Abstract
Background
Nasopharyngeal small cell carcinoma (SmCC) is a rare histological type of nasopharyngeal cancer, and its prognosis remains poor. This study aimed to determine the clinical characteristics and survival prognostic factors of nasopharyngeal SmCC.
Methods
Detailed clinicopathologic and therapeutic characteristics of a patient diagnosed with nasopharyngeal SmCC were determined. Nasopharyngeal SmCC cases reported previously were reviewed and summarized. Furthermore, a retrospective analysis was performed on data from the Surveillance, Epidemiology, and End Results (SEER) Program database. Kaplan–Meier analysis was conducted to compare survival within groups. Univariate and multivariate analyses were performed to investigate prognostic factors.
Results
A nasopharyngeal SmCC patient treated with chemoradiotherapy who achieved 46 months long-term survival was reported. In reviewing 16 reported cases with epidemiologic and therapeutic details, we found most of nasopharyngeal SmCC patients were diagnosed with advanced grades and received chemoradiotherapy. In total, 13,993 cases of nasopharyngeal cancer were extracted from the SEER database, from which 57 nasopharyngeal SmCC cases were eventually screened out. The mean age of the patients was 55.70 years, and 64.9% of these cases were either grade III or IV; the median overall survival (OS) was 18 months. Statistically significant differences were observed in the OS values of groups categorized by age (P = .025) or radiotherapy (P = .037). Age (<70 years) and radiotherapy were identified as independent survival and prognostic factors.
Conclusion
Patients with nasopharyngeal SmCC are usually diagnosed with advanced grades and have poor prognoses; nevertheless, they can benefit from radiotherapy with prolonged overall survival.
Keywords
Introduction
In 2018, 129,079 new cases of nasopharyngeal carcinoma (NPC) were reported globally 1 ; more than 70% of these cases occurred in East and Southeast Asia. 2 In recent years, most NPC patients have exhibited substantially reduced mortality and prolonged survival times, owing to the sensitivity of NPC to chemoradiotherapy. 2 Nevertheless, the prognosis of nasopharyngeal small cell carcinoma (SmCC), which is a relatively rare type of nasopharyngeal cancer, remains poor. 3 A population-based analysis of the National Cancer Database revealed that only .3% of all head and neck cancer patients were diagnosed as having SmCC, among which 10% were nasopharyngeal in origin. 4 Moreover, nasopharyngeal SmCC has been reported to have the worst prognosis of all SmCCs of the head and neck. 5
According to previous studies, the age of patients with SmCC varies from 5 to 80 years, and most nasopharyngeal SmCC cases have been found to be incredibly lethal, with poor survival outcomes; it has been demonstrated that patients are mainly in advanced stages when diagnosed with SmCC, and that they are prone to metastasis.3,6-19 To date, there are no standard therapeutic schemes or guidelines for nasopharyngeal SmCC, and this condition is so rare that to date it has not been listed in the World Health Organization (WHO) classification of nasopharyngeal cancer. Furthermore, its diagnosis and treatment are challenging. Current strategies for the therapeutic treatment of nasopharyngeal SmCC comprise chemotherapy and/or radiotherapy; surgery can also serve as a therapeutic modality for early stage nasopharyngeal SmCC.20-24 Unfortunately, there are few available studies in this regard. Various therapies, including surgery, radiotherapy, chemotherapy, immune therapy, and combined therapies,5,21,25,26 have been studied for SmCCs of the head and neck.
However, to date, there has been no large-scale population-based analysis of the specific clinical characteristics and survival prognostic factors of nasopharyngeal SmCC. Accordingly, in this study, we aimed to determine the clinical characteristics and survival prognostic factors of nasopharyngeal SmCC by studying the case of a patient with nasopharyngeal small cell neuroendocrine cancer who presented at the Fifth Affiliated Hospital of Sun Yat-sen University, reviewing relevant literatures and by screening and analyzing primary nasopharyngeal SmCC cases from the Surveillance, Epidemiology, and End Results (SEER) Program database.
Methods
Case Presentation
A patient was diagnosed with primary nasopharyngeal small cell neuroendocrine carcinoma at the Fifth Affiliated Hospital of Sun Yat-sen University, Zhuhai, China. According to the search results of medical records, this was the only case of nasopharyngeal SmCC in our hospital in the recent 10 years. Detailed information on his radiological and pathological images and results, course of treatment, and follow-up were collected and analyzed.
Immunohistochemistry was performed as described in previous studies. 27 Tissue specimens were fixed, sectioned, and stained according to standard procedures. Hematoxylin and eosin (H&E) staining was performed on tumor tissue samples, followed by immunohistochemical staining with Ki67 rabbit monoclonal antibody (1:400, #9027, Cell Signaling Technology, Boston, the United States), CD56 mouse monoclonal antibody (1:800, #3576, Cell Signaling Technology, Boston, the United States), pan-keratin mouse monoclonal antibody (1:200, #67306, Cell Signaling Technology, Boston, the United States), synaptophysin (SYN) rabbit monoclonal antibody (1:200, #36406, Cell Signaling Technology, Boston, the United States), chromogranin A (CgA) rabbit monoclonal antibody (MAB-0707, MXB Biotechnologies, Fuzhou, China), p40 monoclonal antibody (RMA-0815, MXB Biotechnologies, Fuzhou, China), and p63 rabbit monoclonal antibody (1:450, #39692, Cell Signaling Technology, Boston, the United States). Epstein-Barr virus (EBV)-encoded small ribonucleic acid (EBER) was detected by an EBER detecting kit (ISH-7001, ZSGB-BIO, Beijing, China).
Literature Review
An updated review of nasopharyngeal small cell cancer (SmCC) plus original report through October 2021 was conducted by searching PubMed for articles in any languages as well as their references, with the terms “SmCC,” “small cell cancer,” and “nasopharyngeal cancer.” And then, the epidemiologic features, therapeutic strategies, and survival status of reported cases including the present case were gathered and summarized.
Patient Selection From the SEER Database
Data concerning SmCC cases were obtained from the SEER database, which provides detailed patient outcomes and covers approximately 27.8% of the population of the United States of America (USA). In accordance with the primary site code C11.1-C11.9 and the International Classification of Disease, Oncology, third revision (ICD-O-3) code 8041/3, 60 cases were screened from 1975 to 2015; the primary site of each case was the nasopharynx, and the histology type was SmCC. Three cases with an overall survival (OS) of less than 1 month were excluded in the survival analysis session, considering that they might be in poor performance status or the time frame is too short for the treatment to be effective. The variables analyzed included age (for further analysis, patients were divided into 2 groups [age <70 or ≥70 years] according to previous studies28,29), sex, race, year of diagnosis, grade, histology, metastasis, surgery, radiotherapy, chemotherapy, and survival time.
Statistical Analysis
The clinical characteristics of patients with SmCC were examined and summarized in a table. The Kaplan–Meier estimator was used to describe the distribution of survival time. The reverse Kaplan–Meier methods were applied to estimate the median follow-up time. The log-rank test was used to compare survival between different strata. The OS was defined as the period of time from the date of diagnosis to the date of either death or the end of follow-up. Univariate and multivariate Cox regression analyses were performed to analyze the variables associated with OS. We checked the validation of proportional assumptions of the cox models as previously described, 30 and there were no significant violations. Variables with P-value <.1 in univariate analysis were included for multivariate analysis. The corresponding hazard ratios (HRs) and 95% confidence intervals (CIs) were estimated. A two-tailed P < .05 was considered to represent statistical significance. All statistical analyses were performed using the “survival” R package (v. 3.6.3) and IBM SPSS 26.0 software (IBM Corporation, Chicago, IL, USA).
Results
Case Presentation of a Nasopharyngeal SmCC Patient Exhibiting Complete Remission
On January 8, 2018, a 44-year-old male patient visited the head and neck surgery clinic of the Fifth Affiliated Hospital of Sun Yat-sen University, Zhuhai, China, complaining of nasal bleeding and a headache that had lasted for 4 days. A nasal endoscopic examination indicated the presence of a malignant mass in the nasopharynx, following which a biopsy of the nasopharyngeal neoplasm was performed. The final pathological diagnosis was nasopharyngeal small cell neuroendocrine cancer. Microscopic analysis revealed clusters of small cells (Figure 1 A and B), which were found to be immunopositive for CD56, creatine kinase (CK), Ki67, and SYN (Figure 1 (C)-(F)) and immunonegative for CgA, p63, p40, and EBER (Figure 1 (G)-(J)). (
The patient was then admitted to the Cancer Center of the Fifth Affiliated Hospital of Sun Yat-sen University for further diagnosis and treatment. Magnetic resonance imaging (MRI) confirmed the presence of a tumor mass, which was primarily located in the left side of the nasopharynx. It had invaded the sphenoid bone and the ethmoid, with retropharyngeal lymph node metastases occurring on both sides. A chest X-ray, abdominal ultrasound, and emission computed tomography bone scan all confirmed that there were no distal metastases. Thus, the tumor was staged as T4N2M0, IVA. The Eastern Cooperative Oncology Group performance status (ECOG-PS) of this patient was 0. Subsequently, the patient received intensity modulated radiotherapy for a total dose of 66 Gy with 33 fractions. They received concurrent chemotherapy, comprising 1 course of cisplatin followed by 2 courses of cisplatin (40 mg, day 1–3) and etoposide (150 mg, day 1–3). The patient completed radiotherapy and chemotherapy in June, 2018, following which they received a regular follow-up. MRI images of the tumor before and after therapy are shown in Figure 2 (A) and (B), respectively. The response was assessed as complete remission. As of the last follow-up date (23 November, 2021), the patient remains in complete remission, indicating that his OS is 46 months. Magnetic resonance imaging (MRI) images of the patient (
Literature Review of Reported Cases of Nasopharyngeal SmCC Patients
Literature review of reported nasopharyngeal small cell carcinoma cases.
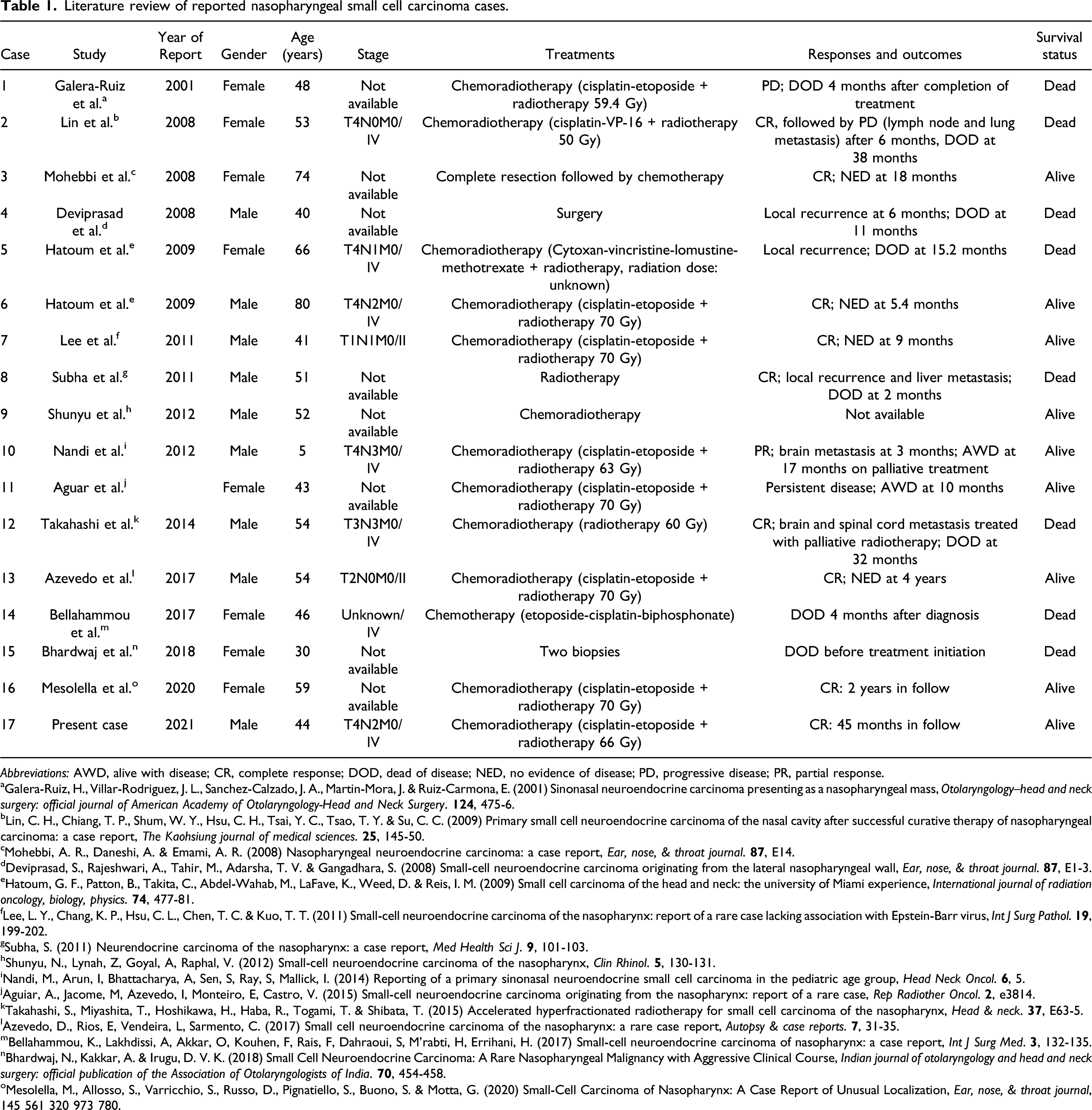
Abbreviations: AWD, alive with disease; CR, complete response; DOD, dead of disease; NED, no evidence of disease; PD, progressive disease; PR, partial response.
aGalera-Ruiz, H., Villar-Rodriguez, J. L., Sanchez-Calzado, J. A., Martin-Mora, J. & Ruiz-Carmona, E. (2001) Sinonasal neuroendocrine carcinoma presenting as a nasopharyngeal mass, Otolaryngology--head and neck surgery: official journal of American Academy of Otolaryngology-Head and Neck Surgery.
bLin, C. H., Chiang, T. P., Shum, W. Y., Hsu, C. H., Tsai, Y. C., Tsao, T. Y. & Su, C. C. (2009) Primary small cell neuroendocrine carcinoma of the nasal cavity after successful curative therapy of nasopharyngeal carcinoma: a case report, The Kaohsiung journal of medical sciences.
cMohebbi, A. R., Daneshi, A. & Emami, A. R. (2008) Nasopharyngeal neuroendocrine carcinoma: a case report, Ear, nose, & throat journal.
dDeviprasad, S., Rajeshwari, A., Tahir, M., Adarsha, T. V. & Gangadhara, S. (2008) Small-cell neuroendocrine carcinoma originating from the lateral nasopharyngeal wall, Ear, nose, & throat journal.
eHatoum, G. F., Patton, B., Takita, C., Abdel-Wahab, M., LaFave, K., Weed, D. & Reis, I. M. (2009) Small cell carcinoma of the head and neck: the university of Miami experience, International journal of radiation oncology, biology, physics.
fLee, L. Y., Chang, K. P., Hsu, C. L., Chen, T. C. & Kuo, T. T. (2011) Small-cell neuroendocrine carcinoma of the nasopharynx: report of a rare case lacking association with Epstein-Barr virus, Int J Surg Pathol.
gSubha, S. (2011) Neurendocrine carcinoma of the nasopharynx: a case report, Med Health Sci J.
hShunyu, N., Lynah, Z, Goyal, A, Raphal, V. (2012) Small-cell neuroendocrine carcinoma of the nasopharynx, Clin Rhinol.
iNandi, M., Arun, I, Bhattacharya, A, Sen, S, Ray, S, Mallick, I. (2014) Reporting of a primary sinonasal neuroendocrine small cell carcinoma in the pediatric age group, Head Neck Oncol.
jAguiar, A., Jacome, M, Azevedo, I, Monteiro, E, Castro, V. (2015) Small-cell neuroendocrine carcinoma originating from the nasopharynx: report of a rare case, Rep Radiother Oncol.
kTakahashi, S., Miyashita, T., Hoshikawa, H., Haba, R., Togami, T. & Shibata, T. (2015) Accelerated hyperfractionated radiotherapy for small cell carcinoma of the nasopharynx, Head & neck.
lAzevedo, D., Rios, E, Vendeira, L, Sarmento, C. (2017) Small cell neuroendocrine carcinoma of the nasopharynx: a rare case report, Autopsy & case reports.
mBellahammou, K., Lakhdissi, A, Akkar, O, Kouhen, F, Rais, F, Dahraoui, S, M’rabti, H, Errihani, H. (2017) Small-cell neuroendocrine carcinoma of nasopharynx: a case report, Int J Surg Med.
nBhardwaj, N., Kakkar, A. & Irugu, D. V. K. (2018) Small Cell Neuroendocrine Carcinoma: A Rare Nasopharyngeal Malignancy with Aggressive Clinical Course, Indian journal of otolaryngology and head and neck surgery: official publication of the Association of Otolaryngologists of India.
oMesolella, M., Allosso, S., Varricchio, S., Russo, D., Pignatiello, S., Buono, S. & Motta, G. (2020) Small-Cell Carcinoma of Nasopharynx: A Case Report of Unusual Localization, Ear, nose, & throat journal, 145 561 320 973 780.
Clinical Characteristics of Nasopharyngeal SmCC Patients in the SEER Database
Clinical characteristics of nasopharyngeal small cell carcinoma.

aOther: American Indian/Alaska Native, Asian/Pacific Islander.

Patient selection flow diagram.
Survival Analysis of Different Clinical Features
The median OS of patients (n = 57) was 18 months (95% CI = 13.00–35.00 months; Figure 4) and the median follow-up time was 138 months. Survival analysis showed that the OS values of patients who were <70 years old (P = .019), male (P = .022), grade III (P = .048), and received radiotherapy (P = .029), were significantly better than those patients who were ≥70 years old, female, grade IV, and were in the “no/unknown radiotherapy” group (Figure 5(A), (C), (E), and (J)). No statistically significant differences were observed upon categorizing the groups by race, year of diagnosis, T stage, N stage, surgery, and chemotherapy (Figure 5(B), (D), (F), (G), (H), and (i)). Using the “Survival” R package, the one-, three-, and 5-year survival rates of all patients were determined to be 62.6, 33.9, and 29.6%, respectively. The one-, three-, and 5-year survival rates of patients who received therapies featuring radiation and chemotherapy were 62.9, 62.5, and 56.3%, respectively. Survival analysis using Kaplan–Meier method and log-rank test; P < .05 was considered statistically significant. Median overall survival (OS) is marked with horizontal and vertical dashed lines. Median OS of all patients (n = 57) = 18 months; 95% confidence intervals (CIs) = 13.00–35.00 months. OS among groups divided by (

Prognostic Factors of Nasopharyngeal SmCC
Univariate and multivariate Cox analysis.

aOther: American Indian/Alaska Native, Asian/Pacific Islander.
Discussion
This study presents the first large-scale population-based study revealing the clinical features and prognostic factors of nasopharyngeal SmCC. The clinical characteristics, survival outcomes, and prognostic factors of nasopharyngeal SmCC were analyzed. Here, nasopharyngeal SmCC was found to be an aggressive carcinoma with poor one- (62.6%), three- (33.9%), and 5-year (29.6%) OS rates. Furthermore, the tumor pathological grade was found to be important for prognosis, with 64.9% of nasopharyngeal SmCC patients being in grade III or IV (poorly differentiated or undifferentiated); this might have contributed to their poor prognosis. The mean age of patients with nasopharyngeal SmCC was 55.70 years. Patients aged under 70 years showed better survival outcomes than those aged over 70 years. The combination of radiotherapy and chemotherapy was the most common choice. Radiotherapy was revealed to be an independent prognostic factor for a longer OS; chemotherapy might also have been associated with a longer OS. Based on the findings presented in this study, nasopharyngeal SmCC should be listed as a distinct classification of nasopharyngeal cancer.
Kaplan–Meier survival analysis revealed different OS values within groups, but it was not possible to clearly identify patients who did not receive radiation therapy based on the SEER database. That is, those patients in the “no or unknown radiotherapy group” might have also received radiotherapy or other treatments. Nevertheless, radiotherapy, chemotherapy, and surgery were grouped together during multivariate analysis, which theoretically corrected for confounders among groups. This analysis revealed that a better OS was achieved in the radiotherapy group than in the “no or unknown radiotherapy group.” Furthermore, multivariate analysis revealed the statistical significance of radiotherapy, showing that it was still an independent predictor of OS. The P-value of chemotherapy was revealed to be <.1 through multivariate analysis, indicating that chemotherapy might also be an independent predictor. However, this needs to be further confirmed by larger clinical studies in the future.
Pertaining to treatment delivery, though most previous studies have focused on extrapulmonary SmCC cases where the patients underwent surgery, 31 surgery is not usually recommended for nasopharyngeal SmCC patients. Here, no difference in survival was observed between the surgery and non-surgery groups. However, patients with nasopharyngeal SmCC were shown to have been mostly treated with radiation therapy combined with chemotherapy and had appreciable one- (62.9%), three- (62.5%), and 5-year (56.3%) OS rates. Similarly, most of the cases reported in the literature have received radiation therapy concurrent with chemotherapy, suggesting that chemoradiotherapy benefits SmCC patients, compared to chemotherapy or surgery alone.6,9-13,15-19 This finding indicates that chemoradiotherapy might be a possible curative therapeutic choice for nasopharyngeal SmCC. The complete response and sustained survival observed in the case presented here also suggest the potential benefit of combination therapy for patients with nasopharyngeal SmCC. To abrogate the bias, however, larger number of patients need to be analyzed. Furthermore, stratification is generally recommended, but here, considering that more biases would be generated if stratification was performed, the previous results of multivariate analysis were retained. In summary, combined therapies, particularly the combination of radiation and chemotherapy, should be considered as the first-line choice of treatment for nasopharyngeal SmCC.
Undifferentiated nasopharyngeal carcinoma type (UNCT) is the most histological type of NPC; it has been verified as a distinct entity and has been demonstrated to exhibit radiosensitivity and chemosensitivity. However, because of its rarity, most treatment options for nasopharyngeal SmCC have instead been determined depending on experience. Furthermore, females with UNCT have exhibited better outcomes than males, which is contrary to the trend observed for SmCC. 32 Thus, more clinical trials are needed to validate treatment options for nasopharyngeal SmCC in the future. In addition, further efforts should focus on screening the molecular and genetic characteristics of nasopharyngeal SmCC. Studies are being conducted to further explore different approaches to treat extrapulmonary SmCCs, especially in the field of immunotherapy.31,33,34 Similarly, immunotherapy could also play an effective role in treating nasopharyngeal SmCC. To confirm the above-mentioned hypothesis, genetic and tumor microenvironment characteristics should be taken into consideration,35-37 which would help to reveal the landscape of tumor mutation burden, immune infiltration, and the distribution of immune checkpoint biomarkers of nasopharyngeal SmCC. This will permit the exploration of more specific schemes to make nasopharyngeal SmCC a potentially curable tumor.
This study has some limitations. First, authorization was not obtained to access detailed data regarding chemotherapeutic regimens, dosages of radiation therapy, and the specific time of surgery. Consequently, it was not possible to identify the suitable chemotherapy regimen or the most beneficial radiation dose and its fraction. Second, the population of nasopharyngeal SmCC cases was small because only a small proportion of American cancer patients have been recorded in the SEER database. Moreover, the information regarding the stages of each case was incomplete, and the metastasis status was unknown for more than half of the patients. Hence, some biases could have existed in the analysis of the prognostic roles of stages. In addition, the lack of ECOG-PS data from the SEER database may also contribute biases in our analysis. Furthermore, variations in genetic backgrounds may be one of the reasons for different therapeutic responses and outcomes in the nasopharyngeal SmCC patients studied here. Therefore, more nations and races, especially patients from areas where nasopharyngeal cancer is endemic, need to be included in future studies.
In conclusion, nasopharyngeal SmCC patients were found to have advanced grade cancers and poor prognoses. Treating nasopharyngeal SmCC remains challenging. This study revealed the clinical characteristics and prognosis factors of nasopharyngeal SmCC. It was found that nasopharyngeal SmCC patients can benefit from radiotherapy, which could contribute to decision-making regarding the clinical treatment of this condition. In view of the lethal nature of this cancer, more studies and clinical trials regarding nasopharyngeal SmCC should be performed in the future.
Footnotes
Acknowledgments
The authors thank the efforts of the Surveillance, Epidemiology, and End Results (SEER) Cancer Registries.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The ethical approval (number: K306-1) for reporting individual cases or case series was obtained from our institution (the Affiliated hospital of Sun Yat-sen University, Zhuhai, China). Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Data Availability Statement
All data support the findings of this study are openly available in the SEER database after official registration and authorization.
