Abstract
CD44 is a transmembrane glycoprotein and is regarded as a potential marker in various tumors. The aim of our study was to analyze the expression of the standard form of CD44 (CD44s) and its isoforms in localized prostate cancer (PCa), and to correlate these data with the classical prognostic factors and biochemical recurrence.
Ninety-four surgical specimens were analyzed in this study. The expression levels of CD44s and all its 9 variants were analyzed by quantitative real time PCR (qRT-PCR). The control group consisted of 14 specimens from patients with benign prostatic hyperplasia. We correlated all the expression profiles with biochemical recurrence, as defined by a PSA >0.4 ng/mL in a mean follow-up period of 53.3 months.
In PCa, CD44s was underexpressed and all the other isoforms were overexpressed. The mean expression level of most variants was higher in patients who had not recurred, and a higher expression of CD44v2 independently correlated with a better recurrence-free survival rate (p=0.045). This variant was also underexpressed in metastatic PCa cell lines. There was no correlation between the expression levels of any of the CD44 isoforms and the classical prognostic factors.
We here demonstrated that PCa cases are characterized by a change in the expression of CD44, with a loss of CD44s and an overexpression of all the other CD44 variants. However, during cancer progression we found a loss of expression of all CD44 variants, and a correlation between higher expression of CD44v2 and a better recurrence-free survival rate. The understanding of the CD44 expression patterns in PCa could contribute to its use as a new prognostic marker.
Keywords
Introduction
Prostate cancer (PCa) is the most common cancer in men and the second leading cause of cancer-related death in western countries (1). With the introduction of prostate-specific antigen (PSA) as a diagnostic marker, as well as of screening programs for PCa early diagnosis, the stage at which these tumors are diagnosed has shifted (2). However, despite PSA being an organ-specific marker, it is not disease-specific, as PSA levels may also rise in benign conditions (3). Therefore, efforts are being made to find new diagnostic and prognostic markers.
CD44 is a multifunctional cell surface molecule and a transmembrane glycoprotein that is involved in various cellular activities, such as cell-to-cell and cell-to-extracellular matrix interactions, cell trafficking, migration, lymph node homing, presentation of growth factors, regulation of chemokines and cytokines, signal transmission from the cell surface to the interior of the cell, apoptosis, and cell survival. CD44 is expressed by cancer initiating cells, which are highly resistant to apoptosis and are essential for metastasis formation after prolonged dormancy (4, 5). Because of its broad role in cellular homeostasis, CD44 has been studied as a potential marker of aggressiveness in various neoplasms.
Tumorigenesis promotes alternative splicing and post-transcriptional modifications in the CD44 messenger RNA (mRNA) to produce a number of CD44 isoforms, including isoforms that have a higher affinity for hyaluronic acid (HA), a factor that is implicated in tumorigenesis (6). The CD44 mRNA consists of 20 exons, of which 10 undergo alternative splicing to form 10 different variants (v1-v10), collectively known as CD44v. The other 10 exons comprise the standard form of CD44, known as CD44s (7).
CD44v expression was previously found to be associated with the development and aggressiveness of breast cancer (8), bladder cancer (9), and colorectal carcinoma (10). However, only a few studies until now have explored the CD44 variants expressed in PCa, mostly using immunohistochemistry.
The purpose of this study is to analyze the expression of all CD44 variants (v2, v3, v4, v5, v6, v7, v8, v9, v10) and CD44s using quantitative real time PCR (qRT-PCR) in clinical specimens of PCa, and to correlate these values with classical prognostic factors and biochemical recurrence. This is the first study to evaluate the expression profile of CD44s and all its variants in this disease.
Materials and Methods
The study population consisted of 94 patients with PCa who underwent radical prostatectomy between December 1997 and August 2001. The mean age of the patients was 63.6 years (SD, 8 years), the mean PSA serum level was 9.4 ng/mL (SD, 4.8 ng/mL), 56.8% of the patients were staged pT2, and the mean Gleason score was 7.1 (SD, 1.3). Fourteen subjects with benign prostatic hyperplasia (BPH) were enrolled as controls. The mean age of the subjects in the control group was 70.1 years (SD, 6.2 years), and their mean PSA was 2.4 ng/mL (SD, 1.4 ng/mL). All patients underwent surgery by the same surgeon and provided informed consent to participate in the study and to donate their biological samples for analysis. The study was approved by the Institutional Board of Ethics (n° 0340/10).
For the part of the study conducted in vitro, we used 3 different cell lines (DU145, PC3, and LNCaP), which were obtained from ATCC and were maintained in DMEM supplemented with 10% fetal bovine serum, and 100 U/mL penicillin.
RNA extraction
Fresh surgical specimens were examined immediately after resection by the pathologist, and 1-cm3 sections were frozen and stored at -170°C in liquid nitrogen. A mirror fragment was processed routinely and stained with hematoxilin and eosin to guarantee that the frozen fragment contained at least 75% of tumor cells. With regard to the cell lines, 1x105 cells were used for RNA extraction.
Total RNA was isolated with an RNAaqueous Kit (Applied Biosystems, Foster City, CA, USA) according to the manufacturer's instructions. RNA concentration was determined by measuring absorbance at 260/280 nm using a Nanodrop ND-1000 spectrophotometer (Thermo Scientific, Asheville, NC, USA). cDNA was generated using a High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA). The reactions were incubated at 25°C for 10 minutes, followed by 120 minutes at 37°C, and 5 minutes at 85°C. The cDNA was stored at -20°C until use.
qRT-PCR and gene expression
Expression levels of all variants and CD44s were analyzed by qRT-PCR using an ABI 7500 Fast Real-Time PCR System (Applied Biosystems). Target sequences were amplified in a 10-µL reaction containing: 0.75 µL of the reverse primer and 0.75 µL of the isoform-specific forward primer (Tab. I), 5 µL of SYBR® Green (Applied Biosystems, Foster City, CA, EUA), and 1 µL of cDNA. The PCR cycling conditions were 2 minutes at 50°C, 10 minutes at 95°C, followed by 40 cycles of 15 seconds at 95°C, 1 minute at 60°C, and 20 seconds at 58°C. The β2M assay was used as the endogenous control.
Primer Pairs Used For Quantification Of Cd44 Expression

Statistical analysis
The ΔΔCT method was used to calculate the relative expression of the target genes using the formula ΔΔCT=(CT target gene, PCa sample - CT endogenous control, PCa sample) – (CT target gene, BPH tissue sample - CT endogenous control, BPH). The fold change in gene expression was calculated as 2-ΔΔCT.
To analyze the expression levels according to pathological stage, grade, PSA value, and recurrence, we used the Student's t-test for homogeneous variables and the Mann-Whitney test for heterogeneous variables. The ANOVA test was used for multivariate analysis considering the expression of CD44v2, Gleason score, and tumor stage. Statistical analysis was performed using SPSS 19.0 for Windows, and the results were considered significant if p≤0.05. Kaplan-Meir curves were constructed for the analysis of recurrence-free survival.
Results
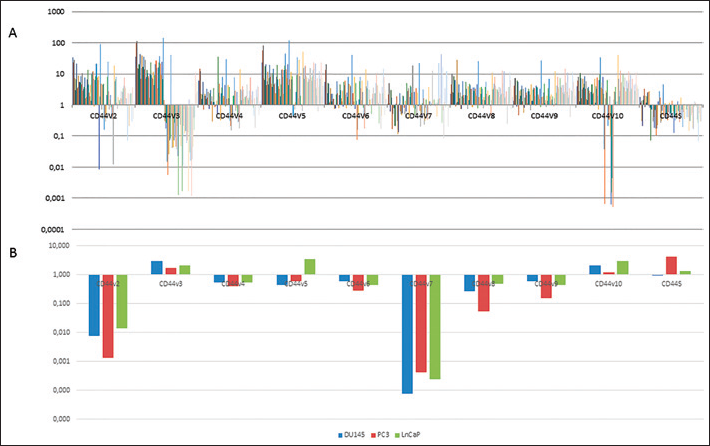
In 71.2% of the PCa cases we found a decrease in the expression of CD44s together with an overexpression of all variants generated by alternative splicing, compared to the BPH controls. By contrast, in a representative metastatic PCa cell line, we found a loss of expression of almost all CD44v, except for CD44v3 and CD44v10. The CD44v5 was overexpressed only in LnCaP, and the CD44s was expressed only in PC3 and LnCaP (Fig. 1, Tab. II).
Expression Of Cd44S And Its Splicing Variants In Prostate Cancer Patients Compared To Bph
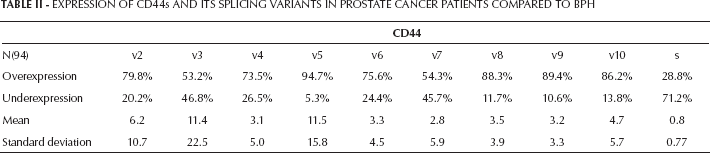
(A) CD44v and CD44s expression profiles in 94 patients with PCa compared to the BPH controls, calculated using the 2-ΔΔCT method. (B) CD44v and CD44s expression in metastatic prostate cancer cell lines compared to BPH controls.
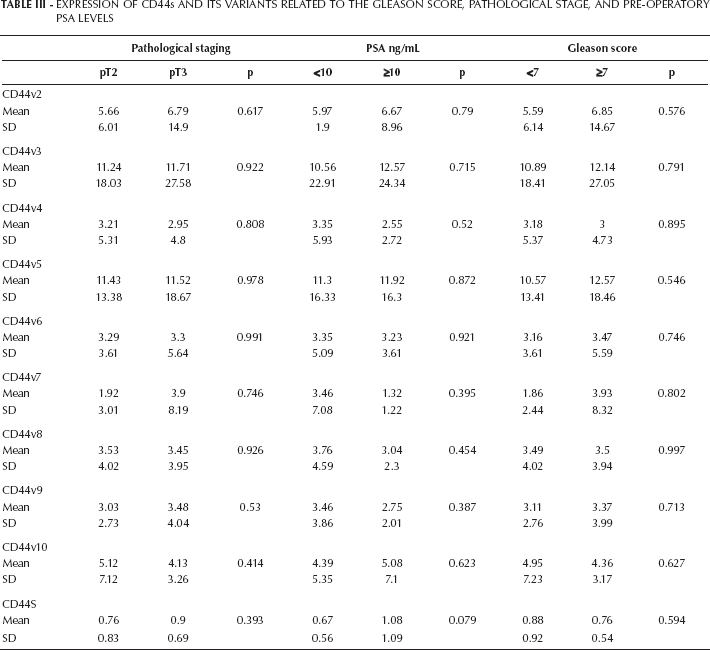
We did not observe a correlation between the expression profile of CD44s nor all its variants and the Gleason score, pathological stage, or pre-operatory PSA levels (Tab. III).
Expression Of Cd44S And Its Variants Related To The Gleason Score, Pathological Stage, And Pre-Operatory Psa Levels
The relationship between the expression of CD44v and the recurrence-free survival rate was examined in 90 patients with PCa during a mean follow-up period of 53.3 months. Biochemical recurrence was considered to occur when the PSA level was >0.4 ng/mL. In addition to a change in the expression levels of CD44 from the standard form to the variant forms, the mean expression of all variants, except CD44v7, was higher in patients who did not recur (Tab. IV). In addition, the Kaplan-Meier curve demonstrated that patients with higher expression levels of CD44v2 had a better survival rate (p=0.044) (Fig. 2). Our multivariate analysis including Gleason score, tumor stage, and CD44v2 expression showed that a higher expression of CD44v2 was independently related to the biochemical recurrence-free survival rate, with a risk to recur of 3.96 when CD44v2 ≤6.16 (p=0.045).
Cd44 Expression Related To Biochemical Recurrence After Radical Prostatectomy
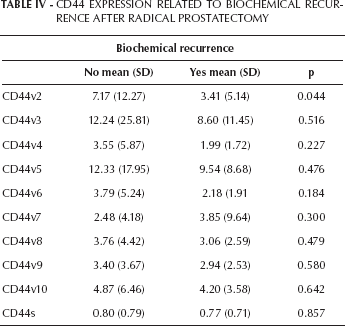
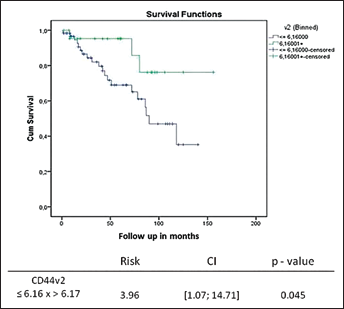
Recurrence-free survival curve of 90 patients in relation to their CD44v2 expression. Patients who did not experience biochemical recurrence had a higher expression of CD44v2 (p=0.044).
Discussion
This is the first study to quantify the expression of all variants of CD44 in clinical specimens of PCa. Our results show that although CD44s was substituted by all its splicing variants, a lower expression of CD44v2 independently correlated with a 4 times greater risk of biochemical recurrence.
Previous studies that only used immunohistochemistry demonstrated a loss of CD44s in PCa and increased levels of the variant forms (11, 12).
CD44 is the main receptor for the glycosaminogly-can hyaluronan, which is the main component of the extracellular matrix in mammalian tissues. During the rapid matrix remodeling that occurs during embryogenesis, inflammation, and tumorigenesis, there is an increase in the amount of hyaluronan attached to enzymes and receptors. The regulation of the interaction between hyaluronan and hyaluronan-binding proteins is crucial for several physiological processes, and the disturbance of these interactions has been related to tumorigenesis. As to be able to function in signaling, hyaluronan must bind to the constitutively active CD44 variant, which is one of the 3 forms of CD44. The other 2 forms of CD44 include a form that is unable to bind to hyaluronan and a form that can bind only when activated by physiological stimuli. CD44 is encoded by a single gene, but it undergoes alternative splicing to generate several different isoforms. The shortest form is CD44s, expressed by normal tissues, while its isoforms contain a variable number of exon insertions at the proximal plasma membrane external region and are expressed by cancer cells. The additional exons in the ectodomain can be decorated with chondroitin sulfate and/or heparin sulfate, which enable CD44 to bind to growth factors. The standard and smaller form of CD44 binds only to ankyrin and ezrin/radixin/moesin proteins to provide a link to the cytoskeleton while abrogating binding to growth factors. However, the interaction between CD44 and hyaluronan synthases or hyaluronidases is much more complex and varies among different organs. This complexity may explain why CD44 is related to a poor prognosis in some neoplasias, but not in PCa (13).
Previous research suggested that CD44s is only expressed in benign prostate tissue and that this expression is lost during carcinogenesis (12). It is expected that methylation of the CD44s promoter would be the mechanism through which CD44s expression is downregulated, and methylation of the CD44 promoter has been correlated with PCa progression (14, 15). As an example, Lou and colleagues demonstrated that methylation of the CD44 promoter occurs in 77.5% of primary PCa specimens and 75% of distant metastases, but in only 10% of matched normal tissues (16).
In addition, microRNA (miRNA)-mediated post-transcriptional regulation may play an important role in CD44s downregulation in cancer. A previous study published by Yang et al showed that the miRNAs 373 and 520c are expected to target CD44s to promote its downregulation, and overexpression of these miRNAs is related to carcinogenesis (17).
Methylation, however, cannot account for the increased expression of the variant isoforms. Since CD44v expression is characteristic of cancer tissues, alternative splicing must also play a major role in tumorigenesis. These variants have an increased ability to interact with HA, which in turn activates several tyrosine kinase receptors and can serve as co-receptors for growth factors, such as hepatocyte growth factor (HGF), fibroblasts growth factor (FGF) and vascular endothelial growth factor (VEGF). These interactions with growth factors upregulate the MAPK and PI3K pathways, facilitating cell invasion of tissues (4). However, despite some studies have tried to address the mechanism through which the CD44 variants interfere with tumor behavior, the role of these variants is not completely understood. CD44v9 forms a complex with HGF and its receptor, c-Met, which activates the tyrosine kinase signaling pathway to promote cell proliferation. HGF promotes the synthesis of CD44v9, which interacts with stromal-derived hyaluronan to stabilize the androgen receptor and inhibit apoptosis (18). Furthermore, Robbins et al demonstrated that calcitonin acts as a paracrine growth factor to upregulate CD44 variants in PCa by facilitating the splicing process through the protein kinase A and p38 pathways (19).
In accordance with our results, the higher expression level of the isoforms CD44v7-10 has been described in the literature as being important in the development of PCa (especially, CD44v9 has been shown to increase cell invasiveness), but none of these studies refer to PCa behavior. We believe that the overexpression of CD44 variants may promote cancer during the first steps of carcinogenesis, and that after cell dissemination their expression may act more as a tumor suppressor (20). To support this theory we showed that there is no correlation between the expression of CD44s and CD44v and the main prognostic factors.
Interestingly, when considering patients with biochemical recurrence, we observed that all variants, except CD44v7, were downregulated compared to patients who did not experience biochemical recurrence. In addition, by analyzing the biochemical recurrence-free survival, the Kaplan-Meier curve showed that a higher expression of CD44v2 correlated with tumors that have not recurred (p=0.044). Interestingly, in 3 different cell lines representative of metastatic PCa this variant was downregulated. In addition, the multivariate analysis including Gleason score and tumor stage showed that expression of CD44v2 was independently correlated to biochemical recurrence-free survival rates. Recently, Tei et al analyzed 160 surgical specimens of PCa and found that CD44v6 expression was also an independent predictive factor of BR (21). In esophageal squamous cell carcinoma, Gotoda et al found by immunohistochemistry that CD44v2 expression correlated with a significantly better prognosis compared to tumors without CD44v2 expression (22).
In conclusion, in PCa there is a loss of CD44s expression during tumorigenesis and, as a result of alternative splicing, there is a corresponding increase in the expression of its variants. We believe that this phenomenon is important in the early stages of carcinogenesis due to modifications of signaling pathways and changes in cellular activities. During cancer progression these variants are also lost, and this loss facilitates tumor recurrence and dissemination. Based on our results, we propose CD44v2 as a prognostic marker for PCa.
