Abstract
Background
RIPK4 is essential for the identification of keratinocyte differentiation and cutaneous inflammation. The mechanisms involved in the tumorigenesis of tongue squamous cell carcinoma (TSCC) have not been well characterized yet.
Objective
To assess RIPK4 expression in various differentiated TSCC and to determine its basic biological function.
Methods
Overall, 109 human samples (36 normal tongue tissues and 73 surgically excised TSCC tissues with different degrees of differentiation) were collected for the present study. The expression of RIPK4 was assessed by real time PCR and immunocytochemistry. Multivariate logistic regression analysis was used to investigate the association between RIPK4 and clinical characteristics. In vitro analyses by scratch wound healing assay and invasion assays, as well as confocal laser scanning microscopy and flow cytometry analysis were performed using the Tca-8113 tongue cancer cell line. Cisplatin sensitivity analysis and cisplatin-induced activation of caspase 8 analysis were also performed.
Results
A significantly higher level of RIPK4 was found in well-differentiated TSCC samples than in the poorly differentiated ones (p<0.01). Male patients had relatively higher levels of RIPK4 (p=0.03). RIPK4 levels were also increased in older patients. In vitro, RIPK4 reduced the migration and invasion of the Tca-8113 cell line, but did not affect apoptosis and cell cycle. Both RIPK4 up or downregulation induced cell sensitivity to cisplatin, determining cell apoptosis.
Conclusions
In the present work we found a correlation between RIPK4 expression and TSCC degree of differentiation, age, and gender. The better understanding of the molecular mechanism of RIPK4 in TSCC may provide a promising biomarker for tongue cancer prognosis and treatment.
Keywords
Introduction
Oral squamous cell carcinoma (OSCC) is the sixth most common cancer worldwide (1). As one of the most common malignancies in China, the incidence of OSCC is increasingly rising (2, 3). Among OSCCs occurring in tongue, bucca, gingiva, and palate, tongue carcinoma has the poorer 5-year survival rates (50%) and the lowest rate of local tumor control (4). However, the mechanisms involved in the tumorigenesis of tongue squamous cell carcinoma (TSCC) have not been well characterized yet.
The RIPK4 (receptor-interacting serine/threonine kinase protein 4) was first described as an unidentified protein found to interact with protein kinase C (PKC) β1 and PKCδ in yeast two-hybrid assays (5, 6). RIPK4 contains an N-terminal RIP-like kinase domain linked to a unique intermediate domain that can be cleaved by caspases (7). Overexpression of RIPK4 in HEK 293T cells activates the nuclear factor-kB (NF-kB), c-Jun N-terminal kinase (JNK), and activator protein-1 (7–9). Due to the abnormal epidermal differentiation, RIPK4 deficiency in mice results in perinatal lethality. The epidermis of RIP4-/- mice is thickened with expanded spinous and granular layers, and contains an outermost layer of parakeratotic cells instead of enucleated squamous cells. RIPK4 autonomously functions in a cell within the keratinocyte lineage (10). RIPK4 is an ankyrin repeat-containing kinase essential for keratinocyte differentiation (10) and cutaneous inflammation (11–13).
The effects of RIPK4 in human cancer differentiation are not known. The goal of the present study was to determine the role of RIPK4 in the various grades of differentiation of TSCC.
Materials and Methods
Patients and Controls
For this study we obtained 36 samples of normal tongue from paracancerous normal tissue and 73 samples of surgically excised TSCC from patients who were treated at the First Affiliated Hospital of Zhejiang University. Of these, 32 patients had highly differentiated tumors (according to WHO), 23 had moderately differentiated tumors, and 18 had poorly differentiated tumors. Among TSCC patients, 20 had lymph node metastasis. None of the patients underwent radiotherapy, chemotherapy, or any other interventional palliative or therapeutic measure prior to sampling. Representative blocks, corresponding to the area of greatest tumor diameter, were selected and analyzed. All specimens were obtained from the central region of the tumor and pulverized in liquid nitrogen in a cold mortar, or fixed in 10% formalin and embedded in paraffin. For all patients OSCC diagnosis was confirmed by histology; tumor samples were checked to ensure that the tumor tissue was present in more than 90% of the specimen. Clinical characteristics of the patients were recorded, including age, gender, secondary education, alcohol consumption, smoking habit, diabetes, hypertension, and lymph node metastasis.
Immunocytochemistry and RIPK4 quantification
Paraffin-embedded sections (5 μm) were immunohistochemically stained with rabbit-anti-RIPK4 monoclonal antibodies (Abcam, Cambridge, UK) using the streptavidinperoxidase technique. Instead, sections used as negative controls were incubated with phosphate buffered solution (PBS). IHC results were reported using the ASCO/CAP modified Hercepscore scoring scheme for the membrane marker RIPK4 (14) as follows: 0 (negative), 1+ (incomplete, weak membrane staining in >10% of tumor cells), 2+ (intermediate, complete membrane staining in >10% of tumor cells), 3+ (strong, uniform, and complete membrane staining in >30% of tumor cells). Each sample was measured in triplicates.
Cell culture
The human TSCC cell line Tca-8113 was obtained from the Chinese Center for Type Culture Collection, in China. Tca-8113 cells were maintained in DMEM (GIBCO, CA), containing 10% FBS (Hyclone, CA), 100 IU/mL ampicillin (Hyclone, CA), and 100 μg/mL streptomycin (Hyclone, CA) and incubated at 37°C in 5% CO2. Cells were seeded at a density of 4×105 cells per well and 18 hours later the cells were transfected with 4 μg of the pRIPK4 plasmid (donated by Prof. Mathieu JM Bertrand, VIB/Ghent University, Belgium) or the pRIPK4-shRNA plasmid (Genepharma, shanghai, China) using Lipofectin (Invitrogen, US) at a 1:4 ratio. The solution was placed in serum-free medium, was added to the cells, and incubated at 37°C for 6 hours. The transfection medium was replaced with regular growth medium after incubation.
Real-time PCR analysis and Western blot
Total RNA was extracted from excised TSCC samples and cultured cells by using TRIzol® reagent according to the manufacturer's protocol. RNA yield and purity were quantified by UV absorbance spectroscopy. cDNA was synthesized using Promega M-MLV (Promega, WI, USA) following the supplier's instructions. Real-time PCR was performed using SYBR® Premix Ex Taq™ (Takara biotechnology, Shanghai, China). Primers' sequences for RIPK4 were: 5′- TGG CTG CCA CTG CAC TAC GC −3′ (forward) and 5′- GGC GTT CAC ACT CAC CCC CG −3′ (reverse). Primer sequences for GAPDH were: 5′- GAG GAC CAG GTT GTC TCC TG −3′ (forward) and 5′- GGA TGG AAT TGT GAG GGA GA −3′ (reverse) (15). Each sample was measured in triplicates.
We analyzed the RIPK4 expression in Tca-8113 cells transfected with different plasmids by Western blot. Cells were lysed in RIPA buffer (Beyotime Institute of Biotechnology, Shanghai, China) and centrifuged at 3000 g for 10 minutes at 4°C, and protein concentrations were determined using a bicinchoninic acid protein assay with bovine serum albumin (BSA) as the standard (Sigma, Missouri, USA). After quantitation, equal amounts of protein (50 μg) from each sample were loaded on sodium dodecyl sulfate (SDS) polyacrylamide gel and transferred to a polyvinyldifluoride membrane (Bio-Rad, Milan, Italy). The primary antibodies used were anti-RIPK4 (Abcam, Massachusetts, USA) and β-actin (Abcam, Massachusetts, USA). Protein expression was determined with horseradish peroxidase-conjugated secondary antibodies (Abcam, Massachusetts, USA) and enhanced chemiluminescence (Beyotime Institute of Biotechnology, Shanghai, China). The protein levels were normalized to the β-actin levels.
Morphologic observation and RIPK4 expression
The cell morphology was assessed and evaluated by a confocal laser scanning microscope (Olympus IX-UCB, X-Cite 120®, Tokyo, Japan). The normal oral keratinocyte cell line (NOK, Shanghai Zhonghua Company, China) was used as a positive control. Cells (1×105) were grown on glass bottom culture dishes and fixed with acetone for 10 minutes, then they were incubated further with rabbit-anti-RIPK4 monoclonal antibodies (Abcam, Cambridge, UK), Cy-3 (Millipore, Darmstadt, Germany) and 10 μg/mL 4′,6-diamidino-2-phenylindole dihydrochloride (DAPI, Yuanye Biological Technology, shanghai, China). The cells were then washed in PBS and immediately examined.
Scratch wound healing assay and matrigel invasion assays
Cells were seeded and grown to reach a confluent monolayer. Wounds were created with a 20-μL pipette tube, and washed twice with PBS. After incubation for 72 hours, images were taken and the migration distance was determined by subtracting the values obtained at 0 hours and 72 hours.
The invasion and growth ability of cells was measured by using the Matrigel-coated transwell and the non-coated transwell. Briefly, cancer cell lines with or without RIPK4 overexpression were seeded in 1% FBS on the upper chamber of Matrigel invasion chambers (8 mm pore size, BD Biosciences, San Jose, CA), while the lower chamber contained 20% FBS, which served as the chemoattractant. After 72 hours, non-invading cells on the upper surface of the inserts were removed. The cells on the lower surface of the insert were fixed in methanol and stained with crystal violet. The number of cells migrated through the Matrigel was counted under light microscope. All experiments were carried out in triplicates.
Cell cycle and apoptosis assessment by flow cytometry
Different cell cycle phases (G1, S, and G2/M phase) are characterized by different DNA contents. After the treatments, cells were collected by trypsinization and fixed overnight with 70% ethanol. The cell pellets were stained with 10 μg/mL DNA PREP Stain (Beckman coulter, USA) and fluorescence signals from the stained cells were collected in the FL-2 detector. The samples were analyzed by using flow cytometer (BD LSR II, USA). A total of 100,000 events were collected.
Apoptosis was measured by Annexin/propidium iodide (PI) double staining (Pharmingen, San Diego, USA) according to the manufacturer's instructions. Briefly, the cells were incubated with 10 mg/mL of Annexin V-fluorescein isothiocyanate and 5 μg/mL of PI, and analyzed with a flow cytometer.
Cisplatin sensitivity analysis and cell proliferation analysis
First, 1×104 cells per-well were seeded into a 96-well plate and incubated for 24 hours. Cisplatin (Sigma, MO, USA) was added at the concentrations of 25 μM, 50 μM, and 75 μM, and cells were then incubated. After 24 hours, the cell proliferation ability was assessed using the CCK8 reagent (Longteng Bio-Science, Shanghai, China) according to manufacturer's instructions. Briefly, 10% CCK8 labeling reagent was added and, after incubation for 4 hours, the absorbance was read at 450 nm with a micro-plate reader (Bio Tek Instruments, Inc., Vermont, USA).
Analysis of cisplatin-induced activation of caspase 8
We cultured Tca-8113 cells transfected with pRIPK4, pRIPK4-shRNA, or control pNC-shRNA. After 24 hours, 95-D cells were treated with 50 μM cisplatin (Sigma, MO, USA) for 24 hours. After 24 hours, 100 μL of Caspase-Glo 8 reagent (Promega, Shanghai, China) was added to each well and cells were incubated at room temperature for 2 hours. Luminance was measured using Luminometer (Promega, Shanghai, China) and each sample was measured in triplicates.
Statistical analysis
Data are expressed as means ± SD and represent at least 3 independent experiments. Continuous variables from the in vitro studies were compared with the Student's t test or ANOVA, as appropriate. Categorical variables in tissue assessments were compared using the Chi-square test. Multivariate logistic regression analysis was used to investigate the association between RIPK4 levels and age, differentiation grades, gender, secondary education, alcohol consumption, smoking habit, diabetes, hypertension, and lymph node metastasis. Differences in experiments comparing more than 2 groups were assessed using ANOVA with Bonferroni post hoc correction. Differences were considered as statistically significant when p<0.05.
Results
Involvement of RIPK4 levels in TSCC epithelia differentiation
The expression of RIPK4 in normal tongue epithelia from paracancerous normal tissue and tumor samples were examined by immunohistochemistry. In 36 paracancerous normal tissues (N), 33 samples scored 3+ and only 3 samples scored 2+. In 32 highly differentiated tumors (H), 19 samples scored 2+. In 23 moderately differentiated tumors (M), 16 samples scored 1+, and 6 samples scored 2+. Nine of the 18 poorly differentiated tumor (L) samples scored 0, 7 samples scored 1+, and only 2 patients scored 2+. RIPK4 expression gradually decreased in highly, moderately, and poorly differentiated TSCC (Figs. 1A and B, r=0.34, p<0.01): from a mean of 2 in highly differentiated TSCC, to 1.2 in moderately differentiated TSCC, to 0.54 in poorly differentiated TSCC (Fig. 2). Paracancerous normal tissues had the highest RIPK4 levels, while the poorly differentiated TSCC had the lowest RIPK4 mRNA levels (Fig. 2B). This indicated that there is a significant correlation between the RIPK4 levels and the TSCC degree of differentiation. RIPK4 levels increased with age (r=0.66, p=0.04), from a mean of 1.13 in patients from 31 to 55 years old, to 1.65 in patients from 55 to 69 years old (Tab. I, r=0.03, p<0.01).
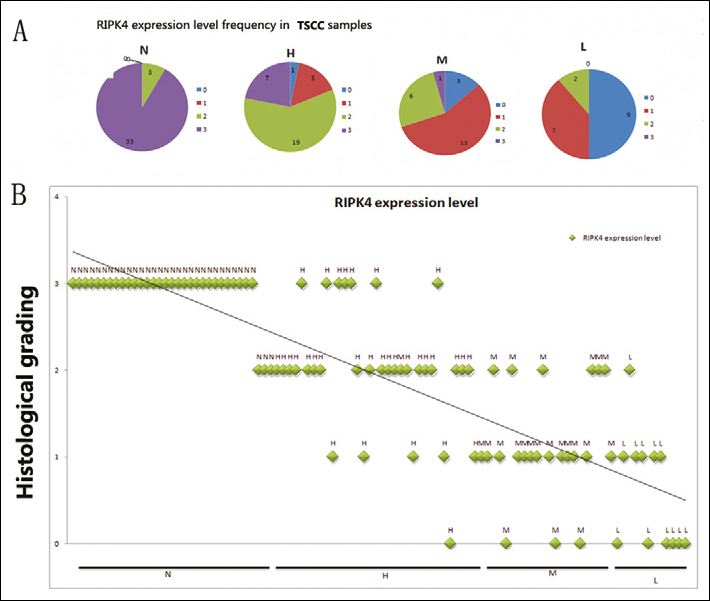
Distribution of RIPK4 in paracancerous normal tissue and surgically excised TSCC samples.
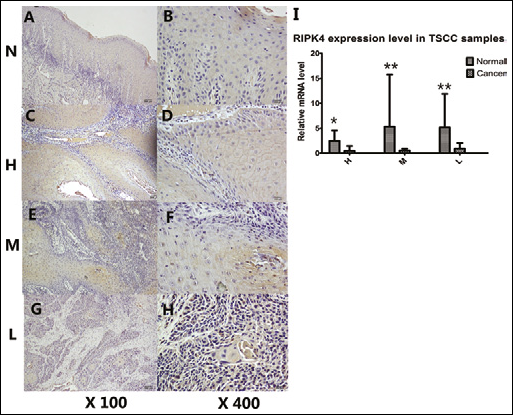
Tumor samples (L) and paracancerous normal tissues were examined by immunohistochemistry. Photomicrographs were taken at 400× and 100× magnifications. Representative immunohistochemical membrane staining of RIPK4: (N) RIPK4, 3+ (strong, uniform, and complete membrane staining in >30% of tumor cells); (H) RIPK4, 3+ (strong, uniform, and complete membrane staining in >30% of tumor cells); (L) RIPK4, 0 (negative); and (M) RIPK4, 1+ (incomplete, weak membrane staining in >10% of tumor cells). A, C, E, and G (×100); B, D, F, and H (×400).
Subjects' Characteristics
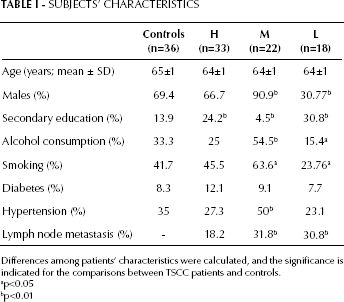
Differences among patients' characteristics were calculated, and the significance is indicated for the comparisons between TSCC patients and controls.
p<0.05
p<0.01
Additionally, we found a significant association between RIPK4 levels and gender (Tab. II). The male group had relatively higher RIPK4 levels (p=0.03, Chisquare test). By contrast, educational level, alcohol consumption, smoking habit, diabetes, hypertension, and lymph node metastasis showed no impact on RIPK4 levels.
RIPK4 Levels by Selected Patients' Characteristics
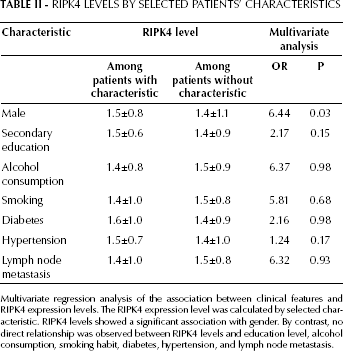
Multivariate regression analysis of the association between clinical features and RIPK4 expression levels. The RIPK4 expression level was calculated by selected characteristic. RIPK4 levels showed a significant association with gender. By contrast, no direct relationship was observed between RIPK4 levels and education level, alcohol consumption, smoking habit, diabetes, hypertension, and lymph node metastasis.
RIPK4 regulates the migration and invasion in Tca-8113 cell lines
To determine whether in vitro RIPK4 plays an important role in regulating cell differentiation and affects the cellular migration and invasion, the Tca-8113 cell line was transfected with pRIPK4, pNC-shRNA, or pRIPK4-shRNA. The RIPK4 transfection rates of the 4 different groups were all above 60% and did not show any significant difference (Fig. 3E). The RIPK4 mRNA expression and the migration and invasiveness of Tca-8113 were measured. As shown in Figs. 3A and 3D, an upregulation of RIPK4 mRNA was found in Tca-8113 cells that were transfected with a plasmid vector encoding human RIPK4. Correlating with the decline in RIPK4 expression in cells transfected with pRI-PK4-shRNA (Figs. 3A, 3D), significant increases in migration (Fig. 3B) and invasiveness (Fig. 3C) were observed in Tca-8113 cells. These results show that RIPK4 plays an important role in Tca-8113 cell migration and invasion. Confocal laser-scanning images showed that RIPK4 levels dramatically declined in the cytoplasm of Tca-8113 cells and TSCC samples, when compared with RIPK4-strongly expressing NOK cells and normal tongue tissue (Fig. 4). No statistically significant differences were observed in Tca-8113 apoptosis and cell cycle progression when cells were transfected with pRIPK4 and pshRNA for 48 hours without cisplatin induction (Fig. 6).
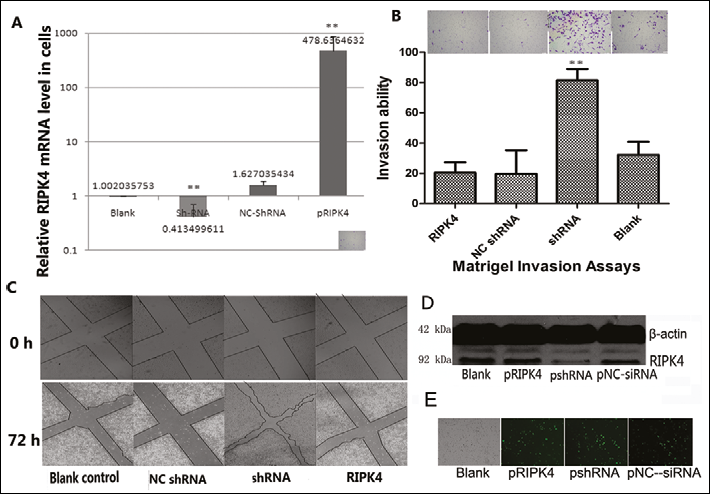
A. Tca-8113 cells with or without a plasmid vector encoding the human RIPK4, NC-shRNA, or RIPK4-shRNA were analyzed for their RIPK4m-RNA expression level. The expression level of RIPK4 mRNA was significantly lower in shRNA-transfected cells. Results show the relative mRNA expression level of the RIPK4 gene in Tca-8113 cells. Data represent 3 independent experiments and are presented as mean ± SD; **p<0.01.
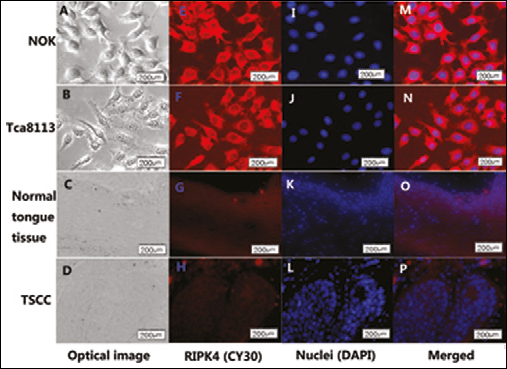
The cells were incubated with the anti-RIPK4 antibody Cy-3, then the nuclei were stained with DAPI. The fluorescence intensity taken up into the cells was observed under a confocal laser scanning microscope. The red color indicates RIPK4 and the blue indicates the nuclei. Graphs A, B, C, and D are the optical images, while graphs E, F, G, and H are the fluorescent images of RIPK4 with Cy-3 staining (red); graphs I, J, K, and L are the fluorescent images of nuclei with DAPI staining (blue), while graphs M, N, O, and P are the merged images. Representative images of positive controls (NOK, normal epithelium) indicate a strong expression of RIPK4. The picture of the Tca-8113 and TSCC samples shows RIPK4 expressed in the cytoplasm and cytomembrane.
Overexpression of RIPK4 increased the cisplatin-induced activation of caspase 8
We then assessed whether an elevated RIPK4 expression could increase chemosensitivity to cisplatin in tongue cancer cells. Different concentrations of cisplatin were used to treat shRNA/RIPK4-transfected Tca-8113 cells for 24 hours. We found that cell growth was significantly increased in knockdown cells after cisplatin treatment (Fig. 5A). RIPK4 knockdown could increase chemosensitivity to cisplatin in tongue cancer Tca-8113 cell lines. Additionally, RIPK4 activated caspase 8 when Tca-8113 cells were treated with 50 μM cisplatin (Fig. 5B).
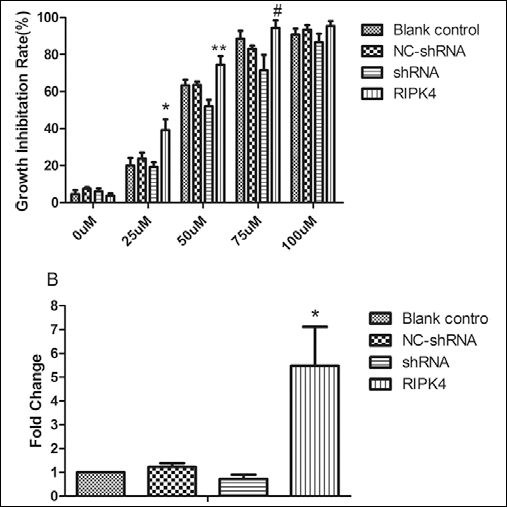
A. pRIPK4, pRIPK4-shRNA, pNC-shRNA, and blank control transfected cells were treated with the indicated concentrations of cisplatin for 24 hours. *p=0.003; **p=0.003; #p=0.0039.
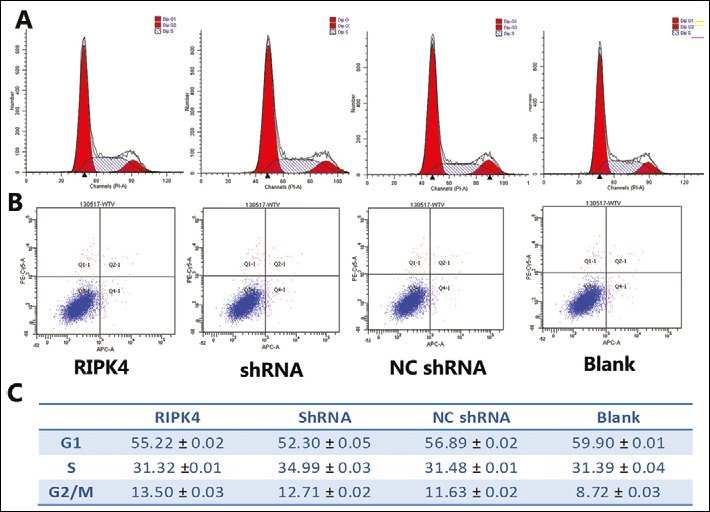
A, C. representative cell cycle progression of Tca-8113 cells. Cell phase percentages were obtained from experiments in triplicates. No statistically significant differences were found among the cell cycle of Tca-8113 cells transfected with the different plasmids and not cisplatin-induced (one-way ANOVA, p=0.45, n=3).
Discussion
RIPK4 promotes epidermal differentiation and wound repair (10–12). Little attention was paid until now to the expression level, subcellular localization, biological function, and tissue localization of RIPK4 in human tumors. In this study, we showed that RIPK4 is involved in cancer pathogenesis of TSCC.
Immunocytochemistry for RIPK4 is currently the gold standard for quantitative assessment of RIPK4 levels in breast cancer tissue and has been shown to provide clinically important prognostic and predictive information (16–18). The scoring system of this assay is based on the number of receptors on the membrane of cancer cells. Our results showed that a reliable assessment of RIPK4 levels in TSCC samples with various differentiation grades should result in a similar classification as the one obtained with by IHC. We found that the lower the differentiation grade, the lower was the expression level of RIPK4. Furthermore, older patient, as well as females, showed a higher RIPK4 expression and a higher differentiation grade. Therefore, RIPK4 could be used by the pathologist as a marker to objectively distinguish the TSCC differentiation grade.
Histological images showed that in moderately and poorly differentiated tumors RIPK4 was expressed predominantly in the cytoplasm (Fig. 2), while highly differentiated tumors and normal tongue tissues mainly expressed RIPK4 in the cytomembrane and cytoplasm. Different translocation patterns may induce a different molecular biological function, which may be confirmed in future experiments. However, Western blot analysis of the tissue samples did not detect RIPK4; this may be due to the low RIPK4 protein expression in mucosa, which could be confirmed by immunohistochemistry.
During cancer progression, poorly differentiated cancers appear to present a higher degree of cell migration, thus promoting the dissemination of cells from the tumor mass (19). Our study showed that RIPK4 knockdown resulted in morphologic changes as well as in increased capabilities of migration and invasion of tongue cancer cells. A higher degree of cell migration and the invasive behavior are associated with the reduced expression of RIPK4, which could be further confirmed by the statistical analysis of lymph node metastasis (Tab. I, r=2, p<0.01).
Caspases are central regulators of apoptosis, cleaving cellular proteins at specific aspartate residues in the apoptotic process (20). It is already known that apoptotic proteins target and affect mitochondria in different ways. The kinase properties of RIPK4 are critical for NF-κB and JNK activation and are triggered during apoptosis (7). As RIPK4 contains an N-terminal kinase domain that, when cleaved by caspases, plays essential roles in apoptosis, necrosis, and inflammation (5, 6, 20, 21), we evaluated the involvement of caspase 8 in this process by using the human tongue cancer cell line Tca-8113. Our results demonstrate that cisplatin-induced apoptosis in Tca-8113 cells was mediated by the overexpression of RIPK4. These data are consistent with those reported in a previous study indicating that RIPK4 could interact with caspase 8 (5).
However, our transfection results showed that there were no significant changes in Tca-8113 apoptosis and cell cycle progression without cisplatin induction. Other cancer cell lines should be used to investigate the effect of RIPK4 in apoptosis and cell cycle progression. Future developments on this issue can be focused on exploring the mechanism of action, or investigating other cell lines.
In the scratch wound healing assay and matrigel invasion assay, we either overexpressed or knocked down RIPK4. The overexpressing Tca-8113 cells did not show any difference from the blank controls, while the cells transfected with shRNA showed an increase in their invasion ability.
Our results indicate the possibility that RIPK4 may be involved in human tumor differentiation. Additional research is needed not only to confirm these findings in other cancer samples, but also to clarify the importance of RIPK4, and to determine whether tumor RIPK4 levels are a cause of tumor differentiation. RIPK4 levels were not associated with education level, alcohol consumption, smoking habit, diabetes, hypertension, and lymph node metastasis, but were associated with gender and age.
Footnotes
Acknowledgment
We thank Prof. Mathieu JM Bertrand (VIB/Ghent University, Belgium), who kindly provided the plasmid RIPK4.
