Abstract
Purposes
For several years S100A4 has been implicated in tumor progression and prognosis. However, the prognostic value of S100A4 overexpression in patients with gastric cancer remains unknown. Therefore, we performed a meta-analysis to assess the relationship between S100A4 overexpression and clinical outcome of gastric cancer.
Methods and Results
Candidate studies were searched from PubMed, Embase, Cochrane Library, and ISI Web of Science. We included studies that evaluated the prognostic value of S100A4 expression in gastric cancer patients with regard to survival and a series of clinicopathological parameters. The pooled hazard ratios (HR) and odds ratios (OR) with 95% confidence intervals (CI) were used to estimate the effects. Ten studies, all from Asia, were included in the meta-analysis. The pooled analysis showed that S100A4 overexpression was significantly associated with worse overall survival (OS) (HR=1.86, 95% CI: 1.45-2.38, p<0.00001) without heterogeneity in the data (I2=43.6%, p=0.131). Furthermore, our results showed that S100A4 overexpression was significantly correlated with some clinicopathological parameters such as tumor grade, stage, metastasis, invasion, and relapse.
Conclusions
The results of our meta-analysis indicate that S100A4 overexpression correlates with more adverse clinical features and a poor prognosis of gastric cancer patients in Asia, thus suggesting that S100A4 could be a useful marker to evaluate progression and prognosis of Asian gastric cancer patients. More studies from Western countries with a larger number of tumors and standardized methods are required before significant conclusions can be drawn.
Introduction
Gastric cancer is one of the most common malignancies and a frequent cause of cancer-related death worldwide (1). The incidence is particularly high in East Asia and, generally, the prognosis is rather poor, with a 5-year relative survival below 30% in most countries (2). Surgical resection remains the mainstay for the curative treatment of gastric cancer, but many patients eventually die of relapse or distant metastasis after curative resection (3). A major strategy to tackle this healthcare problem is the early detection of this disease (4, 5). Recent advances in molecular oncology may identify additional predictive markers of tumor progression; nevertheless, suitable predictors that can be widely used in clinical settings are not currently available. The development of reliable biomarkers and simple tests that are routinely applicable for early detection, progression, prognosis, and therapy monitoring is strongly needed.
S100A4 (also known as calvasculin) belongs to the S100 family that contains two calcium (Ca2+) binding sites, including a canonical EF-hand structural motif, and has been classified as a metastasis-related gene (6, 7). S100A4 has been found to be both overexpressed and associated with reduced survival in several cancer types, among which breast cancer (8), bladder cancer (9), esophageal squamous cancer (10), lung cancer (11), and colorectal cancer (12). In particular, growing evidence has indicated that S100A4 is overexpressed in gastric cancer (13–24). Multiple studies have shown that gastric cancer patients with strong S100A4 expression had a significantly poor overall survival (OS) or disease free survival (DFS) (13, 20, 21, 23, 24); however, other studies could not confirm this (15, 22). With regard to the associations between S100A4 expression and clinicopathological parameters, such as lymph node metastasis (LNM), tumor grade, and vascular invasion, the studies were generally heterogeneous. Until now, no quantitative systematic review has focused on the relationship between progression and prognosis of gastric cancer and S100A4 overexpression. As mentioned above, there is a need for a comprehensive meta-analysis to derive a more precise estimation.
We herein present a meta-analysis performed to precisely estimate the prognostic value of elevated S100A4 levels for survival among patients with gastric cancer. Additionally, the relation between S100A4 expression and clinicopathological parameters that may be predictors of poor clinical outcome of gastric cancer was also examined.
Methods
Search strategy
Studies were identified by searching the PubMed, Embase, Cochrane Library, and ISI Web of Science databases (last search updated to October 24, 2013). The following search strategy was used: “gastric cancer OR gastric carcinoma OR stomach carcinoma OR stomach neoplasms” AND “S100* OR S100A4” AND “prognos* OR progression OR survival”. No language restrictions were applied. To ensure that no studies were overlooked, the reference lists of relevant articles and review articles were manually searched to identify additional studies.
Selection criteria
All articles retrieved were included or excluded according to specific inclusion/exclusion criteria. The inclusion criteria were: (i) all studies had to be associated with S100A4 expression and gastric cancer; (ii) studies reported explicit methods for the detection of S100A4 expression in gastric cancer; (iii) the endpoints were OS, DFS, or a series of clinicopathological parameters; (iv) studies reported a hazard ratio (HR) and 95% confidence interval (CI) or the data sufficient to estimate the HR and 95% CI from survival analysis; and (v) full text articles. The exclusion criteria were: (i) letters to the editor, reviews, and articles published in a book; (ii) animal and cell-line studies; (iii) articles that did not provide information on survival or clinicopathological parameters; or (iv) the HRs of OS could not be extracted based on the given information. When multiple publications on the same study population were identified or when study populations overlapped, only the most recent or complete article was included in the analysis. Two authors performed the articles' search and identification independently, and the inclusion of an article was decided by consensus.
Data extraction
Two authors independently extracted information using predefined data abstraction forms. Discrepancies were resolved by discussion until reaching a consensus. The following information was extracted from each included trial: first author's name, year of publication, country, study size, mean or median age of patients, type of treatment, tumor characteristics, cutoff value of S100A4 expression, and follow-up time. Outcome measures were also recorded including data allowing us to estimate the impact of S100A4 expression on DFS and OS, and data from categorical variables allowing estimation of the impact of S100A4 expression on clinicopathological parameters.
Statistical methods
The expression of S100A4 was considered high or low according to the cutoff values provided by the authors. HRs and their 95% CIs were combined to measure the effective value. If HRs and corresponding 95% CIs were not available, they were calculated from available numerical data using the methods reported by Parmar et al (25). The data from Kaplan-Meier survival curves were read by Engauge Digitizer version 4.1. Two independent investigators read the curves to reduce the reading variability. For the pooled analysis of the relation between S100A4 overexpression and clinicopathological parameters (such as tumor grade and stage), the original binary variables were recorded and odds ratios (ORs) and their 95% CIs were combined to give the effective value. Statistical heterogeneity was assessed by visual inspection of forest plots, by performing the χ2 test (assessing the p value), and by calculating the I2 statistic (26, 27). If the p value was less than 0.10 and I2 exceeded 50%, indicating the presence of heterogeneity, a random-effects model (the DerSimonian and Laird method) was used (28); otherwise, the fixed-effects model (the Mantel-Haenszel method) was used (29). A sensitivity analysis was conducted to evaluate sources of heterogeneity both in the overall pooled estimate as well as within the subgroups. We assessed publication bias graphically using a funnel plot, and quantitatively using the Begg rank correlation test and the Egger regression asymmetry test (30, 31). If publication bias was observed, we adjusted for the effect by using the Duval and Tweedie trim-and-fill method (32). The impact of S100A4 on prognosis was considered statistically significant if the 95% CI for the overall HR did not overlap 1. Conventionally, an observed HR >1 implied a worse survival for the group with S100A4 expression. All p<0.05 (two-sided) were considered as significant unless otherwise specified. All analyses were performed using STATA, version 12.0 (StataCorp, College Station, TX, USA).
Results
Study characteristics
The process of article identification, inclusion, and exclusion is summarized in Figure 1. The initial search yielded 317 records that studied the relationship between S100A4 expression and both gastric cancer prognosis and clinicopathological parameters. After removal of duplicates and screening of the titles and abstracts, 302 trials were excluded, leaving 15 studies for full publication review. Of these, 4 articles were excluded: 3 studies revealed no relation (33–35), and the full-text of one could not be obtained (14). Therefore, according to the inclusion and exclusion criteria, 11 studies were finally retrieved for the meta-analysis (13, 15–24). All studies were published between 2000 and 2013; 10 studies were from Asia and 1 from Europe. The sample size varied from 74 to 598 patients, accounting for a total of 2,149 patients. Gastrectomy as initial treatment for gastric cancer was reported in 9 studies (15–18, 20–24) and gastrectomy plus chemotherapy in 2 (13, 19). Immunohistochemistry was used to detect the S100A4 expression in all but 1 study, and the threshold for differentiating the level of S100A4 varied widely among studies. The prevalence of high-level S100A4 ranged from 12.3% to 68.8% of the studied population. Table I summarizes the characteristics of the included studies.
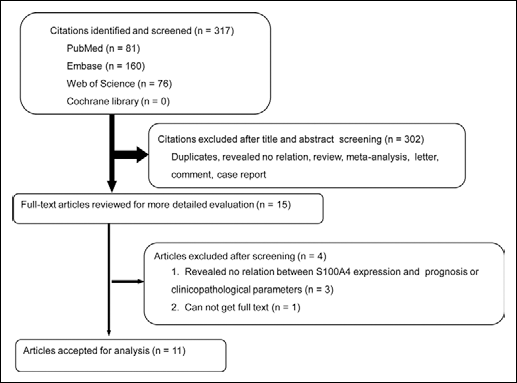
Paper identification, exclusion, and inclusion.
Characteristics of the Studies Investigating the Prognostic Values of S100A4 in Gastric Cancer Patients Included for Meta-Analysis
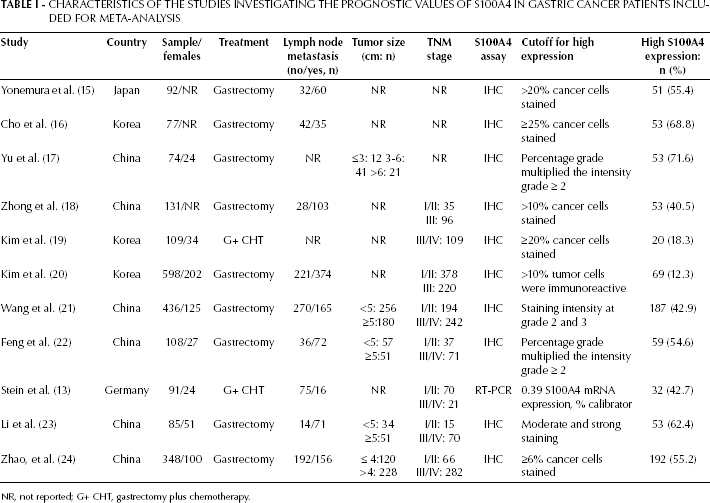
NR, not reported; G+ CHT, gastrectomy plus chemotherapy.
S100A4 expression and OS in gastric cancer
Overall, 5 studies (15, 20–22, 24) including 1,582 patients reported data on S100A4 expression and OS in gastric cancer (Tab. II). All enrolled patients underwent gastrectomy. In all but 1 study (24), the HRs were calculated. As shown in Figure 2, high S100A4 expression was statistically significantly associated with poor OS in 3 studies (20, 21, 24). Combined data from all 5 studies showed that increased S100A4 levels were significantly correlated with OS with a pooled HR estimate of 1.86 (95% CI: 1.45-2.38, p<0.00001; Fig. 2), without heterogeneity in the data (I2=43.6%, p=0.131). After subgroup analysis based on the method used to obtain the HR, the level of significance remained practically unchanged.
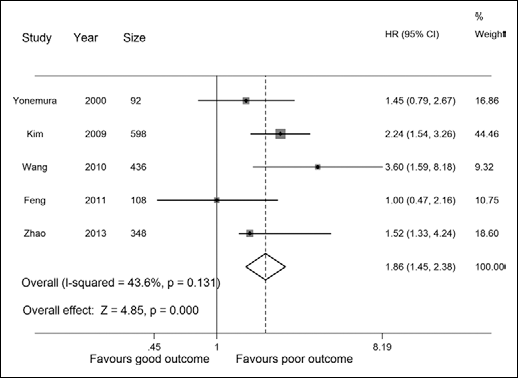
Forest plot of the association between S100A4 overexpression and overall survival (OS) of gastric cancer patients.
The Association of S100A4 Expression with Survival and Clinicopathological Parameters in Gastric Cancer Patients from the Available Published Studies
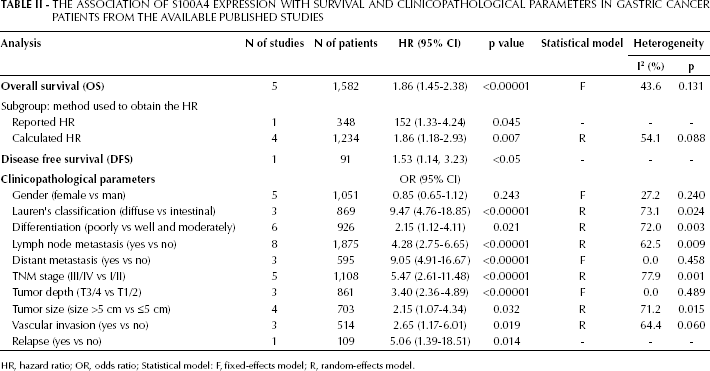
HR, hazard ratio; OR, odds ratio; Statistical model: F, fixed-effects model; R, random-effects model.
S100A4 expression and DFS in gastric cancer
Only 1 study including 91 patients reported data on S100A4 expression and DFS in gastric cancer (13) (Tab. II). In this study, gastric cancer patients received surgery or adjuvant therapy, and S100A4 expression was detected by RT-PCR. With 712 days of median follow-up, an unfavorable DFS was observed for S100A4 high-expressing gastric cancer patients. The calculated HR was 1.53 (95% CI: 1.14-3.23).
S100A4 expression and clinicopathological parameters
All studies reported data on S100A4 expression and at least 1 clinicopathological parameter in gastric cancer (Tab. II). More specifically: 5 studies reported data on gender (17, 21–24); 3 reported data on Lauren's classification (21, 23, 24); 6 reported data on differentiation (15, 17, 18, 21–23); 8 reported data on LNM (15, 16, 18, 20–24); 3 reported data on distant metastasis (17, 21, 23); 5 reported data on TNM stage (18, 21–24); 3 reported data on tumor depth (16, 21, 24); 4 reported data on tumor size (17, 21–23); 3 reported data on vascular invasion (15, 17, 24); and 1 study reported data on relapse and its relationship with S100A4 expression (19). When the data were respectively pooled, we found significant associations between high S100A4 expression and all clinicopathological parameters except gender. Specifically, the pooled ORs (and 95% CIs) were as follows: 9.47 (4.76-18.85) for Lauren's classification (diffuse type vs intestinal type); 2.15 (1.12-4.11) for differentiation (poor differentiation vs well and moderately differentiated); 4.28 (2.75-6.65) for LNM (with LNM vs without LNM); 9.05 (4.91-16.67) for distant metastasis (with distant metastasis vs without distant metastasis); 5.47 (2.61-11.48) for TNM stage (stage III/IV vs stage I/II); 3.40 (2.36-4.89) for tumor depth (T3/4 vs T1/2); 2.15 (1.07-4.34) for tumor size (size >5 cm vs size ≤5 cm); 2.65 (1.17-6.01) for vascular invasion (with vascular invasion vs without vascular invasion); and 5.06 (1.39-18.51) for relapse (with relapse vs without relapse) (Supporting Information Fig. 1). However, there were some limitations to this analysis; data from the study by Yu et al (17) concerning tumor size indicated that a size >6 cm vs a size ≤6 cm had higher S100A4 expression, a comparison which is not consistent with the other studies. However, excluding this study did not significantly affect the overall result. The results from the random-effects model correlated with those from the fixed-effects model.
Sensitivity analysis
Sensitivity analyses were performed to examine whether the effect estimate was robust. A single study included in the meta-analysis was deleted each time to reflect the influence of the individual data set to the pooled HRs (or ORs); the corresponding pooled HRs (or ORs) were not significantly altered (data not shown).
Publication bias
Begg's funnel plot and Egger's test were performed to assess the studies' publication bias. The shape of the funnel plot did not reveal any clear asymmetry (on the S100A4 expression and OS analysis) (Fig. 3). Egger's test was then used to provide statistical evidence of funnel plot symmetry; similarly, the results did not suggest any publication bias (p=0.638), indicating that the results of these meta-analyses were relatively stable and that publication bias is unlikely to have affected them.
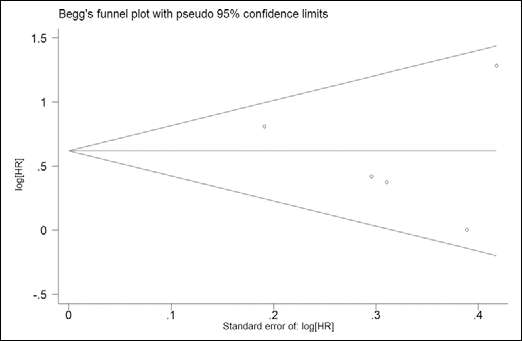
Funnel plots of publication bias for overall survival (OS) analysis.
Discussion
The factors that influence the prognosis of gastric cancer patients are not completely understood. Nowadays, surgical resection remains the mainstay for the curative treatment of gastric cancer; however, not all patients derive clinical benefit from such treatment (3). Tumor metastasis and recurrence often contribute to the poor clinical outcome of gastric cancer patients, whereas invasion is a key step that results in gastric cancer metastasis and recurrence (36). Nevertheless, the molecular factors associated with tumor progression and invasion of gastric cancer have not been well characterized. A candidate marker for the progression and prognosis of cancers is S100A4. Here, we examined the effect of S100A4 in the progression and prognosis of gastric cancer.
The present study showed that S100A4 overexpression significantly predicted poor OS for gastric cancer patients treated by gastrectomy, with no significant heterogeneity and no publication bias. Therefore, S100A4 expression might be a useful prognostic marker for individual gastric cancer patients.
Although the results of this analysis were positive, the discrepancies in OS among gastric cancer patients with high and low S100A4 expression were still not completely elucidated. Notably, tumor invasion and metastasis were usually correlated with poor prognosis of gastric cancer. It is well known that S100A4 is overexpressed in many tumors and is involved in tumor metastasis (6, 7), but the mechanisms of the metastasis-promoting function of S100A4 are not fully understood. It has previously been demonstrated that transfection of rodent or human S100A4 into non-metastatic mammary tumor cells resulted in the acquisition of metastatic properties when re-introduced into rodents (37); this supported the suggestion of a direct role of S100A4 in the promotion of metastasis.
In view of the association of tumor aggression with gastric cancer outcome, we also conducted a pooled analysis on the correlation between S100A4 expression and gastric cancer patients' clinicopathological parameters. As expected, the results suggested an evident trend towards higher S100A4 expression with the presence of LNM, distant metastasis, vascular invasion, and relapse. Besides, the results also showed that a high S100A4 expression was significantly associated with advanced tumor TNM stage, tumor depth, and tumor size. Taken together, the pooled results of the present study support the hypothesis that S100A4 overexpression might promote gastric cancer invasion and metastasis through direct or indirect mechanisms, and thus lead to the poor prognosis of gastric cancer patients.
The identification of reliable molecular prognostic markers for tumor aggression and survival is of great significance in gastric cancer; such markers would help guide clinical decision-making with regard to cancer treatment and outcomes. To our knowledge, this is the first meta-analysis on the association between S100A4 expression and survival and clinicopathological parameters in gastric cancer. Elevated expression of S100A4 may play a role in the progression and prognosis of gastric cancer; thus, S100A4 may serve as a biomarker for progression of gastric cancer and a potential molecular target for biotherapy.
Nevertheless, some limitations of this meta-analysis should be acknowledged. First, the studies included in the meta-analysis were mainly retrospective; it is thus possible that other unknown confounders may bias the data. Second, the low number of articles available and their quality pose a major limitation: despite searching the most pertinent articles in the PubMed, Embase, Cochrane Library, and ISI Web of Science databases, the number of eligible studies in this meta-analysis is still limited. Third, differences in immunohistochemical staining protocols, primary antibodies, evaluation standards, and cutoff values for high S100A4 expression may also contribute to heterogeneity. The different cutoff values between studies may affect the results and account for the inconsistencies. Therefore, further multicenter studies using standardized methods are encouraged. Fifth, when we evaluated the correlations between S100A4 expression and tumor invasion and grade, the between-study heterogeneity was significant. Generally, meta-regression analysis should be conducted to identify variables that result in heterogeneity. In this study, however, meta-regression could not be performed owing to the small number of studies included. Finally and importantly, all but one of the selected studies were from Asia, which puts forward a question concerning the external validity of results and applicability to patients from Western countries. Given the abovementioned limitations, our results should be interpreted with caution and, likewise, the conclusions of this meta-analysis should be carefully drawn.
In conclusion, our analysis showed that gastric cancer patients who overexpressed S100A4 had more adverse clinical features and a poorer prognosis. S100A4 expression provided essential information for the prediction of gastric cancer invasion, metastasis, and prognosis, suggesting that S100A4 could be a useful marker in evaluating the prognosis of gastric cancer patients. These results were mostly limited to the Asian population; more studies from Western countries with a larger number of tumors and standardized methods are required before significant conclusions can be drawn.
