy; DFS = disease-free survival; HER2 = human epidermal growth factor receptor 2.
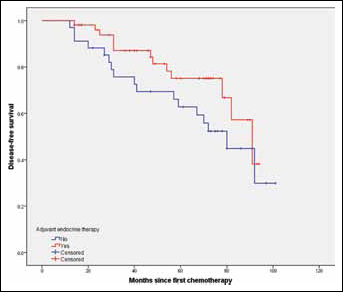
Comparison of DFS between patients given adjuvant ET and those not given adjuvant ET in HER2-negative subgroup. ET = endocrine therapy; HER2 = human epidermal growth factor receptor 2; OS = overall survival.
UNIVARIATE AND MULTIVARIATE PROGNOSTIC ANALYSIS OF DFS AND OS
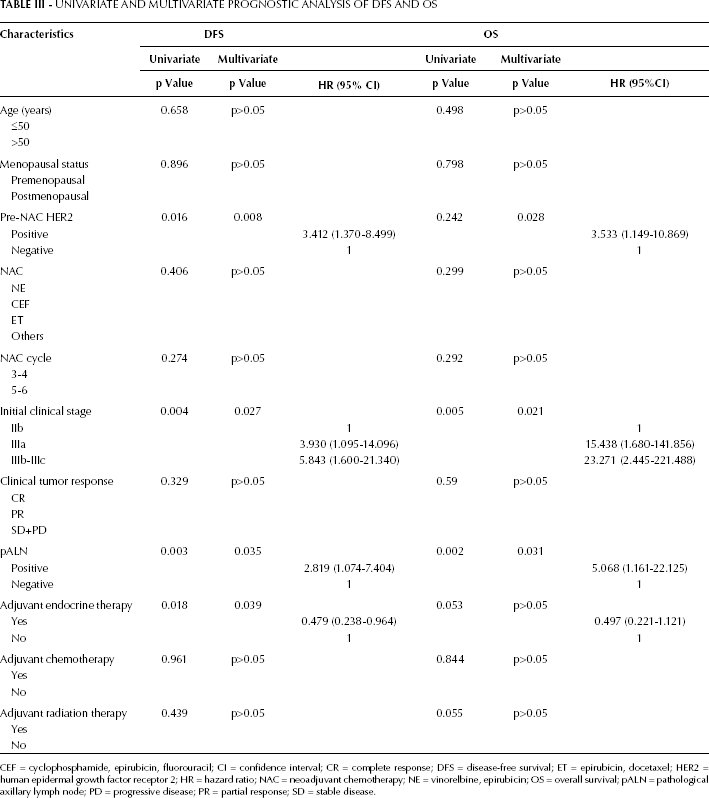
CEF = cyclophosphamide, epirubicin, fluorouracil; CI = confidence interval; CR = complete response; DFS = disease-free survival; ET = epirubicin, docetaxel; HER2 = human epidermal growth factor receptor 2; HR = hazard ratio; NAC = neoadjuvant chemotherapy; NE = vinorelbine, epirubicin; OS = overall survival; pALN = pathological axillary lymph node; PD = progressive disease; PR = partial response; SD = stable disease.
Multivariate Analysis
In a multivariate stepwise Cox regression analysis of DFS and OS illustrated in Table III, the following variables were included as probable prognostic factors in the Cox proportional hazard model: age (<50 vs. >50 years); menopausal status (premenopausal vs. postmenopausal); pre-NAC HER2 status (positive vs. negative); NAC regimen (NE vs. CEF vs. ET vs. others); NAC cycle(3 to 4 vs. 5 to 6), initial clinical stage (IIb vs. IIIa vs. IIIb to IIIc), clinical tumor response (CR vs. PR vs. SD+PD), pathological axillary lymph node status (positive vs. negative) and adjuvant therapies (ET, chemotherapy and radiation therapy, yes vs. no). Four of these variables – pre-NAC HER2 status, initial clinical stage, pathological axillary lymph node status and the use of adjuvant ET – were identified by the stepwise Cox regression model as independent predictors for DFS. Likewise, 3 of these variables – pre-NAC HER2 status, initial clinical stage and pathological axillary lymph node status – were identified as variables affecting OS.
Discussion
The discordance in HR and HER2 status between CNB and excision specimens has been reported in ample retrospective studies with inconsistent results. A meta-analysis by Chen et al confirmed the high diagnostic accuracy of CNB in evaluating HR and HER2 status compared with open excision biopsy in breast cancer patients without NAC (14). Zhang et al concluded in their meta-analysis that in patients receiving NAC, HR status is significantly altered by chemotherapy, and reevaluation of HR status after NAC should be considered (18). Taking the high accuracy of CNB in evaluating HR status into account, this kind of discordance is primarily caused by NAC itself.
Reported discordances in HR status range from 8% to 33% in small retrospective studies (10-11-12-13), and the reported changes are equally distributed between a positive and negative switch in HR status. ET would not be negligible in patients with a negative-to positive switch of HR status. Therefore, we paid more attention to the effect of ET in patients whose HR status changed from positive to negative. Our study is the first to focus on the effect of ET in patients who displayed a positive-to-negative switch in HR status after NAC. The long-term benefit of ET in this subpopulation of patients was confirmed. The 5-year DFS rates in ET-administered patients were significantly higher than those of ET-naïve patients (77.0% vs. 55.5%), whereas the 5-year OS rates had a marginally significant increase after the use of ET (81.3% vs. 72.7%). Similar results were reported in a previous study: in 59 patients who showed HR status conversion after NAC, the DFS of 47 ET-administered patients was significantly longer than that of the 12 ET-naïve patients (HR=0.19; 95% CI, 0.06-0.60, p<0.004) (11). However, this only included 30 patients with a positive-to-negative change of HR status, and all ET-naïve patients displayed a negative-to-positive change.
The mechanisms for a change in HR status in breast cancer caused by chemotherapy are complicated. In their critical review, van de Ven et al speculated that a change in HR status could be the result of lowered circulating levels of estrogens caused by ovarian insufficiency during chemotherapy, which might cause down-regulation of the ER and/or PR of the tumor, leading to estrogen-independent growth (19). Another theory postulated that chemotherapy could target chemosensitive tumor cells, particularly HR-negative cells and leave insensitive tumor cells such as HR-positive cells behind in the residual disease, leading to a negative-to-positive change of HR. Other possible explanations for a negative-to-positive change of HR may be the heterogeneity of the breast carcinoma as well as an insufficient CNB specimen sample. The reasoning proposed by van de Ven offers a plausible explanation for the markedly down-regulation of HR after NAC. Regardless of the underlying mechanism, the current study revealed that this type of breast cancer still responded to ET.
According to a recent international expert consensus, breast cancer can be divided into 4 intrinsic subtypes on the basis of ER, PR, HER2 and Ki67 status (20). Patients with different subtypes of breast cancer exhibit distinct responsiveness to systemic treatment as well as diverse outcomes (21-22-23). All of the patients in the current study had luminal breast cancers before NAC and basal-like or HER2-positive breast cancers after surgery. The results suggested that although apparently endocrine nonresponsive (e.g., basal-like or HER2-positive subtypes) after surgery, breast cancers with luminal subtype confirmed before NAC act like hormonally responsive breast cancer and had a favorable prognosis with the use of ET. Furthermore, luminal breast cancers have been reported to vary in sensitivity to ET, which may be largely, but not exclusively, affected by HER2 status. The multivariate analysis indicated that the benefit of ET was independent of HER2 status. HER2-positive breast cancers seemed to be more responsive than HER2-negative ones.
This study demonstrated that HR status can be evaluated in CNB specimens prior to treatment with NAC, and also post-NAC surgery. The use of ET is worth studying in a larger cohort of patients whose HR status changes from positive to negative prior to NAC treatment, and posttreatment.
