Abstract
Introduction/Aim
Methods
Result
Discussion
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) is one of the most common head and neck cancers in Southeast Asia, due to both genetic factors and diet containing carcinogenic nitrosamines such as salted fish or vegetables. In Thailand, the aged-standardized incidence rate (ASR) of NPC is 3.6 per 100,000 people (1) and the trend has been increasing (1). The standard treatment of locally advanced NPC is a combination of chemotherapy and radiotherapy, most commonly used as concurrent chemo-radiotherapy, which has shown survival benefit in several studies (2-3-4) (K. Thephamongkhol, Abstract ASCO 2004).
Although the combined chemo-radiotherapy can help NPC patients gain more survival time, some of them still have an unfavorable outcome. Therefore, some subgroups of NPC patients need different timings of treatment with combination of chemo-radiotherapy. Theoretically, induction chemotherapy has been used in some patients presenting limitations or technical difficulties in Intensity Modulated Radiotherapy (IMRT), such as the tumor being near critical structures (optic nerves and chiasm, brain stem), so as to reduce the tumor bulk near these structures. This strategy supports the use of IMRT more easily to get a potential improvement in local tumor control (5). Furthermore, induction chemotherapy also has the potential to control micro-metastases effectively before concurrent chemo-radiotherapy.
Even though some institutions have used this strategy routinely, either with induction chemotherapy followed by radiotherapy alone or with induction chemotherapy followed by concurrent chemo-radiotherapy, no promising survival benefit has been recorded yet because the pattern of failure of both strategies is from distant metastases. Some preliminary data have shown the benefit of adding further systemic therapy other than chemotherapy (6). Larger amounts of data are actually supporting this strategy in this subgroup of patients. As an example, the staging system from the International Union against Cancer/American Joint Committee on Cancer (UICC/AJCC), using TNM scoring, might not be enough to predict the pattern of failure and overall survival data (7). There might be some unknown prognostic factors that help identifying the high-risk group for distant metastases.
This study is designed to evaluate the prognostic properties of nodal stations in NPC, such as retropharyngeal lymph node (RP), posterior cervical lymph node (PCN, level V cervical lymph node), and supraclavicular lymph node (SPC), in stage IVA/IVB NPC patients in the setting of induction chemotherapy followed by concurrent chemo-radiotherapy.
Materials and Methods
This retrospective study has been approved by the institutional review board of the Siriraj hospital, Mahidol University, on 21th February 2012. The study included consecutive stage IVA/IVB NPC patients who were treated routinely with induction chemotherapy followed by concurrent chemo-radiotherapy due to bulky primary tumor and/or cervical lymph nodes at the division of radiation oncology, at the Siriraj hospital, from January 2007 to December 2010. Before treatment, complete staging was mandatory, including computed tomography (CT) or magnetic resonance imaging (MRI) of nasopharynx, Tc-99m Bone scan, ultrasound of the upper abdomen, chest radiograph, and blood test for liver and renal function. We excluded patients who had previously been treated with radiotherapy or chemotherapy, and had other cancers except basal cell carcinoma, CIN, or CIS of cervix. For the evaluation of treatment response, in addition to routine physical examination, enrolled patients needed to have available post-treatment clinical and/or imaging follow-up after induction chemotherapy and then at least 3 months after the following concurrent chemo-radiotherapy.
Chemotherapy
With regard to induction chemotherapy, several doublet and triplet chemotherapy regimens are available for NPC, such as platinum-based chemotherapy and 5-FU (1, 8, 9) platinum-based chemotherapy and docetaxel (10), and the combination of platinum-based chemotherapy, docetaxel, and 5-FU (11). According to our hospital policy, we used 3 cycles of induction chemotherapy composed of platinum-based chemotherapy and 5-FU for a number of reasons. Firstly, this regimen was extrapolated from Al-Sarraf (23) by substituting 3 cycles of adjuvant cisplatin and 5-FU with 3 cycles of induction platinum-based chemotherapy and 5-FU. Secondly, induction chemotherapy with cisplatin and docetaxel (10) is one of the commonly used regimens for NPC; however, this regimen seems to be too toxic for our patients needing more supportive care than that provided in our routine practice. Finally, the reimbursement system for our patients includes only platinum-based chemotherapy and 5-FU. As a result, most of our patients received only 3 cycles of platinum-based chemotherapy and 5-FU for induction chemotherapy, regardless of differences in WHO classification. Cisplatin was given at 80 mg/m2 on day 1, and 5-FU at 1,000 mg/m2 on day 1-4, for every 3 weeks. This combination was scheduled routinely for 3 cycles unless patient's intolerance or progression of the disease occurred. CT scan of nasopharynx and neck was also scheduled before concurrent chemo-radiotherapy. In concurrent phase, cisplatin was given at 100 mg/m2 every 3 weeks during radiation course. For patients who could not tolerate cisplatin, induction carboplatin was given with AUC5 at day 1 in combination with 5-FU 1,000 mg/m2 at day 1-4 for every 3 weeks, followed by concurrent weekly carboplatin (AUC2) during the radiation course.
Radiotherapy
The patients’ position was supine with neutral neck extension. Immobilizations were provided by a head-neck-shoulder thermoplastic mask and shoulder fixation. The CT simulation with contrast injection was performed with 3-mm thickness from vertex to 5 cm below the head of clavicle. All target volumes and normal structures were contoured by radiation oncologists. To improve the quality of gross target volume (GTV) delineation, the fusion of treatment planning CT with diagnostic CT or MRI was performed.
Target volume delineation and dose prescription were based on RTOG 0225 (12). Residual gross disease at nasopharynx and cervical lymph nodes after induction chemotherapy were delineated as GTV. Initial diseases before induction chemotherapy were contoured and included in the high-risk clinical target volume (CTV).
All patients were treated with 7-field IMRT techniques using simultaneous integrated boost techniques. The delivery of IMRT was obtained from 6 MV energy of photon linear accelerator with dynamic 120 multileaf collimator (MLC). Inverse treatment planning was performed using the Varian Eclipse treatment planning system. The treatment's schedule would be delivered once daily, 5 fractions per week and in 33 fractions, so that total treatment time would be finished within 7 weeks.
For all patients, we reviewed the information from hospital medical records and radiation oncology medical records. For calculating the survival time, we used the definition proposed by the FDA guideline (13). Being this a one-arm retrospective study, we used as starting date the day 1 of the first cycle of chemotherapy. The patients who were lost to follow-up were censored for progression free survival (PFS), distant metastasis free survival (DMFS) and regional recurrence free survival (RRFS). For overall survival (OS), we retrieved data from the citizen governmental registry. The RECIST1.1 classification (14) was used for evaluating the response to treatment and CTCAE v3.0 for evaluating acute and late toxicity (15).
Survival curves and survival rates were estimated by using the Kaplan-Meier method. Compliance, response rate, pattern of treatment failure, and toxicity were all reported as percentages. Univariate analysis was performed with the Cox regression model. The variables considered were the following: age, gender, pathology, ECOG performance status, 7thUICC/AJCC TNM staging, regimen of chemotherapy (cisplatin or carboplatin), number of cycles of chemotherapy, response rate after induction chemotherapy, response rate after following concurrent chemo-radiotherapy, overall treatment time, T stage, and N stage. The specific location of the nodes considered were the following: RP, PCN, SPC (positive with imaging or clinical detection), and laterality of node (unilateral, contralateral). The cutoffs of the variables were pre-established according to previous research in the Chinese population (16). Multivariate analysis was performed by using variables of clinical interest or variables that had a p value <0.2 in univariate analysis. The final model of multivariate analysis was modified by using the p value from the Wald's test and a backward elimination method (by likelihood ratio test and percent change of coefficient factors).
Results
We enrolled 68 patients with stage IVA/IVB NPC who were treated with induction chemotherapy followed by concurrent chemo-radiotherapy between January 2007 and December 2010 at the Siriraj Hospital. Twenty-five patients were excluded from our study: 14 were missing some radiotherapy data, 9 patients were treated without a platinum-based drug or had an unknown chemotherapy regimen, and 2 refused the treatment. Eventually, data from 43 patients were analyzed. The median follow-up time was 30 months (range, 11-63 months).
Twenty-four (55.8%) patients were stage IVA and 19 (44.2%) were stage IVB. All patients had good performance status (ECOG 0-1). In terms of primary nasopharyngeal tumor, 19 patients (44%) had a tumor confined in only one side of the nasopharynx and 24 patients (56%) had a tumor involving both sides of the nasopharynx. Regarding cervical nodal metastases, 22 patients (51.2%) had RP, 20 (46.2%) had PCN, 16 (37.2%) had lower neck node, and 14 (32.6%) had contralateral neck node metastases. However, SPC metastases were found only in 3 patients (7%) in our study. Patient characteristics are shown in Table I.
PATIENT CHARACTERISTICS
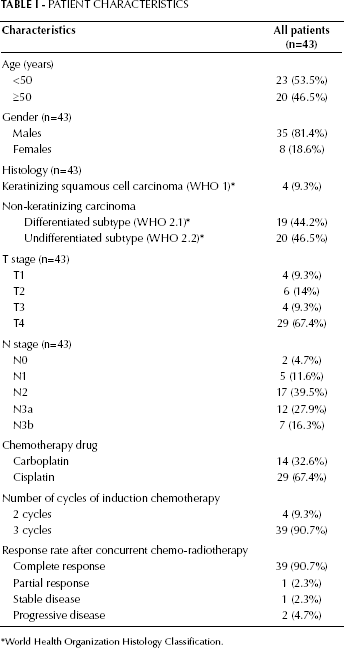
World Health Organization Histology Classification.
The treatment compliance was excellent. Overall, 90.7% of patients could complete 3 cycles of induction platinum-based chemotherapy and the remaining patients (9.3%) received 2 cycles of induction platinum-based chemotherapy. For the concurrent cisplatin chemotherapy administered to 36 patients, 29 (80.6%) received 2 or more cycles of cisplatin. Seven patients (19.4%) received only 1 cycle of cisplatin and then switched to weekly carboplatin due to poor performance status and impaired renal function. For the concurrent weekly carboplatin chemotherapy in 7 patients, 5 (71.4%) had 5 or more cycles of carboplatin. All patients received the complete course of radiation therapy. Only 2 patients had to delay their radiotherapy due to moist desquamation and severe nausea and vomiting. Median overall treatment time with radiotherapy was 51 days (range, 44-65 days). In our study, none of the patients had severe (grade 4-5) toxicity from chemotherapy and radiotherapy.
Regarding treatment response after induction chemotherapy, 23 patients (53.5%) achieved partial response and 20 (46.5%) achieved only stable disease. However, after concurrent chemo-radiotherapy, 39 patients (90.7%) achieved complete response. Among the remaining 4 patients 1 had a partial response, 1 a stable disease, and 2 had a progressive disease. After a median follow-up time of 30 months, the 2-year OS was 79.1%, and the 2-year PFS and DMFS were, respectively, 59.8% and 69.1%. Regarding the first site of failure, distant metastases and local and/or regional recurrences were equally distributed, respectively in 12 and 11 patients.
In univariate analysis, the nodal staging and SPC metastasis, specified as N3b, were not statistically significant in DMFS (HR 3.46; 95% CI 0.46-26.13; p=0.228 and HR 3.39; 95% CI 0.76-15.07; p=0.1, respectively), PFS (HR 2.25; 95% CI 0.52-9.67; p=0.276 and HR 2.59; 95% CI 0.59-11.34; p=0.21, respectively), or OS (HR 3.49; 95% CI 0.46-26.48; p=0.23 and HR 3.67; 95% CI 0.8-16.88; p=0.095, respectively); however, the RP region of nodal metastasis showed statistically significant differences in DMFS, PFS, and OS (HR 5.56, 95% CI 1.57-19.62, p=0.008; HR 2.79, 95% CI 1.12-6.96, p=0.028; and HR 3.12, 95% CI 1.07-9.06, p=0.036, respectively). Similarly, PNC metastasis indicated a significantly worse prognosis in DMFS and OS (HR 3.73; 95% CI 1.31-10.62; p=0.014 and HR 4.59; 95% CI 1.47-14.26; p=0.01). The lower neck node metastasis indicated a significant decrease of DMFS only (HR 3.04; 95% CI 1.15-7.99; p=0.024). Besides the nodal region, staging (IVA and IVB) and response after CCRT (CR and non-CR) were also able to predict some of the outcomes (Tab. II).
UNIVARIATE ANALYSES FOR DISTANT METASTASIS FREE SURVIVAL, PROGRESSION FREE SURVIVAL, AND OVERALL SURVIVAL
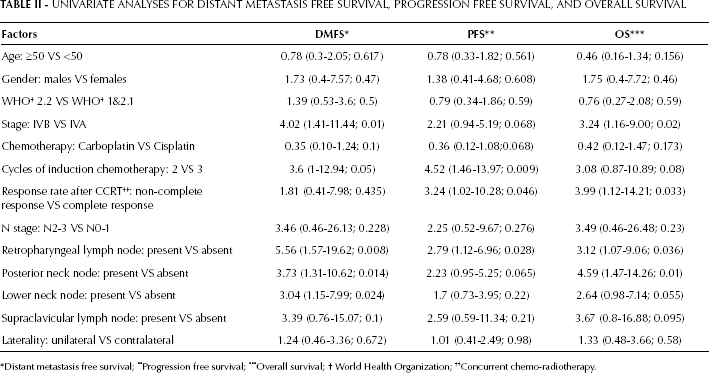
Distant metastasis free survival;
Progression free survival;
Overall survival;
World Health Organization;
Concurrent chemo-radiotherapy.
In a multivariate analysis to predict DMFS, SPC involvement (N3b) was not statistically significant (HR 3.39; 95% CI 0.76-15.07; p=0.1), whereas RP involvement was (HR 5.81; 95% CI 1.08-31.16; p=0.04). Moreover, and more importantly, PCN involvement was the only nodal factor to predict all the outcomes (DMFS, PFS, and OS). In other words, PCN was a strongly independent prognostic factor for DMFS, PFS, and OS (HR 5.57, 95% CI 1.12-27.71, p=0.036; HR 16.05, 95% CI 1.93-133.65, p=0.01; and HR 28.02, 95% CI 2.74-286.22, p=0.005, respectively) (Tab. III). In addition, the response rate after CCRT was also able to predict PFS and OS (HR 28.09, 95% CI 3.19-247.37, p=0.003; HR 31.98, 95% CI 3.55-287.92, p=0.002).
MULTIVARIATE ANALYSES FOR DISTANT METASTASIS FREE SURVIVAL, PROGRESSION FREE SURVIVAL, AND OVERALL SURVIVAL

Distant metastasis free survival;
Progression free survival;
Overall survival;
World Health Organization;
Concurrent chemo-radiotherapy.
Discussion
All our patients had locally advanced NPC stage IVA and IVB, a type of carcinoma that presents some difficulties with IMRT as the first line of treatment. Theoretically, induction chemotherapy could not only reduce tumor bulk but also control micro-metastases. In these groups of patients, IMRT treatment should be easier; however, in our study, the distant metastatic rate is still dominant. Therefore, we may need to find new effective prognostic predictors to enable the use of special treatments to control distant metastasis in this highly specific group of patients.
The most common and useful cancer staging system for solid tumors in adults is the UICC/AJCC, which uses the TNM scoring to classify the prognosis of particular cancer types. For NPC, the T staging predicts local disease and the N staging predicts distant metastasis (17). Furthermore, distant metastasis could affect PFS and OS of a particular patient. In our study, we focused our interest on the nodal group characteristics as an independent prognostic factor of locally advanced NPC treated with induction chemotherapy, a setting that might differ from the concurrent chemo-radiotherapy one.
Chen (18) reported that the 7th edition of the AJCC system defined a better predictor of prognosis than that of the 6th edition, especially with regard to the T1 and T2 groups; however, the N classification of the 7th AJCC edition showed only a slight difference between N0 and N3 in terms of DMFS and DFS when compared with the 6th AJCC edition. Moreover, N3a did not show differences in DMFS and DFS from N1, N2, and N3b. Therefore, besides the known characteristics of laterality and size of lymph node, there might be some unknown nodal characteristics, such as cervical lymph node groups or orderly lymph node station, causing differences in the N staging.
The spreading pattern of cervical lymph node (CLN) in NPC has been shown basically in an orderly fashion. The first echelon is RP and level II CLN, while the second echelon is level III-V CLN (19, 20). The incidence of skipped LN metastasis in NPC is as low as 0.5% (19). Therefore, our hypothesis is that second echelon metastasis in PCN, lower neck node, and contralateral lymph node, may increase the chance of distant metastasis and decrease survival. In other words, some of the second echelons may need to be changed from N1 to N2 disease to better discriminate the survival outcome with the standard staging system.
NPC is one of the few head and neck cancers that have posterior neck node metastasis. Nevertheless, only a limited number of studies has analyzed the specific posterior neck node as a prognostic factor to predict survival outcome of NPC. In our study, in the setting of induction chemotherapy, we found that PCN involvement was the only strongly independent prognostic factor predicting DMFS, which consequently turned to worse PFS and OS outcomes in patients with stage IVA/IVB NPC. On the other hand, a study from Mao (21) showed that level V CLN in nasopharyngeal carcinoma had the same prognosis of distant failure as that of RP, level IB/III CLN, when compared with level II CLN; however, in our study, this might be explained by the different groups compared (IVA stage vs. IVB stage) and the different treatments of NPC. As a result of this, we hypothesize that PCN metastasis may be classified as N2 disease due to obviously poor distant metastases and turning to worse OS. As far as we know, this is the first study to address this issue. This hypothesis needs to be further evaluated in larger cohorts of NPC patients, including different settings such as concurrent chemo-radiotherapy.
For lower neck node metastasis, some studies already investigated this lymph node group and their results present few differences in the classification. Li (7) classified level IV, Vb CLN, and SPC as lower neck node, a conclusion we also reached in our study; however, Mao (21) classified only level IV and SPC as lower group. The studies from Li (7) and Mao (21) show that the patients with positive lower neck node metastasis had significantly worse prognosis in distant metastasis. In our study, lower neck node metastasis showed worse distant control and trended towards a decrease in OS only in univariate analysis. This is possibly due to the very advanced cancer stages of the patients included in our study. The study by Li et al (7) included approximately 62% of patients with stage II and III NPC and their treatments included radiation alone, concurrent chemo-radiotherapy alone, and concurrent chemo-radiotherapy with induction or adjuvant chemotherapy. Clinical SPC-positivity in our study was not a significant prognostic factor in either univariate or multivariate analysis. This was likely due to the contribution of only 3 patients (7%) with positive SPC in our study.
The two studies from Li (7) and Mao (21) both show that laterality has a significantly worse prognosis in distant metastases. However, in our study, laterality did not show statistical significance in either univariate or multivariate analysis. This may be explained by the presence of 56% of patients in our study with primary nasopharyngeal mass involving both sides of the nasopharynx. Therefore, contralateral neck metastasis might not be the second echelon.
Nowadays, the primary purpose of treatment of locally advanced NPC is to control distant metastasis so as to improve OS. For instance, the RTOG 0615 study (6), which adds bevacizumab to standard platinum-based chemotherapy, seems to present better outcomes. Therefore, these prognostic factors from our and other studies might help choosing the specific patients who need an aggressive systemic treatment. Patients with positive posterior cervical neck node, lower neck node involvement, and contralateral neck node metastasis could be considered for targeted therapy, such as bevacizumab, or intensive chemotherapy, such as taxane-based chemotherapy.
We already know that RP is an ominous prognosis that predicted distant metastasis in both negative cervical lymph node and advanced-stage NPC (13, 15, 22). The 7th edition of the AJCC has already classified unilateral and bilateral RP metastasis as N1 staging. In our study, RP tends to have worse prognosis in distant metastasis, as also confirmed in other studies (13, 15, 22). However, this parameter was not shown to significantly decrease PFS and OS. This may be due to the setting of very advanced stage NPC (stage IVA/IVB) in our study or to the limited number of patients included.
Conclusion
PCN metastasis is hypothesized to be a highly accurate predictor for distant failure and worse OS in stage IVA/IVB NPC treated with conventional induction chemotherapy followed by concurrent chemo-radiotherapy. Therefore, regions of cervical neck node as PCN and lower neck node metastasis should also be considered and classified in a new staging system, besides lymph node size and laterality. Besides the classification in new staging, the presence of PCN metastasis might predict which cases need aggressive systemic treatment, such as more intense chemotherapy or added targeted therapy. This hypothesis regarding the possible prognostic factors and their treatment implications is very challenging and needs to be tested in a larger number of patients and in different settings.
Footnotes
List of Abbreviations
This study has been approved by the institutional review board, Siriraj Hospital, Mahidol University on February 21, 2012.
