Abstract
Differentiation is a major histological parameter determining tumor aggressiveness and prognosis of the patient; cancer stem cells with their slow dividing and undifferentiated nature might be one of the factors determining the same. This study aims to correlate cancer stem cell markers (CD44 and CD147) with tumor differentiation and evaluate their subsequent effect on prognosis. Immunohistochemical analysis in treatment naïve oral cancer patients (n = 53) indicated that the expression of CD147 was associated with poorly differentiated squamous cell carcinoma and moderately differentiated squamous cell carcinoma (p < 0.01). Furthermore, co-expression analysis showed that 45% each of moderately differentiated squamous cell carcinoma and poorly differentiated squamous cell carcinoma patients were CD44high/CD147high as compared to only 10% of patients with well-differentiated squamous cell carcinoma. A three-way analysis indicated that differentiation correlated with recurrence and survival (p < 0.05) in only the patients with CD44high/CD147high cohort. Subsequently, relevance of these cancer stem cell markers in patterning the differentiation characteristics was evaluated in oral squamous cell carcinoma cell lines originating from different grades of oral cancer. Flowcytometry-based analysis indicated an increase in CD44+/CD147+ cells in cell lines of poorly differentiated squamous cell carcinoma (94.35 ± 1.14%, p < 0.001) and moderately differentiated squamous cell carcinoma origin (93.49 ± 0.47%, p < 0.001) as compared to cell line of well-differentiated squamous cell carcinoma origin (23.12% ± 0.49%). Expression profiling indicated higher expression of cancer stem cell and epithelial–mesenchymal transition markers in SCC029B (poorly differentiated squamous cell carcinoma originated; p ≤ 0.001), which was further translated into increased spheroid formation, migration, and invasion (p < 0.001) as compared to cell line of well-differentiated squamous cell carcinoma origin. This study suggests that CD44 and CD147 together improve the prognostic efficacy of tumor differentiation; in vitro results further point out that these markers might be determinant of differentiation characteristics, imparting properties of increased self-renewal, migration, and invasion.
Introduction
Multiple histopathological and clinical features are known to be effective determinants of prognosis in oral cancer. Tumor differentiation represents one of the most important histologic markers that predict metastasis and disease recurrence.1–4 At a cellular level, differentiation is a representation of the tumor cell phenotype, wherein well-differentiated tumors, of low mitotic index and high keratinization, are cytologically closer to normal tissues in comparison with poorly differentiated tumors, which show a loss of epithelial characteristics. 5 Although tumor differentiation is used clinically as a prognostic factor, the molecular and cellular mechanisms underlying these changes are not well studied.
Cancer stem cells (CSCs) are currently implicated in different aspects of tumorigenesis and clinical behavior. The inherent slow dividing, drug Resistant, and “undifferentiated” phenotype of these cells correlates with the properties of tumor initiation, relapse, and drug resistance. 6 Studies in this area have identified multiple markers that specify head and neck cancer–specific CSCs. CD44, a type-I transmembrane protein, is one of the most commonly studied marker of head and neck CSCs and is known to have the ability to regulate cell proliferation, differentiation, migration, invasion, and treatment resistance. 7 CD147, another CSC marker of head and neck cancer, is also reported to be involved in oral tumor progression. 8 These markers have been correlated with poor prognosis, metastasis, and higher rates of recurrence.8–11 Given the abundant evidences supporting the undifferentiated nature of the CSCs and their potential to form aggressive tumors, 6 their possible involvement in the genesis of tumor histology and grade is now being postulated. 5 Although there is evidence in gastric cancer pointing out a similar correlation, 12 this is not established in head and neck cancers yet. In this study, we attempted to investigate the possible role of CSCs, as specified by the two markers, CD44 and CD147, in differentiation and prognosis of oral squamous cell carcinoma (OSCC).
Materials and methods
Patients and tissue samples
Patients diagnosed with squamous cell carcinoma of oral cavity between December 2010 and January 2013, at the Head and Neck Oncology Department of a tertiary referral center, were selected retrospectively for inclusion into this study. The inclusion criteria of the patients were (1) treatment naïve, (2) treatment with curative-intent surgery, and (3) availability of histopathology blocks. The exclusion criteria were as follows: (1) patients presenting with recurrent tumor and (2) patients previously treated with chemotherapy and/or radiation. Tumor blocks were collected from the Department of Pathology at Mazumdar Shaw Medical Centre (MSMC), Bangalore after obtaining Institutional Ethical Committee (IEC) approval and categorized into poorly differentiated squamous cell carcinoma (PDSCC), moderately differentiated squamous cell carcinoma (MDSCC), and well-differentiated squamous cell carcinoma (WDSCC) patients. Histological grading was determined by pathologists according to World Health Organization (WHO) grading system. 13 Normal samples were collected from patients undergoing dental extraction. All samples collected from patients and normal controls were obtained after written informed consent. Clinical, demographic, treatment, and follow-up details were obtained from the patient medical records.
Immunohistochemical analysis
Sections from paraffin-embedded tumor tissue blocks were prepared as per standard protocol and independently evaluated (after hematoxylin–eosin staining) by two pathologists. For immunohistochemistry (IHC), the sections were de-paraffinized, re-hydrated, incubated with primary antibodies (CD44: AM310-5M; BioGenex Life Sciences Pvt. Ltd., India; CD147: 1:200, 555961; BD Pharmingen™, USA), and stained using Real™ EnVision™ Detection System (K500711-2; Dako, Denmark) according to manufacturer’s instructions. The immunohistochemical scores were obtained by multiplying the percentage of positive epithelial cells (0%–100%) and staining intensity scores (mild/1, moderate/2, and strong/3). The scoring was done by two independent observers. The CD44 and CD147 expression score in normal mucosa was used as a control.
Cell culture
Six OSCC cell lines UPCI:SCC029B and AW13516 (originated from PDSCC), UPCI:SCC040 and CAL-27 (originated from MDSCC), and UPCI: SCC103 and UPCI: SCC16 (originated from WDSCC)14–16 were obtained from University of Pittsburgh, USA; Institute of Bioinformatics (IOB), India; and ACTREC, India and used in this study. Cells were cultured in monolayer in Dulbecco’s Modified Eagle’s Medium (DMEM) medium (11995-065; Gibco™, Thermo Fisher Scientific, USA) supplemented with 10% fetal bovine serum (FBS, 10082-147; Thermo Fisher Scientific), 1× penicillin and streptomycin (15-240-062; Gibco™, Thermo Fisher Scientific), and 1× non-essential amino acid (11140-050; Invitrogen, Thermo Fisher Scientific). Cultures were maintained at 37°C with humidified atmosphere of 5% CO2.
Flow cytometry analysis
The 70%–80% confluent monolayer cells of all the six cell lines were trypsinized, washed, and re-suspended in the medium with 10% FBS and counted prior to staining for flow cytometry analysis. In 100 µL of buffer (phosphate-buffered saline (PBS) + 0.5% FBS), 1 × 106 cells were incubated with the anti-CD44-APC (559942; BD Pharmingen) and anti-CD147-FITC (555962; BD Pharmingen) on ice for 30 min. The cells were then washed with buffer and fixed in 1% paraformaldehyde for 30 min on ice. After fixation, cells were washed and the fluorescence staining was assessed in the FL1 and FL4 fluorescence channel in flow cytometer (FACS Calibur; BD Biosciences, USA). The percentage of cells positive for each marker/combination was compared across the different cell lines and statistical significance was evaluated.
Stem cell gene expression profiling
Total RNA was extracted from 1 × 106 cells using RNA extraction kit (740933.50; NucleoSpin® RNA/Protein; Macherey-Nagel, Germany). The integrity and quality of RNA were ascertained by gel electrophoresis and by the 260/280 ratio in Nanodrop (NanoVue Plus, USA). A volume of 1 µg of RNA (260/280: 1.8–2.0) was converted into complementary DNA (cDNA) by the cDNA Conversion Kit (4374966, High-Capacity cDNA Reverse Transcription Kit with RNase Inhibitor; Applied Biosystems, USA) as per the manufacturer’s instructions. The primers for selected markers (Supplementary Table 1) were evaluated for specificity using Basic Local Alignment Search Tool (BLAST) analysis (National Center for Biological Information USA). The efficiency of each primer in quantitative real-time polymerase chain reaction (qPCR) was assessed using the slope of the linear regression model as per standard protocols and calculated using the following formula: efficiency = 10(−1/slope). All real-time PCR reactions were performed in triplicates using the LightCycler® 480 Real-Time PCR System (Roche Diagnostics, Germany) using the Kapa SYBR Green PCR Master Mix (KK4602; Kapa Biosystems, USA). A set of reference genes (RGs, n = 4; glyceraldehyde-3-phosphate dehydrogenase (GAPDH), 60S acidic ribosomal protein P0 (RPLP0), 18S ribosomal RNA (rRNA), and hypoxanthine-guanine phosphoribosyl transferase (HPRT)) were assessed using the RefFinder. 17 Across the three cell lines, two of the best RGs were selected for further analysis. The relative change in expression was evaluated using the geometric mean of the selected RGs. 18 The fold level of each marker in SCC029B and SCC040 cell lines was calibrated against the expression in SCC103 cell line.
Spheroid formation assay
Cells were trypsinized and the required number of cells (1200 cells/well) were re-suspended in serum-free DMEM/F12 medium (11320033; Gibco™, Thermo Fisher Scientific) supplemented with 1% N2 supplement (17502048; Gibco™, Thermo Fisher Scientific), 20 ng/mL of human recombinant epidermal growth factor (EGF, E9644; Sigma Aldrich, USA), 5 µg/mL of insulin (1927-5 mL; Sigma Aldrich), and 20 ng/mL human basic fibroblast growth factor (b-FGF, PHG6015; Gibco™, Thermo Fisher Scientific) and cultured in 24-well ultra-low attachment plates for 2 weeks. The medium was supplemented with the growth factors every 3 days. The number of spheroids generated from each cell lines were counted (at the end of first week) using a phase-contrast microscope (Carl Zeiss Scope A1; Zen Software, Germany) in 100× magnification. The assay was carried out in triplicates.
Wound healing assay
Cells were seeded into 24-well tissue culture plates at a density of 1.5 × 105 cells per well. When the cells reached 70%–80% confluence, a scratch was made across the well with the 200 µL micro tip. The wells were replenished with fresh medium, and the cells were grown for an additional 36 h with periodical recording of wound closure. The wound areas were imaged and measured using a phase-contrast microscope (Carl Zeiss Scope A1; 40× magnification), and the percentage of wound closure was compared at various time points (12, 24, and 36 h). The wound was measured as an average (n = 3) of the area between the wound boundaries (in pixels), and the percentage of wound closure is calculated by the formula (A0−An/A0) × 100, wherein A0 is the average area at 0th hour and An at nth hour. The assay was carried out in triplicate.
Matrigel invasion assay
Cells were cultured as a monolayer in serum-free DMEM media for 12 h. Extracellular matrix (ECM) gel (E1270; Sigma Aldrich) was thawed at 40°C overnight. ECM was diluted to 1.5 mg/mL with serum-free DMEM media and 100 µL was added to the upper compartment of the 24-well Millicell insert (Z681946–12EA; Sigma Aldrich). The insert was kept on a 24-well plate at 37°C overnight and 20,000 cells were placed on the gel. After 48 h of incubation at 37°C at 5% CO2, inserts were carefully taken out, and cells and gel in the upper compartment of the insert were wiped gently with cotton swab. Inserts were washed with 1× PBS, and cells on the lower side of the insert membrane were fixed with 3.7% paraformaldehyde for 10 min and washed with 1× PBS. The cells were permeabilized with 100% methanol for 10 min and then stained with Giemsa for 1 h. The cells were washed with 1× PBS and the inserts were then mounted onto a slide. The number of cells on the lower side of the filter was counted under microscope (E200; Nikon, Japan) at 100× magnification, and the percentage of invaded cells was calculated using the following formula: percentage of invaded cells = (total number of cells counted/total number of cells seeded) × 100.
Statistical analysis
The statistical analysis was performed by STATA 11.2 (USA). Chi-square test was used to measure the association between the CD44 and CD147 with recurrence, prognosis, and differentiation. Kaplan–Meier survival curve was used to estimate the association of survival function with co-expression of CD44 and CD147. Comparisons between groups were evaluated by Student’s t-test, and analysis of variance (ANOVA) with Bonferroni correction was applied to find the significant difference between the severities of differentiation with gene/protein expression. Multivariate analysis was done to co-relate clinical parameters and marker expression with prognosis of patients. All data are represented as mean values ± standard error of mean (SEM); p < 0.05 is considered as statistically significant.
Results
CD147 and CD44 marker expression correlates with tumor differentiation
Patient details
During the period of 2010 to 2013, a total of 254 patients were diagnosed with cancers of the oral cavity. Among these patients, 150 patients were excluded as per the exclusion criteria, 51 patients were excluded due to non-availability of samples, and 53 patients met the strict inclusion/exclusion criteria and were analyzed in this study. Of the patients, 23% (12/53) suffered from cancer of the buccal mucosa, 54% (29/53) from cancer of tongue, and 23% (12/53) from cancer of other sites of the oral cavity. Among the patient cohort, 82% (40/49) of patients are with risk habits. These patients include chewers (n = 17), smokers (n = 5), and drinker (n = 1). Additionally, 37% (15/40) of the patients were with multiple risk habits, while information was unavailable for 2 patients. Histologically, a majority of tumors were moderately (24/53, 45%) or well differentiated (16/53, 30%); 40% (21/53) of patients had advanced disease (T4 stage, pathological stage), while 60% had nodal metastases (32/53). Information of metastatic disease was available in 40 patients, among which 3 (8%, 3/40) had distant metastases. Patients were followed up for a median period of 24 months (range 24–46 months). Six patients were lost to follow-up (Table 1). Among the patients (n = 47) with follow-up, 62% (29/47) patients were alive and free of disease during the study period, while 28% (13/47) of the patients developed recurrence during follow-up (Table 1).
Details of patient cohort.

PDSCC: poorly differentiated squamous cell carcinoma; MDSCC: moderately differentiated squamous cell carcinoma; WDSCC: well-differentiated squamous cell carcinoma.
Information unavailable for the remaining patients.
Immunohistochemical analysis
Expression of CD44 and CD147 by IHC was carried out separately with the expression in normal tissues taken as control (Figure 1(a)). Assessment of the average scores of CD147 between the differentiation groups indicated that expression level was significantly more in PDSCC (123.2 ± 7.8, p < 0.0001) and MDSCC (84.3 ± 8.2, p < 0.01) than in WDSCC tumors (53.0 ± 6.6). However, the expression of CD44 in PDSCC (115.7 ± 20.4) and MDSCC (108.8 ± 13.3) in comparison to WDSCC (113.4 ± 15.6) did not show a significant difference (Figure 1(b)).

CD44/CD147 marker expression in PDSCC, MDSCC, and WDSCC patients: the expression profiling of CD44 and CD147 was carried out by IHC in PDSCC, MDSCC, and WDSCC patient cohort. (a) Representative images of these markers in the cohort (400× magnification). (b) Expression of CD44 and CD147 in PDSCC, MDSCC, and WDSCC tumors is shown wherein the expression of CD147 being highest in PDSCC (p < 0.001) than in MDSCC (p < 0.01) as compared to WDSCC tumors. Difference in CD44 expression was not significant. (c) Combinatorial expression of CD44/CD147 in patients wherein CD44high/CD147high and CD44low/CD147high marker combination correlates with degree of differentiation of tumor. (d) Patients with CD44high/CD147high expression with increased percentage of patients in PDSCC and MDSCC as compared to WDSCC tumors (p < 0.001; **p < 0.01, and ***p < 0.001).
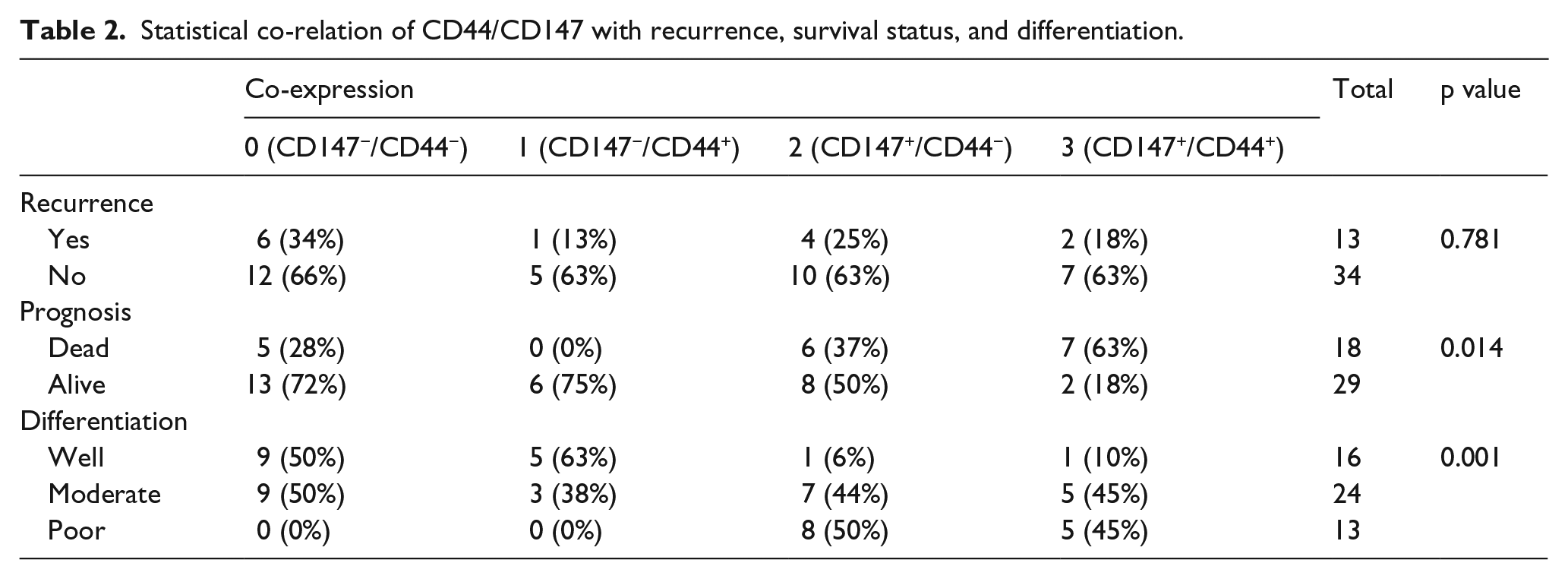
The patients were further categorized into low- and high-expression groups based on the median expression (CD147: 74; CD44: 100) of the markers. Based on the co-expression of the markers, the patients were categorized into four groups, that is, CD44high/CD147high, CD44low/CD147high, CD44high/CD147low, and CD44low/CD147low. CD44high/CD147high and CD44low/CD147high combination co-related with degree of differentiation of tumor (p < 0.001; Figure 1(c), Table 2). In the overall patient cohort, 21% (11/53) of the patients were CD44high/CD147high. Analysis to correlate the risk habits of patients with the marker expression pattern showed that 22% (9/40) patients with risk habits were CD44high/CD147high. Assessment of this expression pattern in the patients with varying severity of differentiation indicated that 45% PDSCC (5/11) and MDSCC (5/11) patients were CD44high/CD147high as compared to 10% of the WDSCC patients (1/11; Figure 1(d)). Furthermore, a combined analysis of differentiation with risk habits indicated that the CD44high/CD147high marker expression co-related with poor differentiation in patients with risk habits (PDSCC 56% (5/9), MDSCC 33% (3/9), and WDSCC 11% (1/9); p < 0.01).
Statistical co-relation of CD44/CD147 with recurrence, survival status, and differentiation.
Expression of CD44 and CD147 correlates with disease prognosis
The patients were evaluated regarding overall survival (OS) status based on their differentiation grades and marker expression patterns. Multivariate analysis revealed that differentiation of tumor, T stage, age, and gender showed a trend of co-relation (not statistically significant) with survival of patients (Supplementary Table 3), while sites of tumor did not show a correlation. Kaplan–Meier analysis indicated poor survival of patients with PDSCC (p < 0.05; Figure 2(a)). Among the four categories based on marker expression, the patients in the CD44high/CD147high and CD44low/CD147high groups had a poor prognosis as compared to the other group (p = 0.06; Figure 2(b)). Co-expression of markers (CD44/CD147) also co-related significantly with survival of patients in univariate analysis and in chi-square test (Supplementary Table 3).

Correlation of CD44/CD147 expression with differentiation and prognosis of patients: (a) KM survival analysis showed the presence of PDSCC as a poor prognosticator in overall survival (p < 0.05). (b) Survival analysis based on the expression pattern indicated that patients with CD44high/CD147high and CD44low/CD147high had poor prognosis, though not statistically significant (p = 0.06). (c) Correlation of marker combination with recurrence indicated that a higher percentage of PDSCC patients recurred in CD44high/CD147high and CD44low/CD147high groups. (d) Correlation of marker expression with overall survival of patients also indicated that an increased percentage of PDSCC patients died in CD44high/CD147high and CD44low/CD147high groups (p < 0.05; *p < 0.05).
To further evaluate the correlation of tumor differentiation with prognosis in the background of CD44/CD147 marker expression, a three-way analysis was carried out. Assessment of prognosis with tumor differentiation in the background of marker expression showed that different grades of differentiation correlated with recurrence (18% PDSCC, 0% MDSCC, and 0% WDSCC) and deaths (27% PDSCC, 27% MDSCC, and 9% WDSCC, p < 0.05) in the patients with CD44high/CD147high expression pattern. Similar results were obtained in patients with CD44low/CD147high expression (25% PDSCC, 0% MDSCC, and 0% WDSCC patients recurred and 31% PDSCC, 6% MDSCC, and 0% WDSCC patients died, p < 0.05; Figure 2(c) and (d), Table 2). However, this correlation of prognosis with tumor differentiation was not found in the other patterns of marker expression.
Data from our lab have proved that grades of differentiation correlate with recurrence and survival in the CD44high group with PDSCC patients showing poor survival and higher recurrence rates (unpublished data). To evaluate the effect of CD147 on prognosis, the three-way analysis was repeated to assess the prognostic efficacy of differentiation in the background of CD147high independently. The correlation was similar in CD147high group, wherein differentiation was associated with recurrence (22% PDSCC, 0% MDSCC, and 0% WDSCC; p < 0.05) and survival (30% PDSCC, 15% MDSCC, and 4% WDSCC; Table 3, Figure 3(a) and (b)). These correlations were not observed in the CD44low (unpublished data) and CD147low groups.
Statistical co-relation of CD147 with recurrence, survival status, and differentiation.

PDSCC: poorly differentiated squamous cell carcinoma; MDSCC: moderately differentiated squamous cell carcinoma; WDSCC: well-differentiated squamous cell carcinoma.

Correlation of independent CD147 expression with prognosis: (a) correlation of marker expression with recurrence indicated that differentiation associated with prognosis in the background of marker expression with a higher percentage of PDSCC patients recurred in CD147high group (p < 0.01). (b) Correlation with overall survival also indicated that a higher proportion of PDSCC patients had poor survival in CD147high group (**p < 0.01).
CD44+/CD147+ enrichment in cell lines of PDSCC and MDSCC origin
The six OSCC cell lines were assessed for the CD44+ and CD147+ cell population by flow cytometry analysis. The results showed that the percentage of CD147+ cells in SCC029B, AW13516, SCC040, CAL-27, SCC16, and SCC103 were 98.93% ± 0.09%, 99.16% ± 0.3%, 99.19% ± 0.10%, 98.3% ± 0.47%, 98.06% ± 0.25%, and 96.87% ± 0.093%, respectively, indicating a stable level of expression across the three cell lines. However, there was a significant increase in percentage of CD44+ population in SCC029B (94.98% ± 1%), AW13516 (98.28% ± 0.17%), CAL-27 (93.2% ± 2.7%), and SCC040 (94.33% ± 0.34%) as compared to SCC16 (50.9% ± 2.1%) and SCC103 (25.91% ± 0.3%; p ≤ 0.001; Figure 4(a) and (c)). Accordingly, assessment of the double-positive cells (CD44+/CD147+) indicated an increase in SCC029B (94.35% ± 1.14%, p < 0.001), AW13516 (96.13% ± 0.6%, p < 0.001), CAL-27 (90.13% ± 2.05%, p < 0.001), SCC040 (93.49% ± 0.38%, p < 0.001), and SCC16 (44.6% ± 1.9%), as compared to SCC103 (23.12% ± 0.49%; Figure 4(b) and (d), Supplementary Figure 1).

Profiling of SCC029B, SCC040, and SCC103 cell lines for the CSC/EMT markers: (a) FACS profiling of the cell lines indicated the presence of >90% CD147+ cells in all the three cell lines. CD44+ cells were higher in SCC029B and SCC040 cell lines compared to SCC103 (p < 0.001). (b) Co-expression of CD44 and CD147 in SCC029B, SCC040, and SCC103 indicated that both SCC029B and SCC040 cells had a higher percentage of CD44+/CD147+ cells in comparison to SCC103 cells (p < 0.001). (c) CD44 and CD147 profiling of six cell lines by FACS indicated the presence of >90% CD147+ cells in all the six cell lines. CD44+ cells were higher in SCC029B, AW13516, CAL-27, and SCC040 cell lines compared to SCC103 (p < 0.001). (d) CD44/CD147 co-expression indicated the presence of more amount of CD44+/CD147+ cells in SCC029B, AW13516, CAL-27, and CSS40 in comparison to SCC103 (p < 0.001). (e) Relative gene expression of CSC markers indicated that CD44 and Oct3/4 were significantly higher in SCC029B and SCC040 (p < 0.001) with SCC029B showing high CD147 expression. (f) Vimentin and Snail expressions were significantly higher in SCC029B and SCC040 cells (p ≤ 0.001), while Collagen 3A1 gene expression was significantly higher in SCC029B (p < 0.001) compared to SCC040 and SCC103. (g) Assessment of markers of invasion also indicated that SCC029B showed a significantly elevated gene expression of N-cadherin, MMP2, and MMP9 (p ≤ 0.001) compared to SCC040 and SCC103. E-cadherin expression did not show any significant difference between the three cell lines (****p < 0.001).
Expression profiling of markers determining CSCs, epithelial–mesenchymal transition, and invasion
Out of the six cell lines, three cell lines (SCC029B, SCC040, and SCC103) were profiled for the expression pattern of markers associated with CSCs, EMT, and invasion using specific primers. All the primers used in the study showed an efficiency ranging from 1.9 to 2.1 with the percentage efficiency being 93%–110%. Analysis by RefFinder indicated that among the RGs assessed, HPRT and 18SrRNA were identified as the most stable genes. qPCR validation of the selected genes was then carried out using the relative quantification method using the geometric mean of the two RGs for normalization. The gene expression profiling of CD44 and CD147 showed a trend similar to that of the flow cytometric analysis. Expression of CD44 was significantly higher (p < 0.001) in SCC040 (9.98 fold) and SCC029B (4 fold) cell lines with expression in SCC103 cell line as the baseline. Although the difference in expression was less, CD147 also showed ~2-fold increase (1.9 fold) in SCC029B as compared to SCC103 (Figure 4(e)).
Profiling of the panel of stem cell marker, OCT3/4, and markers of epithelial–mesenchymal transition (EMT; SNAIL, MMP2, MMP9, VIMENTIN, COL3A1, E-CADHERIN, N-CADHERIN) and invasion (matrix metalloproteinases (MMPs) and cadherin) was carried out in these cell lines. The gene expression analysis revealed significantly elevated levels of OCT3/4 (p < 0.001) in SCC029B (56.9 fold) and SCC040 (40.7 fold) as compared to SCC103 (Figure 4(e)). Among the EMT markers, SCC029B showed a higher expression (p ≤ 0.001) of VIMENTIN (292.4 fold), SNAIL (4.4 fold), and COL3A1 (3 fold) compared to SCC103. In SCC040, VIMENTIN (53.1 fold) and SNAIL (3.9 fold) expression was significantly higher (p ≤ 0.001) than SCC103 (Figure 4(f)).
Assessment of the expression levels of genes regulating tumor invasiveness showed that SCC029B revealed a significantly elevated gene expression of N-CADHERIN (4.6 fold), MMP2 (2.98 fold), and MMP9 (9.2 fold) (p ≤ 0.001) as compared to SCC040 and SCC103. E-cadherin expression did not show any significant difference between the three cell lines (Figure 4(g)).
Differentiation status of cells correlated with increased self-renewal, migration, and invasiveness
All cell lines (SCC029B, SCC040, and SCC103) were evaluated for their spheroid forming potential. A significant increase was observed in the numbers of spheroids formed by SCC029B (10.6 ± 0.54) and SCC040 (3 ± 0.47) as compared to SCC103, which did not form any spheroids (p ≤ 0.001). An assessment of the minimum number of cells required also indicated that spheroid formation was initiated at a lower cell number by SCC029B (n = 300) as compared to SCC040 (n = 1200; Figure 5(a) and (b)).

SCC029B shows increased self-renewal, migratory, and invasive properties. (a and b) Spheroid culture of SCC029B, SCC040, and SCC103 cell lines indicated that SCC029B formed a higher number of spheroids than SCC040. SCC103 could not form any spheroids (p < 0.001) in 100× magnification. (c and d) A comparison between the percentage wound closure of SCC029B, SCC040, and SCC103 at 12, 24, and 36 h, respectively, showed the increased migratory potential of SCC029B, though not statistically significant (40× magnification). (e and f) Matrigel invasion assay revealed a higher percentage of invasive cells in SCC029B cells as compared to SCC040 and SCC103 after 48 h of incubation (p < 0.001; 100× magnification; **p < 0.01, and ***p < 0.001).
The wound healing assay was performed to evaluate the migratory properties of these three cell lines. A comparison between the percentage wound closure of SCC029B, SCC040, and SCC103 at 12 h (29.8% ± 5.9%, 39.5% ± 11.7%, and 48.7% ± 3.2%), 24 h (67% ± 4.8%, 81.6% ± 2%, and 81.1% ± 13.9%), and 36 h (95.9% ± 1.8%, 90.6% ± 11.9%, and 85.6% ± 10.8%), respectively, showed the increased migratory potential of SCC029B at 36 h, though not statistically significant. All the cells completely closed the wound by 48 h (Figure 5(c) and (d)).
The invasion potential of all these three OSCC cell lines was assessed by in vitro matrigel invasion assay. The results showed that more number of SCC029B cells (17% ± 0.03%) could invade through the matrigel as compared to SCC040 (10% ± 0.02%) and SCC103 (9.17% ± 0.008%) cells indicating that a higher proportion of SCC029B/SCC040 showed invasive potential compared to SCC103 (p ≤ 0.001; Figure 5(e) and (f)).
Discussion
Tumor differentiation, defined microscopically by epithelial cell keratinization and keratin pearl formation, 5 is a major prognostic factor in OSCC.2,5 Poorly differentiated tumors are known to be more aggressive and metastasize earlier, leading to poor prognosis. With evidences that characterize CSCs as undifferentiated and attribute tumorigenic, metastatic, and invasive properties to them, 6 this study attempted to delineate the cellular basis for differentiation in OSCC using CSC-specific markers CD44 and CD147.
In head and neck cancer, multiple markers are reported to identify CSCs (CD44, ALDH1, ABCG2, BMI1, OCT4, SOX2, and NANOG), 6 among them CD44 is the most common and well-established CSC marker. CD44+ CSCs are known to define tumorigenicity, drug resistance, and metastasis in head and neck cancer and thereby are correlated with poor prognosis in the disease.7,19 However, reports also attribute CSC properties to CD44− cells, 20 indicating the necessity to use multiple markers to specify the CSC population. 21 CD147/EMMPRIN is an important marker for anchorage independent growth, angiogenesis, drug resistance, hypoxic survival, and invasion, properties attributed to CSCs and which are essential molecular events for carcinogenesis. 22 Additionally, CD147 is also a marker of undifferentiated human embryonic stem cells 23 and has been reported as a marker of oral CSCs. 24 Recent studies have implicated CD147, along with CD44, in modulation of the Wnt/β-catenin pathway 25 in metastatic prostate cancer, a property which is being investigated as potential therapeutic strategy. 26 Given these evidences, a combination of CD44 and CD147 was used to specify oral CSCs in this study.
In this study, the presence of CSCs with a high-expression pattern of CD44/CD147 co-related with tumor differentiation status. Similar studies have been reported in gastric cancers, wherein ABCG2 and CD133-positive CSCs correlated with degree of differentiation of gastric tumor. 12 Also, Zhong et al. 27 have reported that CK19, which is a marker of stem cells, correlates with differentiation grade and poor prognosis in OSCC. 6 Assessment of the prognostic significance in our study indicated poor differentiation in patients to be a predictor of poor prognosis in the background of CD44high/CD147high expression. Our study also showed that while independent expression of CD147 correlated with differentiation (p < 0.001) and poor prognosis (p < 0.05; Table 3, Supplementary Table 2), CD44 was a major contributor to prognostic significance when assessed in combination with tumor differentiation. Taken together, these evidences further emphasized the need to use multiple identifiers for CSCs to delineate their role in the process of differentiation and other tumorigenic processes.
In vitro studies in the cell lines that originated from tissues of varied grades of differentiation revealed that the cell line originating from poorly differentiated tissue (SCC029B and AW13516) showed increased enrichment of CD44+/CD147+ cells, which is in concordance with the results obtained from the patient cohorts. Additionally, expression profiling revealed elevated levels of stem cell marker, OCT3/4 and EMT markers in the SCC029B. OCT3/4 are known to be involved in the pluripotency and carcinogenesis of oral cancer, 28 while MMPs have been well implicated for facilitating the invasion of the stromal ECM by the tumor cells. 29 The balances in expression of N-CADHERIN and E-CADHERIN along with VIMENTIN and COL3A1 have been implicated in inducing a transition to mesenchymal lineage.30–34 Furthermore, an exploration of the functional behavior of these cells indicated that in accordance with the marker profile, cell line of PDSCC origin showed enhanced self-renewal, migratory, and invasive properties. The increased percentage of CSC population in these cells, accompanied by elevated expression of markers of tumor aggressiveness/EMT in combination with the aggressive cellular properties, indicated a possible role for the CSC population in patterning this behavior. Moreover, this is in accordance with the highly aggressive nature of poorly differentiated tumors that are prone to nodal/distant metastasis and disease relapse.35–39 In the in vitro study, CD147 expression is not differential between the cell lines of varied differentiation type and also does not correlate to the migratory/invasive properties. Although other studies have shown that CD147+ cells in ovarian cancer are highly migratory/invasive, 40 this study indicates that it is the percentage of double-positive (CD147/CD44) cells that correlate with the cellular properties of invasion and migration. CD147 (also termed Basigin), a transmembrane glycoprotein, is activated in vivo by signals from the microenvironment (mostly glycans and hyaluron), leading to activation of monocarboxylate transporters (MCTs), which in turn leads to aggressive tumor properties in patients. 41 Whether the presence of CD44 (receptor for hyaluron) contributes toward activation of CD147 in cell lines and thereby increases invasive properties of the CD147+/CD44+ cells and the lack of other microenvironment signals is responsible for the absence of correlation of CD147 alone with tumorigenic properties, need to be thoroughly investigated. Nevertheless, this further emphasizes the need to use multiple markers to specify CSCs to obtain an accurate representation of these cells.
CD44high/CD147high cells in this study correlated with differentiation status of the tumors/prognosis and in combination with the results that attribute enhanced self-renewal/migration and invasive properties indicate a strong role for these cells in determining the histological grade of the tumor. Studies in other cancers such as colon, stomach, esophagus, lung, pancreas, and gall bladder have highlighted a correlation between the expression of CD44 and the differentiation status of the tumor.42,43 However, there still remains an ambiguity with respect to the correlation between CD44 and tumor differentiation with a few studies showing contradictory results.42–44 This could, possibly, be due to the multiple functions attributed to the molecule in CSCs and cell adhesion.45,46 In this and previous study, although an independent correlation with differentiation was not observed with CD44, the expression of the marker exerted a strong influence on prognostic capability of differentiation. However, CD147 expression showed a direct correlation with differentiation and prognosis; similar results have been observed in gastric and colorectal cancers. 47 Elevated expression of CD147 in combination with HAb18G and MMP2 has also been reported to be indicators of poor differentiation in pancreatic and colorectal cancers.48,49 Additionally, in this study, the elevated expression of these markers was also accompanied by de-regulation of the Vimentin, MMPs, and SNAIL in vitro, which is in accordance with evidences that suggest the role of CD147/CD44 in inducing the tumor-associated stroma to secrete MMPs and initiate stromal degradation and EMT.9,26,50 Although the correlation of these markers with metastasis could not be evaluated in this study due to low incidence, the evidences obtained in vitro do indicate an association between CD147/CD44 and the markers of EMT with significant role in modulating properties such as invasion and metastasis.51–53 Given these evidences, this study does suggest a role for CSCs in patterning differentiation in tumors and thereby influencing prognosis, the use of a combination of markers to specify CSCs being more accurate.
Conclusion
In this study, the presence of CSCs, as specified by CD44+/CD147+ combination, correlated with the loss of cell differentiation and poor prognosis in OSCC in both in vitro models and in patients. However, the correlation with prognosis necessitates validation in a larger cohort of patients. These evidences suggest that the tumor differentiation in oral cancer may be driven by CSC expression, the combination of tumor grade with CSC markers being better prognosticators. Further studies that correlate other histological features in a larger cohort of patients as well as those which delineate the underlying mechanisms of CSC-driven tumor histology and thereby prognosis will enable a deeper understanding of the process.
Footnotes
Acknowledgements
The authors thank Ms Priyanka Chevur for providing help in FACS acquisition.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by Narayana Hrudayalaya Medical Ethics Committee (Ref. No. NHMEC-2013/24Oct2013A).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is funded in part by Mazumdar Shaw Medical Foundation and Department of Science and Technology (DST), Government of India. S.M. received funding through Women-Scientist A award (ID: SR/WOS-A/LS-224/2012) by DST, Government of India.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
