Abstract
The basal cell layer has emerged as a critical player in cancer progression, and understanding the molecular contribution of specific cell types is important in treatment and prevention. The adhesion receptor CD44, which mediates epithelial-stromal and cell-cell interactions, has been shown to both promote and suppress tumor progression. To better understand the normal function of CD44, we have investigated its role in mouse mammary gland development and its expression in human breast and prostate cancer. We have found that CD44 is expressed in the myoepithelium of the developing mammary gland and modulates ductal development of FVB/N mice. The loss of CD44 results in defective luminal-myoepithelial cell-cell adhesion and promotes the mixing of luminal and myoepithelial layers, disrupting epithelial bilayer organization, and CD44-null mice experience delayed ductal outgrowth and impaired terminal end bud formation. The myoepithelial expression of CD44 is also relevant to its expression in cancer, as CD44 is expressed in the basal cells of early-stage breast and prostate cancer but exhibits altered localization with increasing tumorigenicity and is strongly expressed by tumor epithelium.
Keywords
Introduction
The mammary gland is comprised of an adipose-rich parenchyma embedded with a branching network of epithelial ducts, which are organized in 2 distinct cell layers, the basal myoepithelial and luminal epithelial layer. Bilayered myoepithelial-luminal epithelial organization is a critical aspect of not only normal mammary gland function but also of the secretory acini of the prostate. The myoepithelial cells of the breast and basal cells of the prostate share the properties of expressing basal cell–specific cytokeratins 5 and 14, p63, hemidesmosome structures, and function to connect cells to the underlying basal lamina and surrounding stroma.1-3 They differ in that myoepithelial cells function as contractile cells, whereas the basal cells of the prostate are not contractile, with this function being carried out by the smooth muscle cells of the prostate interstitium. 4 Epithelial bilayers are established early during development and persist throughout adulthood, 2 but importantly, the basal/myoepithelium layer is lost in adenocarcinoma of the breast and prostate.
There is significant evidence that the myoepithelium is a natural tumor suppressor.5-8 Myoepithelial cells (MECs) induce growth arrest and apoptosis in breast cancer cell lines
9
and secrete ECM proteins, antiangiogenic factors, and protease inhibitors.10,11 When co-cultured with breast cancer cells, MECs inhibit expression of MMPs
10
and direct polarization and branching morphogenesis during mammary gland development.12,13 Changes to the myoepithelial cell layer occur during the transition from ductal carcinoma
CD44 is a type I transmembrane adhesion receptor involved in mediating the response of cells to their extracellular microenvironment. Research of CD44 in breast and prostate cancer has shown that it both promotes metastatic signaling and acts as a putative tumor suppressor.15-19
To better understand the role of CD44 in cancer, we began by examining its function during normal mammary gland development, as processes that occur in development mirror cancer progression and rely on conserved signaling pathways, making it a powerful model system to understand cancer progression. Here, we report that CD44 is expressed early during pubertal mammary gland development and that it regulates ductal outgrowth and terminal end bud morphology. We found that CD44 is expressed in the myoepithelial cell layer of the developing duct, and its loss impairs myoepithelial-to-luminal epithelial cell-cell adhesion and bilayer organization. This observation prompted us to assess the relevance of myoepithelial- specific CD44 expression in the MMTV-PyV MT transgenic mouse and in human breast and prostate cancer. We found that CD44 is expressed in the myoepithelium/basal layer in prostatic intraepithelial neoplasia (PIN) and in DCIS but is re-expressed strongly in the epithelium of invasive carcinoma. Together, our data demonstrate a role for CD44 in the developing mammary gland, highlighting the changes in cell type–specific expression of CD44 during cancer progression.
Results
CD44 is expressed during pubertal mammary gland development and localizes to the myoepithelial cell layer
Development in the mouse mammary gland occurs from approximately 3 to 8 weeks of age. During this period, epithelial tissue initiating in the mammary enlage undergoes extensive growth and branching to fill the mammary fat pad and form an extensive network of ductal epithelium. To verify CD44 expression during development and phases of active ductal outgrowth, protein lysates were prepared from virgin mammary glands at 3, 4, and 6 weeks of age, and CD44 protein expression was examined by Western blot analysis using a pan-CD44 antibody (IM7), which binds to the nonvariant portion of murine CD44 (Fig. 1A). CD44 standard isoform (80 kDa) and variant isoforms (100 and 250 kDa) are strongly expressed at 4 weeks and persist through 6 weeks.
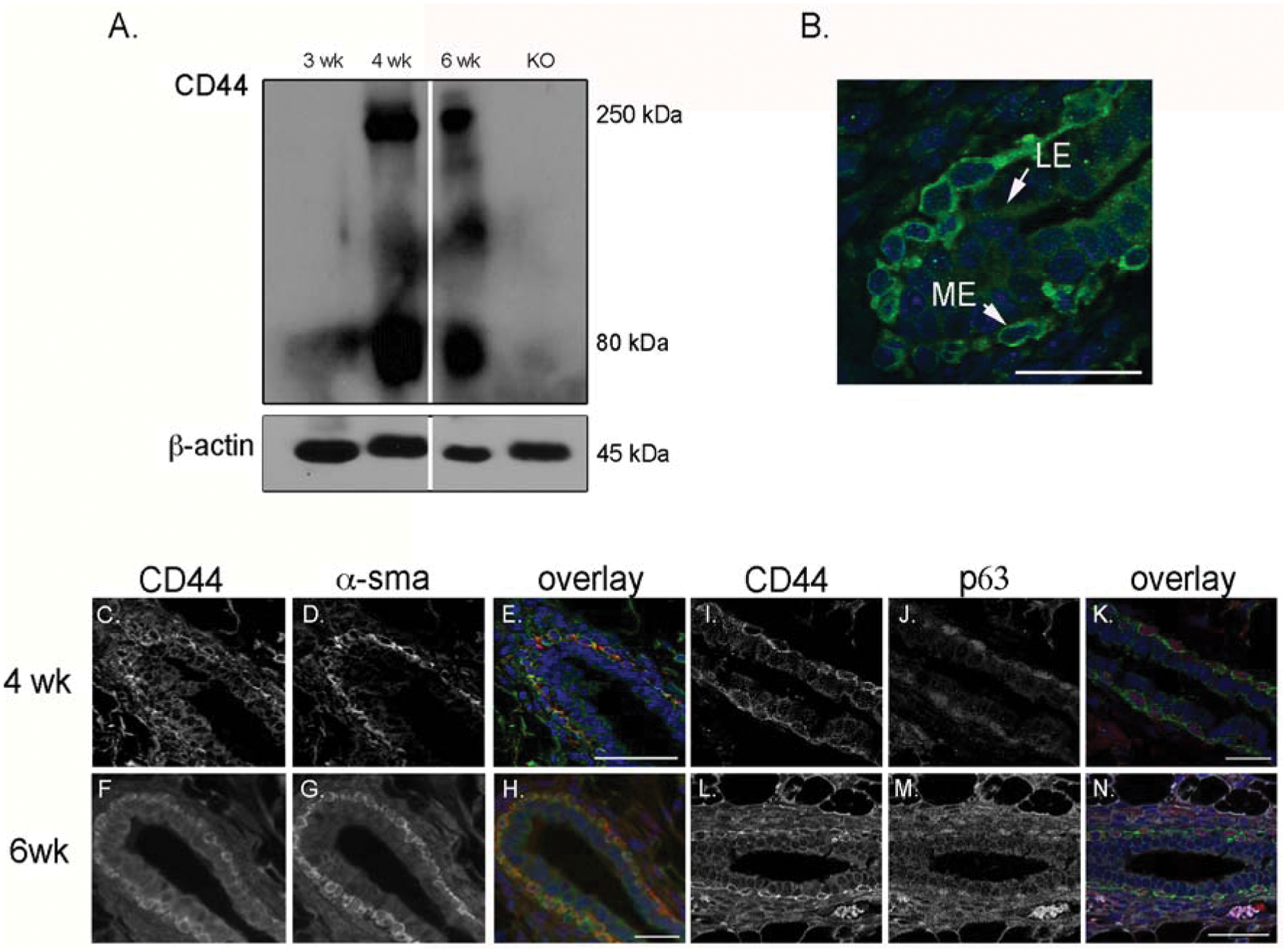
CD44 is expressed in the myoepithelial cell layer during mammary gland development. (
The mammary epithelial duct consists of distinct bilayered cellular organization, containing a basal myoepithelial cell layer and an inner luminal epithelial layer. To examine the localization of CD44, 6-week-old mammary glands were visualized by immunofluorescence. Notably, CD44 expression was detected predominantly in the myoepithelial layer of growing ducts with limited expression in the luminal epithelium (Fig. 1B). To verify myoepithelial- specific expression, we examined CD44 in conjunction with 2 myoepithelial markers, α-smooth muscle actin (αsma) and p63, in 4- (Fig. 1C-E and 1I-K) and 6-week-old (Fig. 1F-H and 1L-N) mammary glands and found that they are co-expressed (Fig. 1E, 1H, 1K, and 1N). Thus, CD44 is expressed in the myoepithelium of the mammary gland during periods of growth and branching.
Loss of CD44 delays ductal outgrowth
We have found that CD44 is expressed during pubertal development and next examined the role of CD44 in this process using a CD44-null mouse. CD44–/– mice are healthy and viable, and although genetic deletion of CD44 in DAB/1 mice has been reported to promote premature involution of the lactating mammary gland,
28
mice on the FVB/N background lactate normally. During the course of our study, we monitored the litter size, frequency of birth, and weight at weaning of CD44-null mice and found no impairments. To assess whether CD44 plays a role in ductal outgrowth, mammary glands were collected from CD44–/– and CD44+/+ mice at 4 and 6 weeks, and the ductal tree was visualized by carmine alum staining of whole-mounted glands (Fig. 2A-D). Absence of CD44 protein in CD44–/– mice was confirmed by Western blot analysis (Fig. 1A, lane 4). Duct length was measured in the fourth inguinal mammary gland, and we observed that at 4 weeks, ductal elongation is impaired in CD44–/– mice, and ducts were, on average, 23% shorter than CD44+/+ mice (
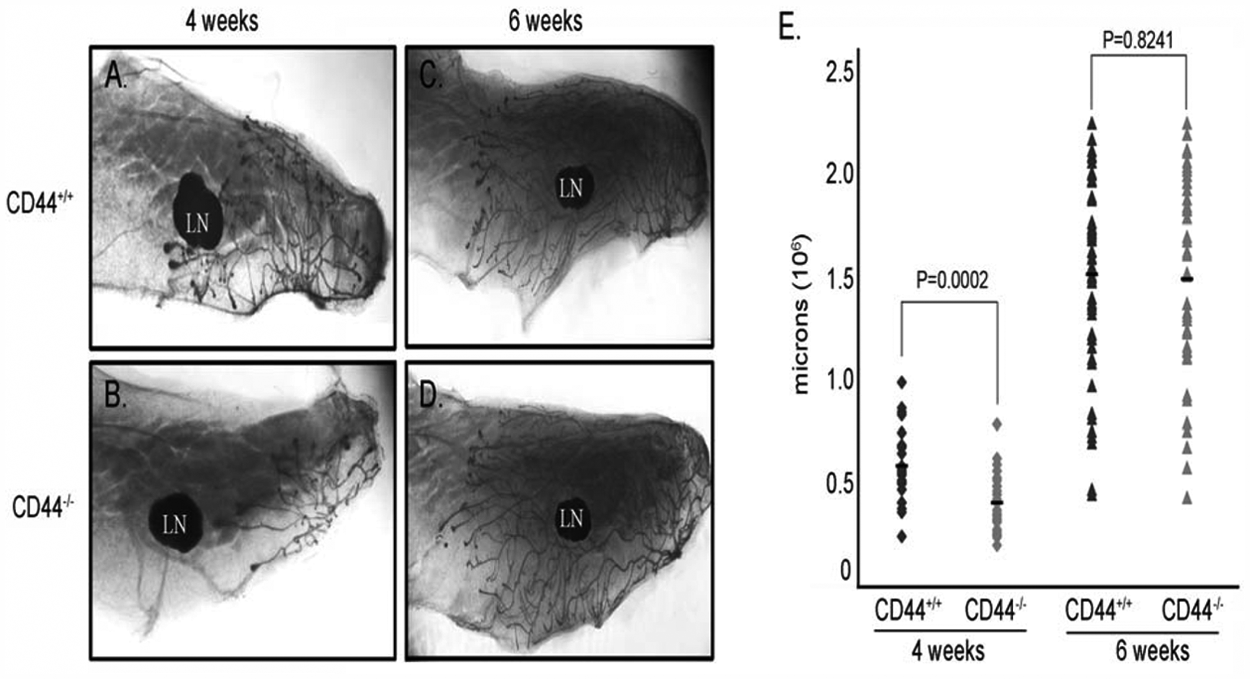
Loss of CD44 delays ductal outgrowth. (
Impaired ductal outgrowth is due to terminal end bud malformation
The terminal end bud (TEB) of the developing mammary gland is a specialized structure that mediates growth and invasion into the mammary fat pad during development. To assess the mechanism of delayed ductal outgrowth in CD44–/– mice, we examined the morphology and size of TEBs in CD44–/– and FVB/N mice (Fig. 3). TEBs were analyzed in whole-mounted glands in 4- and 6-week-old mice by examining TEBs along the leading edge of ducts in the fourth inguinal mammary gland (Fig. 3A-H). Examination of CD44–/– mice revealed TEBs that were significantly smaller than their wild-type counterpart (Fig. 3C, 3D, 3G, and 3H). The size of TEBs was measured, and we found CD44-null mice had significantly smaller TEBs at both 4 and 6 weeks (Fig. 3M). To assess TEB abnormalities on a cellular level, we examined TEBs in H&E-stained glands and examined E-cadherin and αsma expression by immunohistochemistry and found that, although the TEBs of CD44–/– are morphologically smaller (Fig. 3J and 3L v. 3I and 3K), no differences in E-cadherin or αsma were observed, suggesting normal cellular architecture of the TEB (data not shown). In addition, we examined the number of TEBs in wild-type and null mice and determined that there were no differences in TEB number; at 4 weeks, FVB/N mice have an average of 9.2 TEBs per gland, and CD44–/– mice have an average of 9 TEBs per gland (
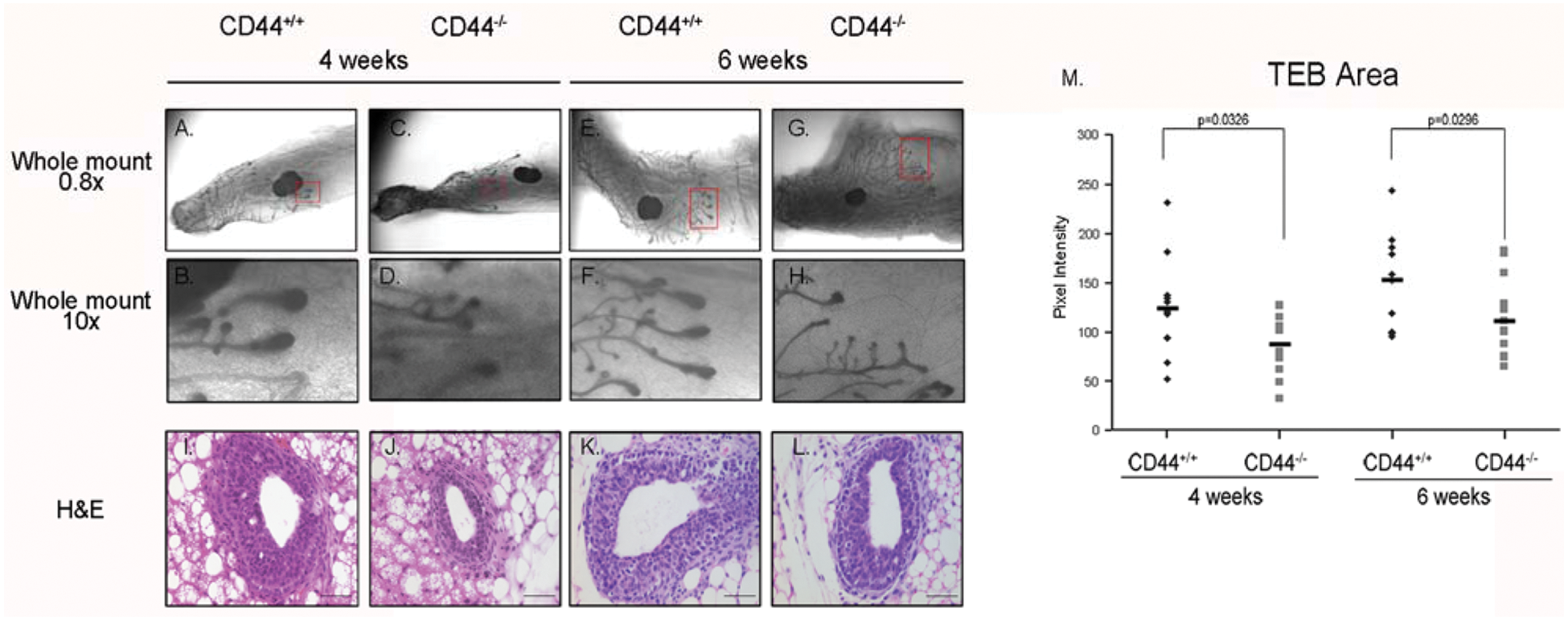
CD44–/– mice have reduced terminal end bud (TEB) area. (
Loss of CD44 impairs bilayer cell organization
The myoepithelium directs polarization of the luminal cells and thus maintains bilayered epithelial cell organization of the mammary duct. 12 Given the myoepithelial-specific expression of CD44, we next determined the effect of CD44 deletion on structural organization of the mammary gland. We first examined the architecture of the epithelial bilayer by examining ducts for the luminal marker, cytokeratin 18, and the myoepithelial marker, αsma, in 4-week-old mammary glands by immunofluorescence (Fig. 4). Wild-type mice display distinct luminal and myoepithelial cell layers, with no mixing of either cell type (Fig. 4A-D). In contrast, the bilayer organization is disrupted in CD44–/– ducts. Although 2 distinct cell layers are present, αsma-positive cells can be seen in the luminal cell layer of CD44–/– ducts but not wild-type ducts (Fig. 4E-H). Examination of mislocalized αsma-positive cells in the luminal compartment reveals that these cells do not express luminal cytokeratins (Fig. 4G and 4H; arrowheads denote myoepithelial cells, and arrows denote luminal cells).
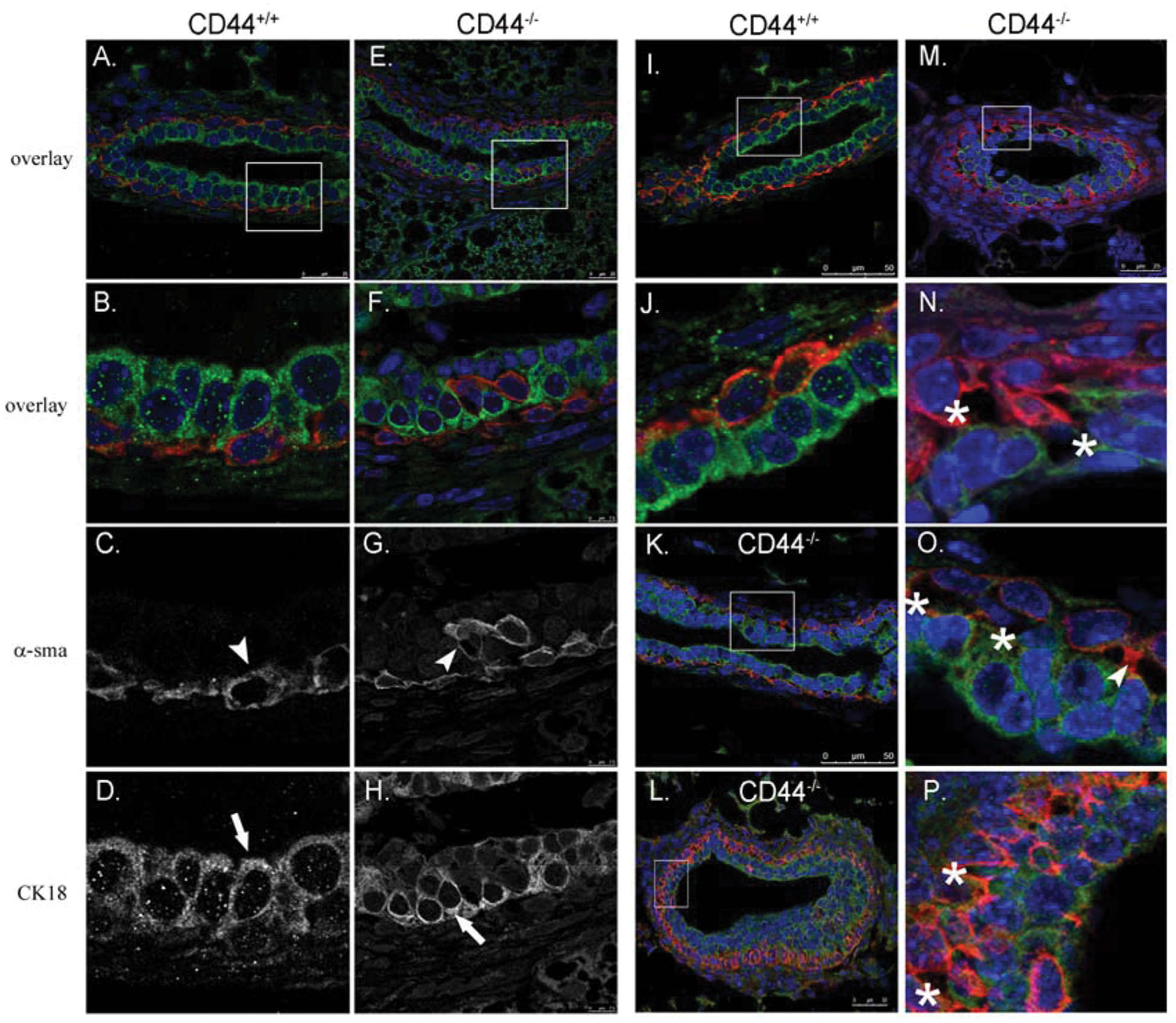
Loss of CD44 causes disruption to bilayered cellular organization and loss of luminal-myoepithelial adhesion. Formalin-fixed mammary glands from 4-week-old CD44+/+ (
Loss of CD44 impairs luminal-myoepithelial cell-cell adhesion
Examination of the bilayer also revealed that the loss of CD44 impairs luminal-myoepithelial adhesion. Normally, MECs are juxtaposed between the luminal cells and the basement membrane, and this adhesion is required for normal epithelial morphogenesis. 13 Mammary ducts of FVB mice display tight luminal- to-myoepithelial adhesion (Fig. 4I and 4J). The loss of CD44, however, dramatically impaired adhesion (Fig. 4K-P). Large spaces are visible between the 2 cell layers, and occasional punctate projections of αsma can be observed between the 2 layers (Fig. 4N-P, asterisks and arrowhead in O).
Myoepithelial CD44 expression correlates with loss of the myoepithelium in MMTV-PyV MT mammary tumors
We have previously demonstrated that the loss of CD44 in the MMTV-PyV MT mouse leads to a 6-fold increase in metastasis. 17 The myoepithelial-specific expression of CD44 during mammary gland development suggests that its cell type–specific expression may regulate antitumorigenic properties given the tumor-suppressive nature of the myoepithelium. We sought to determine if CD44 maintains myoepithelial expression during early tumorigenesis and if CD44 expression changes as the myoepithelial cell layer is lost with increasing invasiveness.
The MMTV-PyV MT mouse model is a useful model for human breast cancer because tumors progress through distinct stages of tumorigenesis in a similar fashion to human cancer. 29 We therefore examined CD44 localization in conjunction with a myoepithelial marker in mammary tumors from MMTV-PyV MT mice. Mammary glands from healthy adult virgin mice, hyperplastic mammary glands, and mammary tumors from MMTV-PyV MT were analyzed for CD44 using the pan-CD44 antibody (IM7), which recognizes all CD44 isoforms and αsma co-localization (Fig. 5). Cells that are positive for CD44 expression may express multiple isoforms, while cells that are negative for CD44 expression do not express any isoforms. In the adult mammary gland, unlike the developing mammary gland, CD44 is predominantly expressed in the luminal epithelium, with limited expression in the myoepithelium (Fig. 5A, 5E, 5I, and 5M). Contrastingly, in hyperplastic mammary glands in which the ductal architecture is intact and a myoepithelial layer surrounds the gland, CD44 strongly localizes to the myoepithelium (Fig. 5B, 5F, 5J, and 5N). In DCIS, where tumor epithelium has filled the lumen of the duct but has not invaded into the surrounding stroma, basal cells form a semicontinuous barrier around the duct but are disperse (Fig. 5C, 5G arrows, 5K, and 5O). In these tumors, CD44 expression correlates with αsma expression (Fig. 5G, 5K, and 5O) and appears to disperse in conjunction with the myoepithelial cell layer. In invasive ductal carcinoma, however, in which there is no αsma expression (Fig. 5L), CD44 is strongly expressed throughout the tumor epithelium (Fig. 5P).
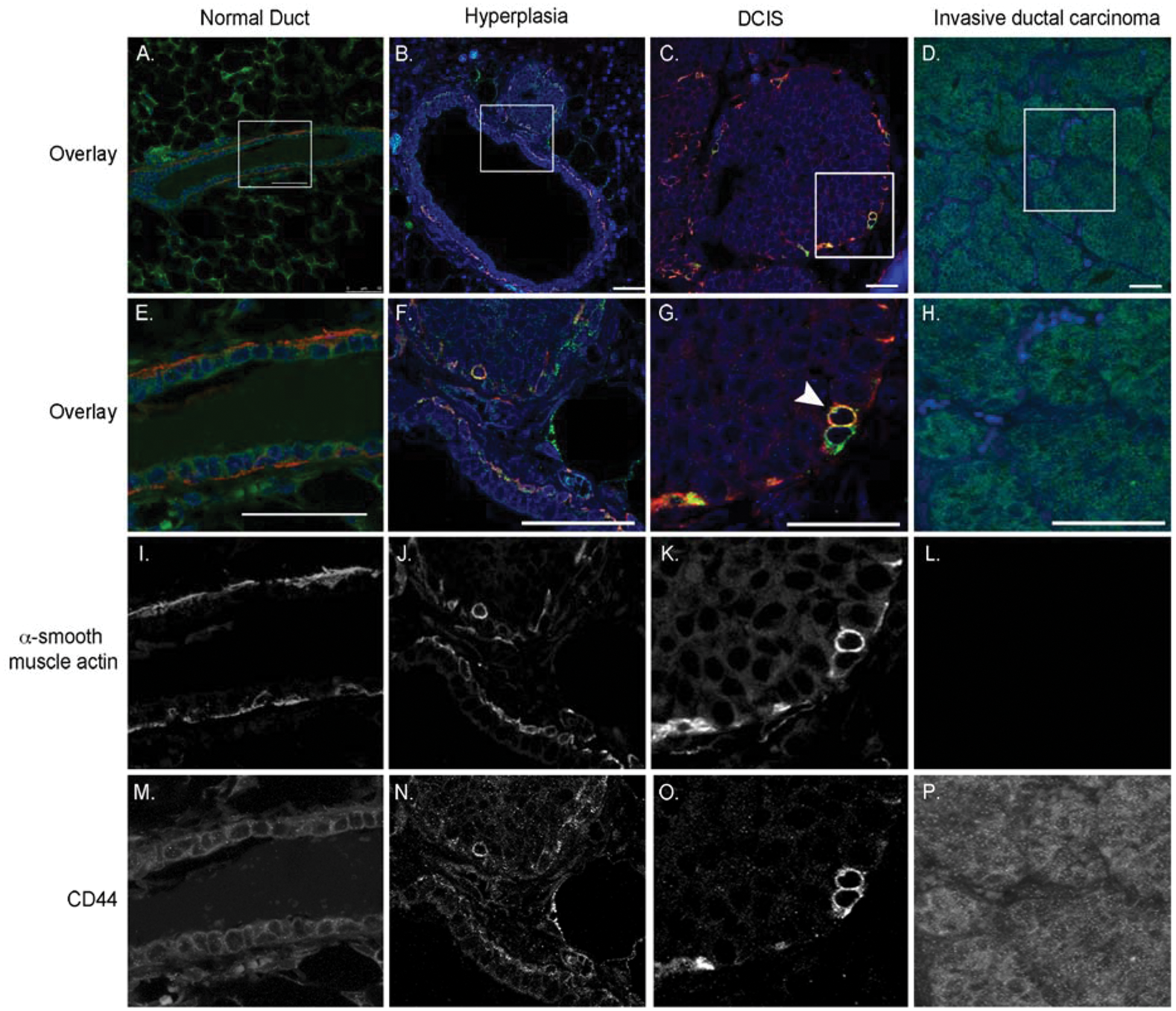
CD44 and αsma localization changes during tumor progression in MMTV-PyV MT mammary tumors. Formalin-fixed sections from normal (
CD44 basal-to-epithelial expression in human breast cancer
CD44 expression has not been correlated with myoepithelial expression in an early stage in cancer progression, DCIS, when the basal cell layer is intact. We therefore examined the expression of CD44 and αsma in 10 cases of DCIS. Immunofluorescence staining of DCIS with a pan-CD44 antibody showed that CD44 is expressed primarily in 2 foci of DCIS, the myoepithelium surrounding a tumor, which correlates with expression of αsma in 60% of DCIS cases examined (Fig. 6B, 6E, and 6K), and in the tumor epithelium, usually in the center of DCIS (Fig. 6B, 6E, and 6K; arrowhead denotes MEC, and arrow denotes CD44-positive tumor epithelium). As in the MMTV-PyV MT mouse, CD44 was found strongly expressed in a foci of microinvasion surrounding DCIS in one of the cases examined and is reportedly highly expressed in invasive ductal carcinoma, particularly basal-like subtypes 30 (Fig. 6F and 6L, arrow). Interestingly, while others have observed CD44 in the myoepithelium of the normal breast ducts, 23 we examined 10 cases of normal breast tissue from mammoplasties and found that, as in mice, CD44 is expressed in the luminal epithelium of adult ducts, with occasional expression in the myoepithelium or both (Fig. 6A, 6D, 6G, and 6J). Additional examples of CD44 localization in DCIS are provided in Supplementary Figure S2.
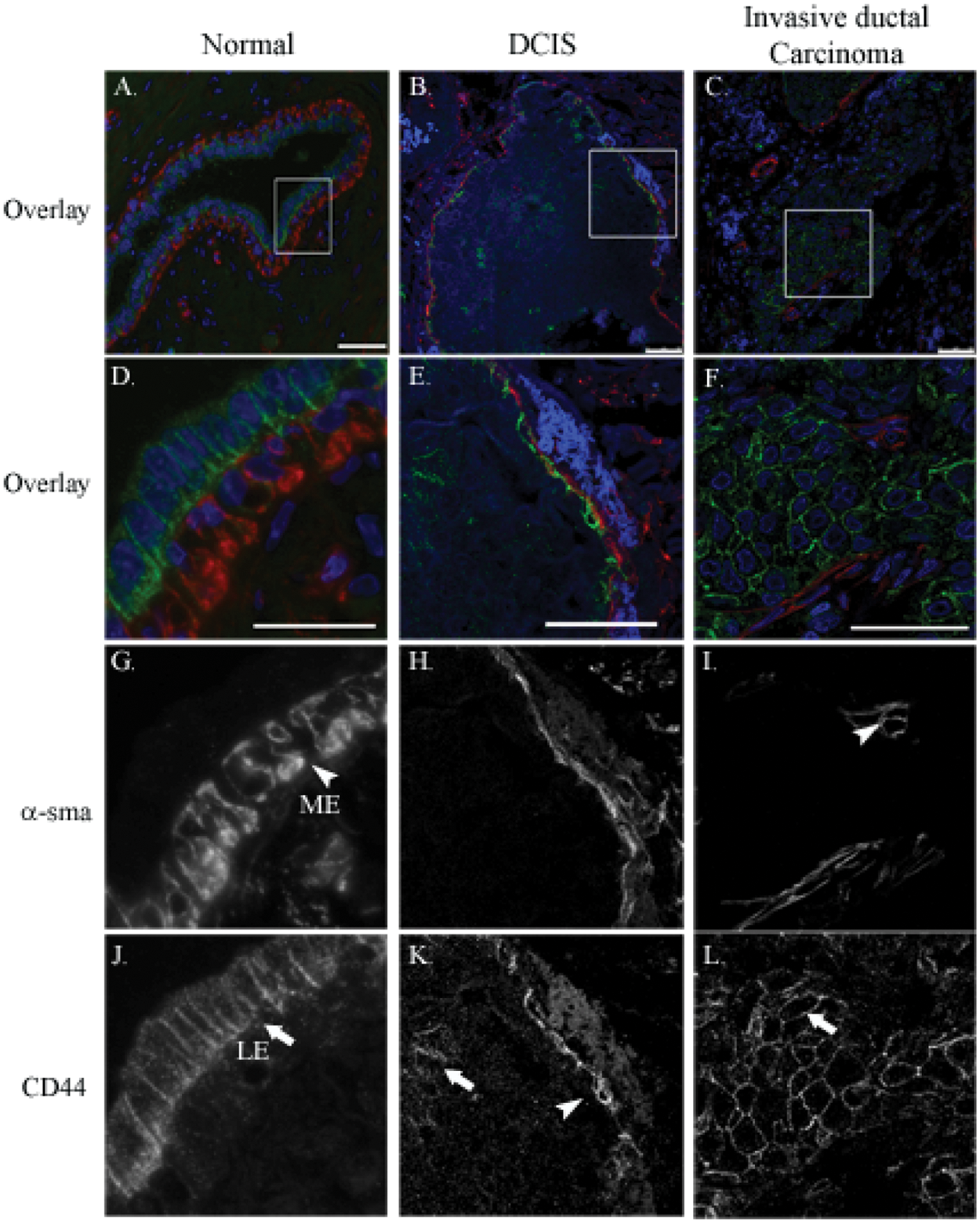
CD44 and αsma localization changes during tumor progression in human breast cancer. Formalin-fixed sections from normal (
In addition, we investigated the co-localization of CD44 with an additional myoepithelial marker, p63. We found that CD44 is co-expressed in p63-positive cells in hyperplastic mammary glands near foci of DCIS and along the edges of DCIS where the basal cell layer remains intact (Suppl. Fig. S2J, S2K, S2M, and S2N). Interestingly, in some cases of DCIS, p63-positive cells form acinar structures in the center of tumors that are positive for p63, and we found that these cells are CD44 negative (Suppl. Fig. S2L and S2O).
CD44 undergoes a basal-to-epithelial switch in human prostate cancer
To address the prevalence of myoepithelial-CD44 expression in other human cancers with bilayer ducts, we examined the localization of CD44 in conjunction with a basal cell marker, cytokeratin 5/14, in human prostate tissue. Prostate cancer progression is identifiable by the presence or absence of a basal cell layer and is, therefore, another model for the role of basal cells during tumor progression. We examined the localization of CD44 and cytokeratin 5/14 in human prostate and found that, in contrast to the breast, CD44 is restricted to the basal cell layer in normal ducts (Fig. 7A, 7D, 7G, and 7J, arrowheads) but, similar to the breast, is acquired by luminal epithelial cells in prostatic intraepithelial neoplasia (PIN) lesions (Figs. 7B, 7E, and 7K, arrows). As in breast cancer, CD44 is expressed throughout the tumor epithelium of invasive cancer (Fig. 7C, 7F, and 7L).
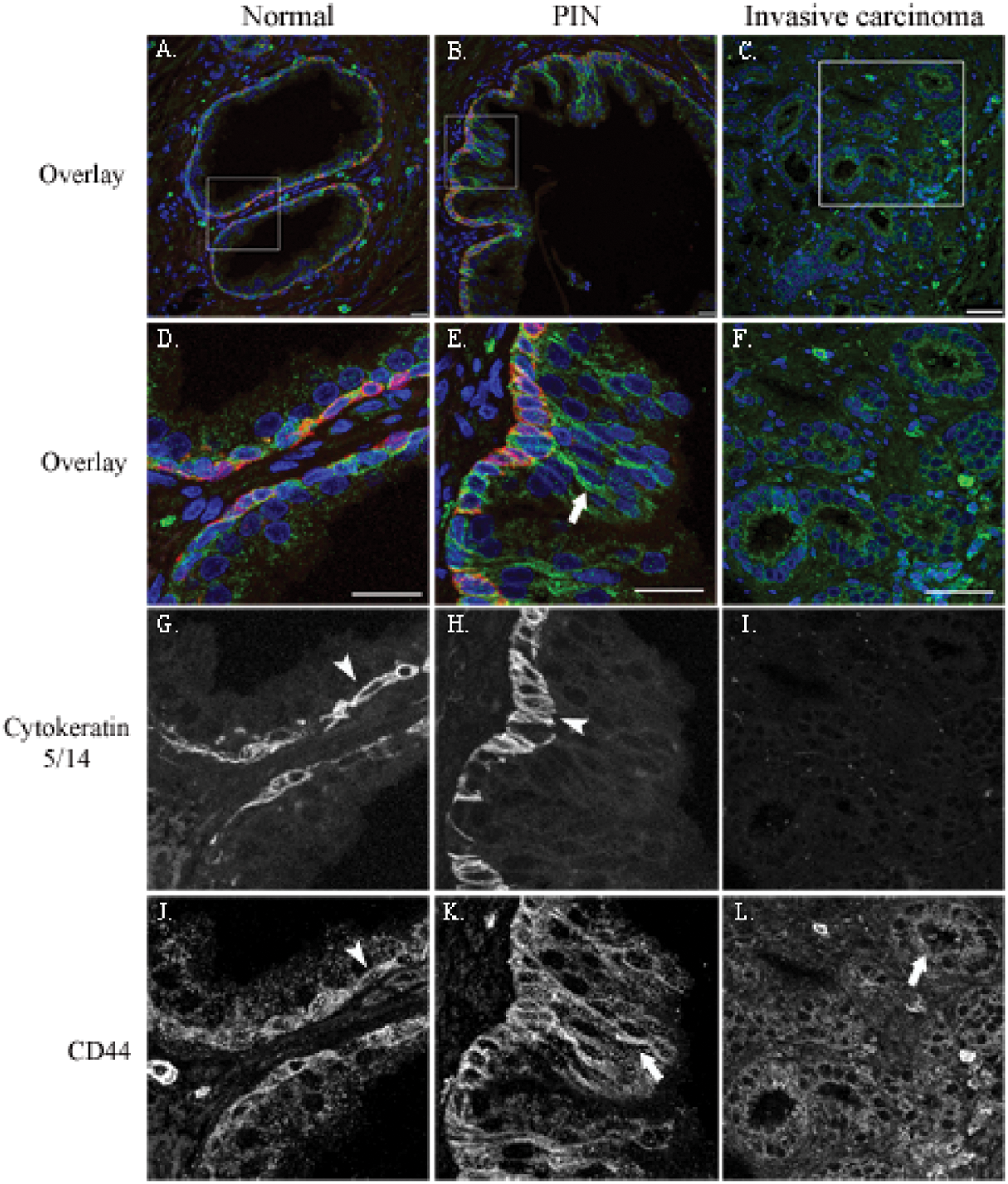
CD44 and cytokeratin 5/14 localization changes during tumor progression in human prostate cancer. Formalin-fixed sections from normal (
These data demonstrate that CD44 expression is subject to changes in cell-type expression during cancer progression in several models.
Discussion
CD44 has garnered significant attention in cancer biology for several decades due to its expression in numerous cancer types, its putative role in prometastatic signaling, and its more recent identification as a stem cell marker. Its expression has been correlated with both a favorable and unfavorable prognosis, and it has been found to act in both tumor suppression and metastasis promotion with no clear consensus about its role in cancer progression.
In the present study, we have shown that CD44 is expressed early during normal pubertal mammary gland development, predominately in the myoepithelium. This expression is functionally important, as CD44-null mice have a delayed ductal outgrowth and morphologically small TEBs. Epithelial-stromal interactions are critical to normal development, and we have found that this phenotype correlates with impaired myoepithelial-luminal identity, cell-cell adhesion, and bilayer organization.
Our findings suggest that the cell-type expression of CD44 also changes during cancer progression, and we therefore examined CD44 localization in conjunction with a myoepithelial marker during cancer progression in the MMTV-PyV MT transgenic mouse, in which the myoepithelial cell layer is lost during DCIS-invasive transition. In this model, CD44 is expressed in MECs in hyperplasia and DCIS, but as the myoepithelium is lost, CD44 is found expressed in the tumor epithelium. We have previously shown that in the MMTV-PyV MT model of breast cancer, CD44 is protective against lung metastasis when lost throughout tumor progression but is strongly expressed throughout the tumor epithelium of invasive tumors. 17 This is in contrast to the loss of CD44 in a mutant p53 model of osteosarcoma, where CD44 loss prevented lung and liver metastasis. 31 These data suggest that myoepithelial-specific expression may regulate its tumor-suppressive function, while expression in epithelium of carcinoma may account for its tumor-promoting function.
To investigate the relevance of CD44 myoepithelial function in human cancer, we examined its localization in breast and prostate cancer. In early neoplasias, CD44 is expressed in the basal/myoepithelial layer with foci of expression in luminal epithelium. As the basal cell layer is lost, however, CD44 remains strongly expressed by tumor epithelium. Interestingly, CD44 is expressed by basal cells of normal prostate but by luminal cells in normal adult mammary epithelium. This may represent differences in cell-type regulation of CD44 expression in mammary and prostate tissue and a luminal-myoepithelial switch in the transition from normal to DCIS. The basal-luminal expression of CD44 in PIN lesions, on the other hand, may represent transitory cells or cells that express markers of both cell types and points to these cells as a key regulator of the normal-PIN transition. 32 Alternatively, CD44-positive cells in invasive tumors may represent CD44-positive progenitor cells from the basal cell layer, suggestive of a tumor of basal cell origin. Cell type–specific deletion studies would lend insight into the contribution of CD44, in addition to contributing to our knowledge about the cell type responsible for transformation.
Our data suggest that CD44 expression in DCIS and PIN resemble developmental CD44 expression patterns while CD44 expression in adenocarcinoma of the breast resembles luminal adult expression patterns. These findings demonstrate that normal myoepithelial function is regulated in part by CD44. This may indicate that CD44 plays a protective role against cancer progression, as the myoepithelium is protective. Alternatively, these data also suggest that tumor progression may not occur in a linear fashion, with DCIS as a precursor of luminal subtypes of breast cancer. Breast cancer has traditionally been thought to arise from the luminal epithelial cell layer, but profiling of breast cancers has demonstrated distinct phenotypic subtypes that differ in their biological characteristics as well as clinical outcome.33-36 The basal subtypes of cancer, in particular, which encompass the most aggressive subtypes and which are most strongly associated with reduced survival, have highlighted the need to understand the molecular profile and the contribution of distinct cell types.37,38 Our data suggest that tumors are subject to molecular regulation of distinct cell types and that cell type–specific contribution to cancer progression could contribute to differences observed in tumor subtype and origin.
Interestingly, other groups have reported that CD44 is expressed in the myoepithelium of normal mammary tissue, 23 which may reflect differences in the reproductive stage of the patients or CD44 isoform changes or stem from the use of different antibodies. CD44 isoform expression represents an additional layer of CD44 regulation; it is known to commonly express at least 10 splice variants with different expression patterns that have varying functions. Isoform expression has been proposed as a mechanism through which CD44 promotes tumorigenesis and metastatic progression. Histopathological examination of CD44 variant expression, however, has produced conflicting results with no clear correlations between variant expression and tumorigenicity. While several groups have reported changes to CD44 variant expression during tumor progression, particularly in breast cancer, there is no consensus about which variants are associated with increased aggressiveness. To exclude the complexity of variant expression, we examined cell-type changes to CD44 expression, which recognizes both standard and variant isoforms; future research should examine variant-specific changes in this context.
Taken together, our data indicate that the cell type– specific expression of CD44 is a key aspect of its biological function. CD44 plays a role in maintaining normal myoepithelial function during development and undergoes a cell-type switch during cancer progression. These data lend insight into the conflicting results regarding the role of CD44 as a tumor suppressor or promoter and highlight the importance of studying the cell type–specific function of proteins important to cancer progression. Future research will investigate the transcriptional regulatory mechanisms of CD44 in basal and luminal cell types to understand what mechanisms control this expression switch.
Materials and Methods
Mice
CD44-null mice were a kind gift from Tak Mak (University of Toronto) and maintained on a FVB/N background. MMTV-PyV MT mice were a gift from William Muller (McGill University). F1 generation–outbred mice (C57Bl/6 X FVB/N) were analyzed for tumor and metastasis formation. Twelve FVB mice from 5 litters and 19 CD44–/– mice from 6 litters were analyzed for mammary gland development. Mice weighing less than 15 g or more than 19 g were determined to be outliers, and so only mice with body weight between 15 and 19 g were used.
Whole mounts
The inguinal mammary glands were collected and fixed in a 1:3 solution of glacial acetic acid/ethanol, subsequently washed in a series of ethanol washes, and defatted in acetone. The glands were rehydrated in a series of ethanol washes, stained in 0.2% carmine/0.5% aluminum potassium sulfate solution, and destained in ethanol. Images of the whole mounts were captured using a Leica MZFLIII dissection scope (Wetzlar, Germany) with Optronics MagnaFire (Goleta, CA).
Immunoblotting
Protein lysates of mammary glands were prepared by harvesting and immediately homogenizing in Triton X-100 lysis buffer (20 mm HEPES, pH 8.0, 150 mm NaCl, 1% Triton X-100, 2 mm EDTA, 2 mm sodium orthovanadate, 50 µm ammonium molybdate, 10 mm sodium fluoride, and Complete inhibitor mixture [Sigma, St. Louis, MO]), followed by the removal of the insoluble fraction by centrifugation, and stored at –80°C until use. The protein concentrations were determined by bicinchoninic acid assay (Pierce, Rockford, IL). Equal protein concentrations were loaded onto SDS-PAGE gels, separated by electrophoresis, and transferred onto PVDF membrane (Millipore, Billerica, MA). The membrane was blocked with either 2.5% nonfat milk in PBS (0.1% Tween20) or 3% BSA in TBS (0.1% Tween20) and immunoblotted. The membrane was then treated with Super Signal West Pico Chemiluminescent Substrate (Pierce), visualized on Image- tech-B film (American X-ray Supply Inc., Oilville, VA), and developed with Konica SRX-101C (Tokyo, Japan). Antibodies used were CD44 (IM7) from Santa Cruz Biotechnology (Santa Cruz, CA) and β-actin (AC-75) from Sigma.
TEB area measurements
Whole mounts were analyzed with Image Pro Plus 5.1Blinded images were artificially colored in Adobe Photoshop CS2 (San Jose, CA), and TEBs along the leading edge of growth were analyzed for area by pixel intensity, and results were analyzed by a Student
Branch length measurements
Measurements were made using Image Pro Plus 5.1 (MediaCybernetics) by calibrating images taken at 0.8x magnification against a photographed ruler. From blinded files, 4 branches growing in the direction of the lymph node were chosen at random and measured in Image Pro Plus 5.1 by tracing the duct from the mammary anlage to the terminal bud against a ruler calibrated in Image Pro Plus. Data were analyzed by Student
Immunofluorescence/immunohistochemistry
Tissues were dissected and fixed in 10% buffered formalin or 4% paraformaldehyde, embedded in paraffin, and sectioned using the Tissue Acquisition and Cellular/Molecular Analysis Shared Service (TACMASS) of the Arizona Cancer Center and immunofluorescence performed as described previously. 39 Antigen retrieval was performed in 1 mM EDTA at 121°C by pressure cooker. Antibodies used were CD44 (H300) and cytokeratin 18 (H80) from Santa Cruz Biotechnology, αsma (1A) from Sigma, cytokeratin 5/14 (KA2) and HABP from United States Biological (Swampscott, MA), and p63 (4A4) from Abcam (Cambridge, MA).
Human breast and prostate tissue
Human tissues were harvested and fixed in 10% neutral buffered formalin for 24 hours at the University Medical Center (Tucson, AZ). Tissue processing and sectioning were done using TACMASS. Routine H&E stains were performed on 3-µm sections of tissue. All tissues were then de-identified and provided by TACMASS. Immunofluorescence/immunohistochemistry was performed as described above.
Footnotes
Acknowledgements
Breast and prostate tissue and histological services were provided by the Tissue Acquisition and Cellular and Molecular Analysis Shared Service, which is supported by a Arizona Cancer Center grant (NIH CA023074). The authors are grateful to Kathy McDaniel for excellent technical assistance and to Ben Bitler and Matthew Callan for their insightful edits of this article.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
This work was supported by the Arizona Cancer Center (to JAS and RBN) [grant number 2P30CA023074]; the Arizona Biomedical Research Commission (to JAS) [grant number 0807]; and the Department of Defense Predoctoral Breast Cancer Research Program (to JMVL) [grant number W81XWH0810247]. The funding sources had no involvement in the design, collection, analysis, or interpretation of the data presented.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
