Abstract
Cyclin E is a critical regulator in cell cycle and promotes the initiation of DNA replication and centrosome duplication in late G1. The overexpression of cyclin E is common in cancers of the digestive system. However, whether cyclin E represents a prognostic biomarker in gastrointestinal cancer remains controversial. We reviewed the published literatures to clarify the association between cyclin E determined by immunohistochemistry (IHC) and survival in gastrointestinal cancer. Literatures were searched in PubMed and Cochrane Library published up to December 1, 2014. A total of 282 articles were initially identified, and 14 articles were included in this study. Meta-analysis was performed for 10 studies with a total of 1300 patients. Combined hazard risk (HR) and corresponding 95% confidence interval (CI) were calculated by random-effect model due to the heterogeneity. The quality of included studies was assessed by the Newcastle-Ottawa Scale and the Methodological Index for Non-Randomized Studies (MINORS). We found that high level of cyclin E was a predicator of poor prognosis among patients with gastrointestinal cancer (HR = 1.67, 95% CI = 1.06-2.63,
Introduction
Progression between phases in cell cycle (G0, G1, S, G2, M) is an orderly process tightly regulated by complex yet redundant mechanisms. It includes multiple checkpoints used to assess growth signals, cell size, and DNA integrity. 1 Cyclins are prime cell cycle regulators, and they play a critical role in the control of cell proliferation by forming a complex with different cyclin-dependent kinases (CDKs). Cyclin D-CDK4, cyclin D-CDK6, and cyclin E-CDK2 drive G1 progression through the restriction point, which commits the cell to complete the cycle. 2 Abnormalities in cell cycle regulators and subsequent deregulation of the G1/S transition may be one of the important biologic events in the development and progression of cancer. Overexpression of cyclins and CDKs and loss of expression of CKIs (CDK inhibitors) are frequently reported in human neoplasia. The overexpression of cyclin E is common in various types of cancer, including breast cancer, 3,4 nonsmall cell lung cancer (NSCLC), 5,6 colon cancer, 7 bladder cancer, 8 and ovarian cancer. 9
Cancers of the digestive system are common worldwide, while the pattern and incidence vary widely between different parts of the world. According to the data of World Health Organization, in 2005 to 2009, the highest overall rates (per 100 000) among men were in the Republic of Korea (25.3). In women, the highest rates (per 100 000) were observed in Ecuador (10.5). 10 It is estimated that there are more than 1.2 million men and women living in the United States with a previous colorectal cancer diagnosis up to January 1, 2014, and the 1-year and 5-year relative survival rates for patients with colorectal cancer are 83.4% and 64.9%, respectively. 11 Mortality rates for both gastric and colorectal cancer decline in the past decade attributed to the prevention, early detection, and treatment, 12 but gastric cancer and colorectal cancer still rank the second and fourth leading cause of cancer deaths worldwide. 13,14
A number of studies have investigated the association between expression of cyclin E and survival in patients with gastric and colorectal cancer. However, whether cyclin E represents a prognostic biomarker remains controversial and no consensus has been reached. Tenderenda 15 reported that increased expression of cyclin E in gastric cancer tissue correlated with poorer prognosis. In contrast, Takano et al 16 and Li et al 17 reported independently that patients with gastric cancer and colorectal cancer whose tumors were cyclin E positive showed a favorable prognosis. However, So et al 18 and Ioachim 19 reported that expression of cyclin E was not related to survival of patients with gastric or colorectal cancer. Hence, we performed a systematic review of published studies with meta-analysis to assess the effects of overexpression of cyclin E in tumor tissue on survival in patients with gastrointestinal cancer.
Methods
Search Strategy and Study Selection
The electronic databases PubMed and Cochrane Library were searched for studies dealing with the prognostic implications of different expression levels of cyclin E protein to include in the present meta-analysis. The references reported in the identified studies and conference papers were also used to complete the search. The deadline of the included articles was December 1, 2014. Searches included the terms “gastric,” “intestinal or colorectal,” “cancer or carcinoma or tumor,” and “Cyclin E.”
Studies were considered eligible if they met the following criteria: (1) peer-reviewed original articles, (2) patients with gastric and intestinal cancer were studied, (3) the expression of cyclin E in cancer tissues was measured with immunohistochemistry (IHC), and (4) the primary outcomes of interest were overall survival (OS) and/or relapse-free survival (RFS), hazard ratios (HRs), and 95% confidence interval (95% CI) according to cyclin E level either had to be reported or had to be calculated from the data presented. The exclusion criteria for the studies were (1) HRs of OS or RFS could not be extracted based on the given information and (2) animal or laboratory studies, non-English articles, reviews or letters, case reports, articles published in a book.
The titles and abstracts of relevant articles were read by 2 reviewers (Lanshan Huang and Fanghui Ren), respectively. The articles which could not be categorized in accordance with title and abstract were retrieved for full-text review. Disagreements were resolved through consensus with a third reviewer (Gang Chen).
Data Extraction
The final articles included were assessed independently by 2 reviewers (Lanshan Huang and Fanghui Ren) using a standard protocol. The following characteristics were extracted: first author, publication year, origin of population, type of cancer, tumor stage, test method, cutoff value, follow-up, and survival data. Differences in the data extraction were resolved together by 2 reviewers.
Statistical Analysis
Included studies were tested by performing chi-square Q-tests for heterogeneity. A
Hazard ratio with 95% CI was used to combine the data. When described in original articles, these values were obtained directly. When these statistical variables were not provided, they were calculated from available numerical data or Kaplan-Meier survival curve. The total values were combined to obtain the overall HR and 95% CI 21 consisted of a DerSimonian-Laird random-effect model.
Sensitivity analyses were performed by removing each study in turn to establish the extent to which they contributed to heterogeneity and to the overall result. Studies with clear survival values provided were included in a sensitivity analysis, excluding the studies in which data could only be obtained from calculation or Kaplan-Meier survival curves.
The quality of included studies was assessed using the Newcastle-Ottawa Scale (NOS) for cohort studies. 22 The risk of bias for included studies was assessed using the NOS and the Methodological Index for Non-Randomized Studies (MINORS). The potential publication bias was assessed using Beggs 23 and Eggers tests. 24 The statistical analyses were performed by Stata version 11.0 and Rev Man 5.3. Again, disagreements were resolved by discussion of 2 reviewers (Lanshan Huang and Fanghui Ren) and consensus with a third reviewer (Gang Chen).
Results
Study Characteristics
A total of 282 potentially relevant articles were found after initial searching of PubMed and Cochrane Library. Finally, the remaining 15 potential eligible studies were obtained for full-text evaluation. 15 -19,25 -34 One study 25 was excluded due to the duplicate for the same population. Fourteen studies 15 -19,26 -34 were included in the systematic review, of which 10 studies 15 -17,26 -29,31,33,34 with sufficient data or with data that could be calculated indirectly were evaluated in the meta-analysis and 4 studies 18,19,30,32 were with lack of eligible HR data. Figure 1 showed the flowchart of study inclusion. The main characteristics of the included studies are summarized in Table 1.
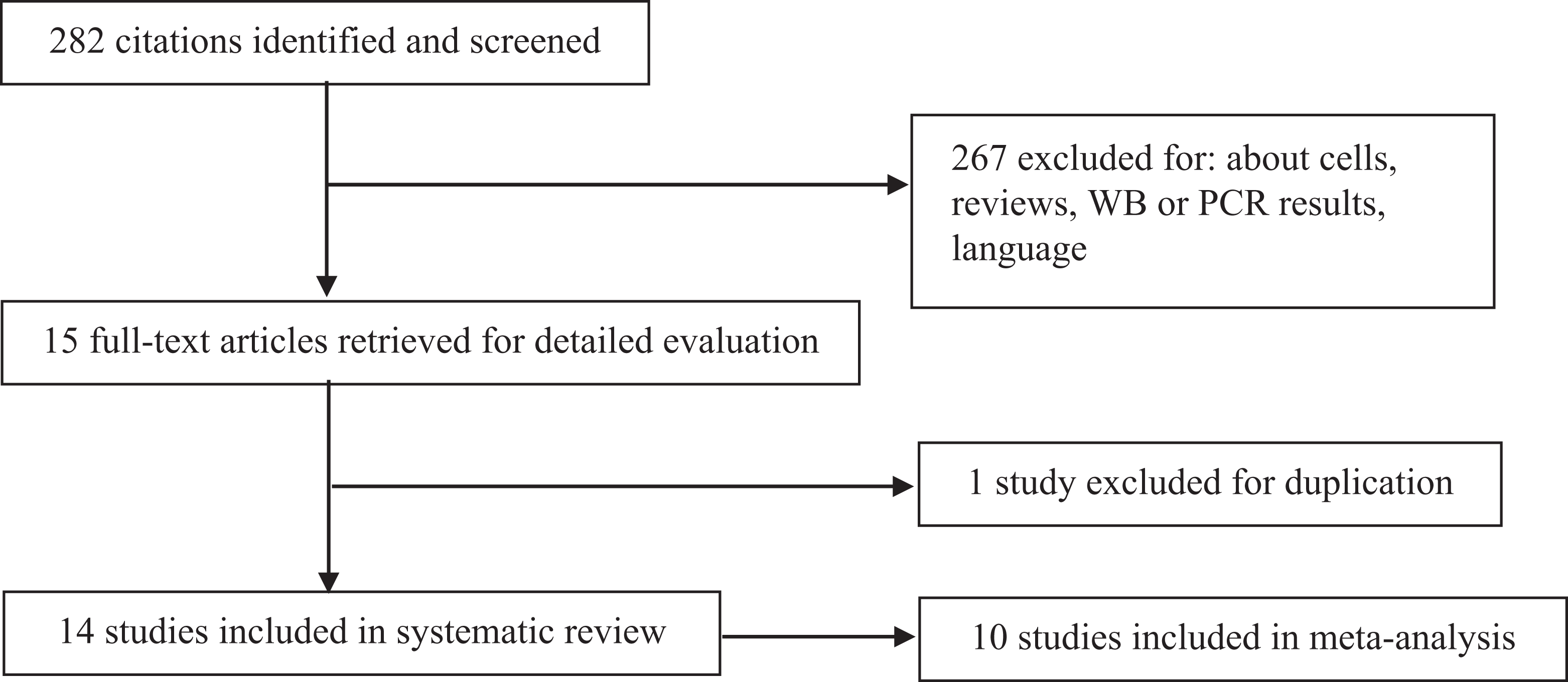
Study inclusion flowchart.
Characteristics of Patients in Different Studies.

Abbreviations: NA, not available; A/B/C/D Dukes classification; I/II/III/IV TNM stage; IHC, immunohistochemistry; OS, overall survival; DFS, disease-free survival; RFS, recurrence-free survival; HR hazard ratio; SC survival curve; CI, confidence interval.
aMedian follow-up.
bTotal follow-up.
cMean follow-up.
dThe 4 studies lack of eligible HR data.
In the meta-analysis, we collected data from the 10 studies between 1999 and 2009 including a total of 1300 patients from Japan, Korea, Germany, and Poland. All of them were retrospective in design. All of the studies reported the prognostic value of cyclin E status for survival in patients with gastric cancer or colorectal carcinomas. Immunohistochemistry was applied for cyclin E expression assessment in tissue samples in all the studies included. Of the 10 studies, 4 studies directly reported HRs while the other 6 studies provided survival curves. Seven studies identified overexpression of cyclin E as an indicator of poor prognosis and the other 3 studies showed inverse results of which 2 studies showed no statistically significant impact of overexpression of cyclin E on prognosis.
Since the HRs of OS could not be obtained based on the data presented in the articles of So et al, Bondi et al, and Ioachim 18,19,32 and HR with 95% CI calculated from Kaplan-Meier survival curve in Lim article 30 was not eligible which may be due to the small number of the study and low-positive rate, these articles were excluded in the meta-analysis and remained in the systematic review.
Meta-Analysis and Sensitivity Analysis
The 10 studies including 1300 cases were evaluated for the effect of overexpression of cyclin E on OS. A random-effect model was used to combine HRs because of the heterogeneity present among the studies by chi-square Q-tests (χ2 = 46.73,

Forest plot of meta-analysis of the effect of expression of cyclin E on survival in patients with gastrointestinal cancer. Random effects model was used when combining data. The combined hazards ratio (HRs) displayed in this figure suggest that high level of expression of cyclin E might be an independent prognostic factor that predicts the adverse outcome in patients with gastrointestinal cancer.
Sensitivity analyses showed that the study by Tenderenda,
15
Bani-Hani et al,
31
and Li et al
17
were the top 3 with heterogeneity, and removal of these studies changed the effect into a significant one with lowest heterogeneity (HR 1.365, 95% CI 1.03-1.81,
Quality and Bias Assessment
The quality assessment for a total of 14 studies are summarized in Table A1 of the appendix. For quality, scores ranged from 0 (lowest) to 9 (highest), and studies with scores of 6 or more were rated as high quality. Scores of the 14 studies ranged from 4 to 7, with a mean of 6.21 ± 1.19. The risk of bias was shown in Figure A1 of the appendix. Baseline equivalence of groups and incomplete outcome data were considered high risk of bias in the 14 studies which indicated information bias and attrition bias.
The Begg funnel plot and the Egger test were applied for assessing publication bias in the meta-analysis. Begg funnel plot revealed evidence of asymmetry in this study (Figure 3). However, the

Begg’s funnel plot for the assessment of potential publication bias. Studies are distributed asymmetrically and suggest the potential meta-analysis publication bias.
Systematic Review
As the detailed results of the 4 studies excluded from meta-analysis were not shown in Table 1, we summarize the information here. In the study of So et al, 18 expression of cyclin E was not related to survival of patients with gastric cancer. In the studies of Lim et al, 30 Bondi et al, 32 and Ioachim, 19 the correlation of cyclin E with prognosis of patients with colorectal carcinoma was reported. According to Lim et al, 30 in patients with stage II colorectal cancer, the overexpression of cyclin E could not predict poor survival. According to Bondi et al, 32 when analyzing the expression of the different cyclin proteins individually (cyclins A, B1, C, D1, D3, E, and H) with regard to survival, only low expression of cyclin A showed a significant association with an unfavorable prognosis. Ioachim 19 reported that the prognostic significance of cyclin E in determining the risk of recurrence and OS showed no statistically significant differences.
In total, among the 14 studies included, 7 studies identified overexpression of cyclin E as an indicator of poor prognosis, 3 studies showed inverse results, and 4 studies showed no association of overexpression of cyclin E with prognosis. Of 14 studies, there were 10 on gastric cancer and 4 on colorectal carcinoma. In the 10 studies on the association of cyclin E with gastric cancer prognosis, 7 studies identified cyclin E as a poor prognosis marker, 2 studies showed inverse results, and 1 study found no relation. In the 4 studies on colorectal carcinoma, 1 study showed that low level of cyclin E associated with poor survival and 3 studies found no correlation between cyclin E and survival.
Discussion
In the previous published literatures, several systematic reviews with meta-analyses on overexpression of cyclin E for tumors have been performed; high level of cyclin E was found to be a poor prognostic factor for survival in breast cancer and NSCLC. 35 -37 As far as we know, no meta-analysis had been undertaken to evaluate cyclin E as a prognostic marker in case of digestive system. In the present meta-analysis, we identified 10 studies concerning the expression of cyclin E on survival of gastrointestinal cancer. The expression of cyclin E was determined by IHC with surgical specimens. Our results showed that overexpression of cyclin E was associated with poor survival for patients with gastrointestinal cancer.
Cyclins E and A and the activator of the cyclin E(A)/CDK2 kinase—the CDC25A phosphatase—are induced in late G1. 38 Cyclin E accumulates at G1/S boundary and forms complex with CDK2 and subsequently promotes initiation of DNA replication and centrosome duplication. The abundant cyclin E eventually becomes phosphorylated and degraded that allows normal cell cycle progression. 39,40 However, the level of cyclin E and activity of cyclin E–CDK2 can be aberrantly regulated and this excessive activity of the cyclinE–CDK2 complex, in turn, drives cells to replicate their DNA prematurely, resulting in tumorigenesis. 41
Alterations of cyclin E have been shown in gastrointestinal cancer including amplification of cyclin E gene and overexpression of cyclin E protein. 4,8 Like other regulators of cell cycle such as cyclin D1, 42,43 the association of cyclin E with prognosis of these cancers also cannot reach a consensus hither to. Inconsistent results were shown in previous studies that were performed with methods of polymerase chain reaction (PCR), Western blot (WB), or IHC. Expression of Cyclin E is easy to be detected in human tissues by standard IHC techniques and IHC is cost effective and relatively easy to perform. In this study, we assessed the prognostic role of cyclin E protein in patients with gastrointestinal cancer. Evaluating the prognosis of patients with gastrointestinal cancer by detecting cyclin E level with IHC techniques might be an alternative scheme.
As a meta-analysis, this study was restricted to some limitations that should be discussed for further consideration. First, there might be biases if negative studies were less frequently published or contained less detailed results. Second, we conducted a literature search in PubMed and Cochrane Library. As studies included were published in English only, selection and language bias might exist. Third, due to the limited published studies on association of cyclin E protein with prognosis of colorectal carcinoma and lack of subgroup meta-analysis, this meta-analysis cannot offer strong evidence on the overall of gastrointestinal cancer. Fourth, extrapolating the survival rates from the survival curves may add inaccuracy in the extracted survival rates and lead to potential bias. Finally, the cutoff used to assess overexpression of cyclin E, the technique used for IHC, and the analysis of univariate or multivariate were variable between studies might contribute to the heterogeneity. Therefore, our results should be interpreted with caution.
In conclusion, despite the limitations described earlier, this study suggests that overexpression of cyclin E is associated with poor prognosis in gastrointestinal cancer. The expression of cyclin E determined by IHC might be a prognostic marker for gastrointestinal cancer in clinical practice. To strengthen our findings, well-designed prospective studies with better standardized assessment of prognostic markers are necessary to help to define the precise prognostic significance of cyclin E in patients with gastrointestinal cancer.
Footnotes
Abbreviations
Appendix A
The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of 14 studies.

| Studies | Selection | Comparability | Outcome | ||||||
|---|---|---|---|---|---|---|---|---|---|
| First author, year | Representativeness of the Exposed Cohort | Selection of the Nonexposed Cohort | Ascertainment of Exposure | Demonstration that Outcome of Interest was not Present at Start of Study | Comparability of Cohorts on the Basis of Metastasis or Age, Stage, Grade, etc | Assessment of Outcome (Death or Recurrence) | Adequacy of Follow-up Length for Outcome (2 years) | Lost to Follow-up Acceptable (less than 25% or reported) | Total Quality Score |
| Jang et al, 26 1999 |
|
|
|
|
|
|
|
|
|
| Muller et al, 28 1999 |
|
|
|
|
|
|
|
|
|
| Kwon, 27 1999 |
|
|
|
|
|
|
|
|
|
| Aoyagi et al, 29 2000 |
|
|
|
|
|
|
|
|
|
| Takano et al, 16 2000 |
|
|
|
|
|
|
|
|
|
| Li et al, 17 2001 |
|
|
|
|
|
|
|
|
|
| Bani-Hani et al 31 , 2005 |
|
|
|
|
|
|
|
||
| Tenderenda, 15 2005 |
|
|
|
|
|
||||
| Choi et al, 33 2009 |
|
|
|
|
|
|
|
|
|
| Kouraklis et al, 34 2009 |
|
|
|
|
|
|
|
|
|
| Lim, 30 2004* |
|
|
|
|
|
|
|
|
|
| So et al, 18 2000* |
|
|
|
|
|
||||
| Bondi et al, 32 2005* |
|
|
|
|
|
|
|||
| Ioachim, 19 2008* |
|
|
|
|
|
|
|||
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
