Abstract
Background
The relationship between tumor necrosis factor (TNF)-related parameters and cardiorenal metabolic factors is still controversial in clinical hypertension.
Methods
Normotensive men (NT, n=60) and treated stage 2 and 3 essential hypertensive men (HT, n=89) were enrolled in this study. The relationship between TNF-related parameters and cardiorenal metabolic factors was examined in NT and HT, separately.
Results
HT showed higher rates of insulin resistance and enhanced chronic inflammation compared with NT. The levels of soluble TNF receptor 1 and 2 were significantly higher in HT than in NT, although TNF-α levels were unexpectedly lower in HT than in NT. Regression analysis indicated that the TNF-related parameters were closely linked with mild renal dysfunction both in NT and HT, and moderately related to chronic inflammation only in HT. HT taking inhibitors of the renin-angiotensin system showed improved insulin resistance, but no difference in the TNF-related parameters.
Conclusion
These results suggest that the disturbed TNF system is closely linked with chronic inflammation rather than with insulin resistance in HT.
Keywords
Introduction
The tumor necrosis factor (TNF)-α system is well known to participate in insulin resistance associated with essential hypertension (1–3). Chronic inflammation of the cardiovascular system may precede the development of hypertension and atherosclerosis (4, 5). Enhanced TNF activity is associated with chronic inflammation in hypertension (6–10). An activated TNF-α system is associated with early target organ damage, such as urinary microalbuminuria and left ventricular hypertrophy (7, 11). Chronic inflammation and endothelial damage are associated with renal dysfunction in essential hypertension (12–15). The relationships between plasma TNF-related parameters, such as TNF-α and soluble TNF receptor 1 (sTN-FR1) and 2 (sTNFR2), and cardiometabolic factors have been investigated together in normotensive subjects and hypertensive patients. However, it remains unclear whether TNF-related parameters differentially correlate with cardiorenal metabolic factors in normotensive subjects and in hypertensive patients.
TNF-α acts through the binding of 2 types of TNF receptors: TNFR1 and TNFR2. TNFR1 was reported to control common effects of TNF-α, whereas TNFR2 mainly controlled its metabolic effects (1). Increased circulating sTNFR2 but not sTNFR1 or TNF-α concentrations were reportedly associated with insulin resistance in healthy volunteers, lean non-diabetic offspring of diabetic subjects, and young obese subjects (1, 16). In contrast, some studies reported that sTNFR1 reflects the activity of the TNF-α network in lean subjects with insulin resistance (17, 18). The importance of the TNF-related parameters on the cardiorenal metabolic factors depends on demographic characteristics, including obesity of the subjects. Antihypertensive medications have differential impacts on insulin resistance (19). However, it has not been fully elucidated whether they have any effect on the TNF system in relation to their differential impacts on insulin resistance (20).
The aim of this cross-sectional study was to elucidate whether altered TNF-related parameters are associated with the condition of hypertension in hypertensive men (HT) compared with normotensive men (NT). Additionally, we aimed at identifying which TNF-related parameters are correlated with cardiorenal metabolic factors in normotensive subjects and in hypertensive patients. The results of this study may lead to a new insight in the field, by considering differences in the relationships between TNF-related parameters and cardiorenal metabolic factors in normotensive subjects and hypertensive patients separately. The clinical impact of antihypertensive medications on the TNF system may be elucidated in relation to insulin resistance in treated hypertensive patients.
Materials and Methods
Study Population
Apparent healthy volunteers were consecutively informed about the study when undergoing an annual health examination between 2000 and 2004 at Keio University (Tokyo, Japan). NT were enrolled when meeting the following criteria: systolic blood pressure (SBP) <130 mm Hg and diastolic blood pressure (DBP) <80 mm Hg; age >50 years; and no familial history of hypertension among their first-degree relatives.
Essential HT were consecutively recruited during the same period among patients at the same University Hospital. Recruitment was limited to patients who met the following criteria: pretreatment SBP ≥160 mm Hg and/or DBP ≥100 mm Hg; at least one hypertensive parent; and onset of hypertension at <60 years of age, regardless of their age at the time of inclusion. By excluding subjects with high normal blood pressure ranges and patients with stage 1 hypertension, we intended to avoid misclassification of the subjects due to blood pressure variability. We also excluded subjects and patients with clinically evident acute or chronic inflammation (defined on the basis of current and past medical history), as well as those with diabetes, serum creatinine levels ≥1.3 mg/dL, heart or endocrinological disease, or secondary hypertension. Diagnosis of essential hypertension was made based on current and past medical history, family history, physical examinations, electrocardiography, routine urine and blood examinations, plasma renin activity and aldosterone concentrations, and, if necessary, the optional radiological, echocardiographic, and endocrinological examinations. All HT used antihypertensive medications and approximately half of the HT used a combination of antihypertensive medications. Overall, 60 HT were using angiotensin-converting enzyme inhibitors (ACEI) or angiotensin receptor blockers (ARB), 59 calcium channel blockers (CCB), and 12 thiazide diuretics. The present study was conducted according to the principles expressed in the Declaration of Helsinki. The ethics review committee of the School of Medicine at the Keio University approved the study protocol. Each subject gave written informed consent before enrollment (9).
Plasma Assays
Measurements of TNF-α, sTNFR1, sTNFR2, high sensitivity C-reactive protein (CRP), and all other biochemical examinations were performed within a few weeks after sampling, as previously described (9, 10). Blood samples were kept at –80°C before measurements. The insulin resistance index, based on a homeostasis model assessment (HOMA-R), was calculated as fasting blood glucose (FBG) (mg/dL) × immunoreactive insulin (IRI) (μU/mL) / 405. Renal function was assessed using the estimated glomerular filtration rate (eGFR), calculated by modification of the diet in the renal disease equation. The Japanese ethnic factor is 0.881. Therefore, eGFR (mL/min per 1.73 m2) = 0.881 × 186.3 × age-0.203 × creatinine (Cr) (mg/dL)-1.154. Chronic kidney disease (CKD) was defined as positive proteinuria or eGFR <60 mL/min/1.73 m2 body surface area. Clinical stages of CKD were classified according to the recommendations of the National Kidney Foundation Kidney Disease Outcomes Quality Initiative guidelines (21).
Statistical Analysis
Many biomarkers showed a skewed deviation. These included IRI, HOMA-R, low-density lipoprotein-cholesterol (LDL-C), triglycerides (TG), high-density lipoprotein-cholesterol (HDL-C), CRP, TNF-α, sTNFR1, sTNFR2 and Cr levels, which were natural log-transformed. Data with a normal distribution are shown as means ± standard deviations, and biomarkers with a skewed deviation are shown as 25th percentile – median – 75th percentile. Comparisons between the groups were carried out with the unpaired t-test. Chi-squared analysis and Fischer's exact test were used to estimate differences in the distributions according to the CKD classification. Receiver operating characteristics (ROC) analysis was performed for natural log-transformed values of HOMA-R, TNF-α, sTNFR1, sTNFR2, CRP, and LDL-C, and compared between NT and HT. Univariate correlations between demographic, TNF-related and cardiorenal metabolic factors were evaluated with Pearson's correlation coefficients in NT and HT, separately. Stepwise multivariate regression analysis was performed to identify significant contributions of TNF-α, sTNFR1, and sTNFR2 in NT and HT, separately. Statistical significance was defined as p<0.05. Statistical analyses were performed using the SPSS statistical program (SPSS V17, Chicago, IL, US).
Results
Comparisons of Clinical Parameters between NT and HT
Clinical characteristics of NT and HT are summarized in Table I. Body mass index (BMI), FBG, IRI, TG, uric acid (UA) levels, and HOMA-R were significantly higher in HT than in NT, whereas HbA1c and LDL-C were similar in the 2 groups. HDL-C levels were significantly lower in HT than in NT. Altogether, these results indicate that HT had insulin resistance. WBC and CRP levels were significantly higher in HT than in NT suggesting an association between HT and chronic inflammation. sTNFR1 and sTNFR2 levels were significantly higher in HT than in NT, although plasma TNF-α concentrations were unexpectedly lower in HT than in NT. These results indicate that a disturbed TNF system is associated with the condition of HT. Due to the higher prevalence of proteinuria in HT, the proportion of subjects with CKD was higher in HT than in NT.
Clinical Characteristics of Normotensive Subjects and Hypertensive Patients
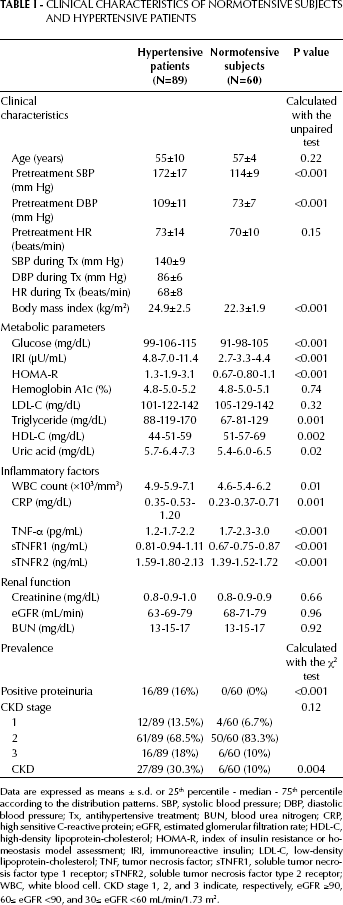
Data are expressed as means ± s.d. or 25th percentile - median - 75th percentile according to the distribution patterns. SBP, systolic blood pressure; DBP, diastolic blood pressure; Tx, antihypertensive treatment; BUN, blood urea nitrogen; CRP, high sensitive C-reactive protein; eGFR, estimated glomerular filtration rate; HDL-C, high-density lipoprotein-cholesterol; HOMA-R, index of insulin resistance or homeostasis model assessment; IRI, immunoreactive insulin; LDL-C, low-density lipoprotein-cholesterol; TNF, tumor necrosis factor; sTNFR1, soluble tumor necrosis factor type 1 receptor; sTNFR2, soluble tumor necrosis factor type 2 receptor; WBC, white blood cell. CKD stage 1, 2, and 3 indicate, respectively, eGFR ≥90, 60≤ eGFR <90, and 30≤ eGFR <60 mL/min/1.73 m2.
Comparison of the Receiver Operating Characteristics Curves between NT and HT
Among the representative biomarkers analyzed (HOMA-R, TNF-α, sTNFR1, sTNFR2, CRP, and LDL-C) ROC curves analysis demonstrated that sTNFR1 was the second best discriminatory biomarker for HT after HOMA-R (Tab. II).
Comparison of the Receiver Operating Characteristics Curves for Several Biomarkers Between Normotensive Subjects and Hypertensive Patients
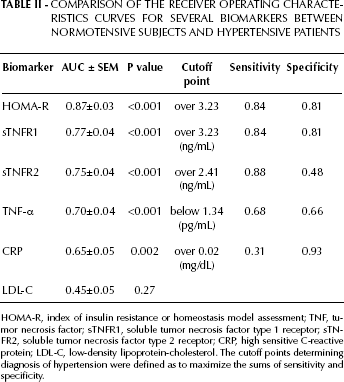
HOMA-R, index of insulin resistance or homeostasis model assessment; TNF, tumor necrosis factor; sTNFR1, soluble tumor necrosis factor type 1 receptor; sTN-FR2, soluble tumor necrosis factor type 2 receptor; CRP, high sensitive C-reactive protein; LDL-C, low-density lipoprotein-cholesterol. The cutoff points determining diagnosis of hypertension were defined as to maximize the sums of sensitivity and specificity.
Correlations between Cardiometabolic Parameters in NT
When analyzing the correlation among demographic characteristics, TNF-related parameters, and cardiorenal metabolic parameters in NT, BMI was significantly correlated with HOMA-R (r=0.55, p<0.001), SBP (r=0.39, p<0.001), and TG (r=0.39, p=0.02). No significant correlation was observed between TNF-α and the other factors. Additionally, sTNFR1 was directly correlated with sTNFR2 (r=0.72, p<0.001), and inversely correlated with HDL-C (r=-0.32, p<0.05) and eGFR (r=-0.45, p<0.001) (Tab. III). sTNFR2 was directly correlated with IRI (r=0.26, p<0.05), and inversely correlated with HDL-C (r=-0.26, p<0.05) and eGFR (r=-0.43, p<0.001). In multivariate stepwise regression analysis with the TNF-related parameters as dependent variables and SBP, HOMA-R, CRP, HDL-C, and eGFR as independent variables (Tab. III), sTNFR1 and sTNFR2 were significantly correlated with lower eGFR levels. These results suggested that sTNFR1 and sTNFR2 were markers of lower eGFR in NT.
Stepwise Regression Analysis in Normotensive Subjects
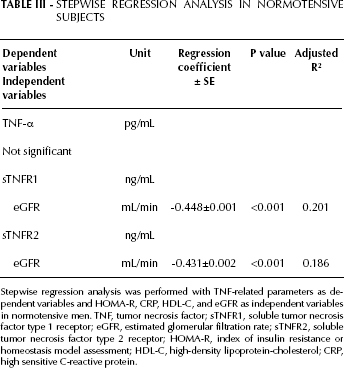
Stepwise regression analysis was performed with TNF-related parameters as dependent variables and HOMA-R, CRP, HDL-C, and eGFR as independent variables in normotensive men. TNF, tumor necrosis factor; sTNFR1, soluble tumor necrosis factor type 1 receptor; eGFR, estimated glomerular filtration rate; sTNFR2, soluble tumor necrosis factor type 2 receptor; HOMA-R, index of insulin resistance or homeostasis model assessment; HDL-C, high-density lipoprotein-cholesterol; CRP, high sensitive C-reactive protein.
Correlations between Cardiometabolic Parameters in HT
When analyzing the correlation among demographic characteristics, TNF-related parameters, and cardiorenal metabolic parameters in HT, BMI was significantly correlated with HOMA-R (r=0.30, p<0.01), TG (r=0.25, p<0.05), UA (r=0.23, p<0.05), and CRP (r=0.36, p=0.001) and inversely correlated with HDL-C (r=-0.23, p<0.05). TNF-α was correlated with sTNFR1 (r=0.28, p<0.05), sTNFR2 (r=0.49, p<0.001), UA (r=0.22, p<0.05), CRP (r=0.37, p<0.001), pretreatment SBP (r=0.26, p<0.05), DBP (r=0.26, p<0.05), and the changes in SBP before and during the treatment (r=0.22, p<0.05) (Tab. IV). Additionally, sTNFR1 was directly correlated with sTNFR2 (r=0.65, p<0.001), and inversely correlated with HDL-C (r=-0.24, p<0.05) and eGFR (r=-0.58, p<0.001). sTNFR2 was directly correlated with HOMA-R (r=0.26, p<0.05) and CRP (r=0.29, p<0.05), and inversely correlated with HDL-C (r=0.22, p<0.05), and eGFR (r=-0.46, p<0.001). In multivariate stepwise regression analysis with the TNF-related parameters as dependent variables and pretreatment SBP, HOMA-R, CRP, HDL-C, and eGFR as independent variables (Tab. IV), TNF-related parameters were significantly correlated with lower eGFR and higher CRP levels. These results suggest that a disturbed TNF system was more closely linked with chronic inflammation than with insulin resistance in HT.
Stepwise Regression Analysis in Hypertensive Patients
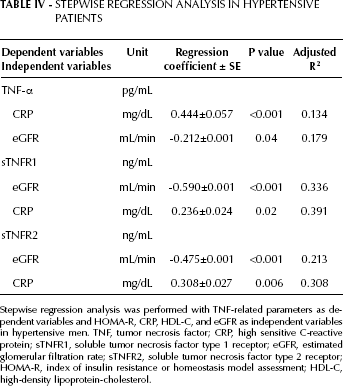
Stepwise regression analysis was performed with TNF-related parameters as dependent variables and HOMA-R, CRP, HDL-C, and eGFR as independent variables in hypertensive men. TNF, tumor necrosis factor; CRP, high sensitive C-reactive protein; sTNFR1, soluble tumor necrosis factor type 1 receptor; eGFR, estimated glomerular filtration rate; sTNFR2, soluble tumor necrosis factor type 2 receptor; HOMA-R, index of insulin resistance or homeostasis model assessment; HDL-C, high-density lipoprotein-cholesterol.
Antihypertensive Medications and TNF-Related Parameters
Among the cardiorenal metabolic factors, IRI and HOMA-R were significantly lower in HT using ACEI or ARB than in HT not using any of these medications (Tab. V). In contrast, pretreatment SBP, IRI, and HOMA-R were significantly higher in HT using CCB than in HT not using CCB. Besides, IRI, HOMA-R, UA, and WBC were significantly higher in HT using diuretics than in HT not using diuretics. No significant differences were observed in either SBP or DBP levels during treatment, or in TNF-related parameters and CRP levels according to the type of antihypertensive medication (ACEI, ARB, CCB or diuretics), although sTNFR2 tended to be higher in HT treated with diuretics (p=0.06). Therefore, the various antihypertensive medications had differential effects on insulin resistance, but no significant impact on the TNF system or chronic inflammation state.
Differences in Cardiorenal Metabolic Parameters According to Antihypertensive Medications
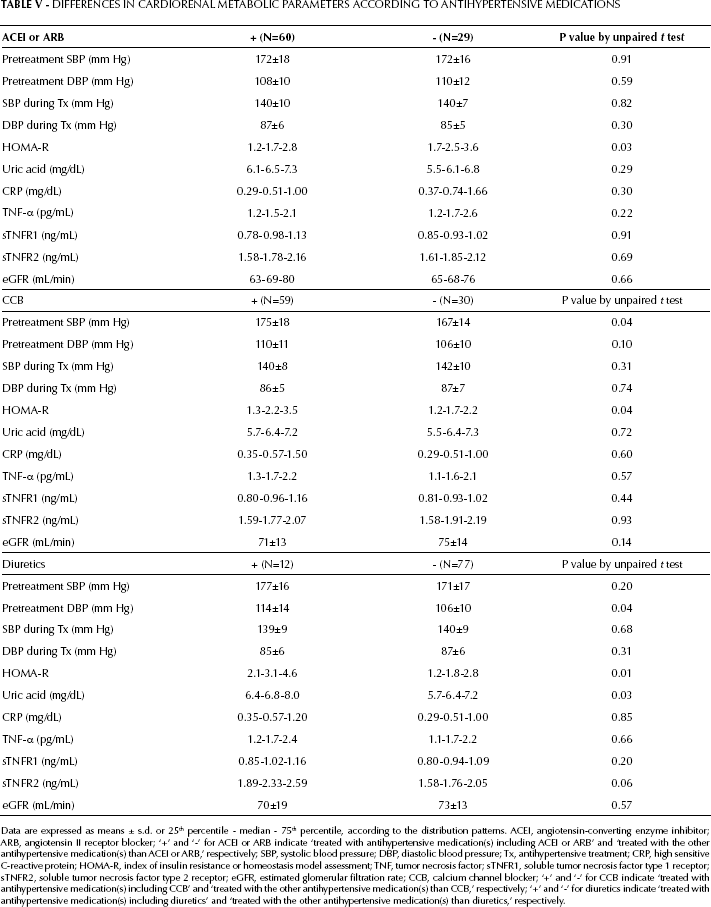
Data are expressed as means ± s.d. or 25th percentile - median - 75th percentile, according to the distribution patterns. ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin II receptor blocker; ‘+’ and ‘-’ for ACEI or ARB indicate ‘treated with antihypertensive medication(s) including ACEI or ARB’ and ‘treated with the other antihypertensive medication(s) than ACEI or ARB,’ respectively; SBP, systolic blood pressure; DBP, diastolic blood pressure; Tx, antihypertensive treatment; CRP, high sensitive C-reactive protein; HOMA-R, index of insulin resistance or homeostasis model assessment; TNF, tumor necrosis factor; sTNFR1, soluble tumor necrosis factor type 1 receptor; sTNFR2, soluble tumor necrosis factor type 2 receptor; eGFR, estimated glomerular filtration rate; CCB, calcium channel blocker; ‘+’ and ‘-’ for CCB indicate ‘treated with antihypertensive medication(s) including CCB’ and ‘treated with the other antihypertensive medication(s) than CCB,’ respectively; ‘+’ and ‘-’ for diuretics indicate ‘treated with antihypertensive medication(s) including diuretics’ and ‘treated with the other antihypertensive medication(s) than diuretics,’ respectively.
When analyzing the correlation between TNF-α and sTNFRs, TNF-α was significantly correlated with sTNFR1 and sTNFR2 in HT treated with ACEI or ARB and in HT treated with CCB. However, this correlation was hidden in HT treated with diuretics, HT without ACEI or ARB, and HT without CCB. The strong correlation between sTNFR1 and sTNFR2 was preserved in all the subgroups of HT subdivided according to antihypertensive medications.
Discussion
This study confirmed that HT had insulin resistance and chronic inflammation. It also showed that HT had decreased TNF-α levels, increased sTNFR1 and sTNFR2 levels, or a disturbed TNF system. Taken separately, both the 2 study groups of NT and HT showed strong inverse correlations between soluble TNF receptors and eGFR levels. Furthermore, for HT we found significant correlations between the TNF-related parameters and chronic inflammation measured by CRP. Therefore, the disturbed TNF system seems to be closely linked with chronic inflammation in HT.
Earlier pioneer studies reported that low-grade inflammation was an early marker of renal dysfunction in the general population (22, 23). Recently, Gohda et al reported, in 2 longitudinal cohort studies (24, 25), that sTNFR1 and sTNFR2 levels predict renal dysfunction in type 1 and type 2 diabetic patients. Our results suggest that increased sTN-FR1 and sTNFR2 could be markers of mild renal dysfunction and chronic inflammation in HT. Keller et al reported that TNF-α, sTNFR1, and sTNFR2 were directly correlated with plasma cystatin C concentrations and inversely correlated with eGFR (26, 27). Their findings are generally consistent with ours. One attractive hypothesis is that chronic systemic inflammation can accelerate renal dysfunction in hypertension. In a rat model, renal interstitial inflammation with increased circulating TNF-α levels has recently been reported to play a key role in early renal dysfunction in hypertension (28). Another possibility is that renal dysfunction results in low clearance of TNF-α (29). The strong inverse correlations between soluble TNF receptors and eGFR were consistent both in NT and HT. The fact that TNF-related parameters were significantly correlated to CRP levels only in HT supports the hypothesis that activation of the TNF system contributes to the development of hypertension via chronic inflammation. Besides, the correlations between sTNFR2 and IRI in NT and between sTNFR2 and HOMA-R in HT support the hypothesis that sTNFR2 plays a role in insulin resistance in NT as well as in HT.
In our study, the TNF-α levels were lower in HT than in NT, a finding that was apparently inconsistent with earlier studies, including those conducted with obese patients (30, 31). These studies reported that TNF-α was higher in hypertensive subjects than in normotensive controls. Possible explanations for this discrepancy may be: first, a difference in the inclusion criteria of the subjects in the study. The present study was conducted exclusively in males, relatively leaner subjects (BMI: 22.3 in NT and 24.9 in HT). The low proportion of obese subjects might have kept low the TNF-α levels. In addition to this, differences in racial and cultural backgrounds, including dietary habits, may have affected the results. Second, antihypertensive medications were prescribed in HT: 60 of them were using ACEI or ARB, 59 CCB, and 12 diuretics. Antihypertensive medications can potentially lower the TNF-α levels (32). Patel et al reported that TNF-α levels were lower in treated hypertensive patients compared with either patients who were newly diagnosed with hypertension or with healthy controls (33). We could not detect significant differences in TNF-α levels according to antihypertensive medications. However, TNF-α levels positively correlated with the changes in SBP before and during antihypertensive treatment. These results do not support the hypothesis that antihypertensive medications markedly decrease the TNF-α levels in HT, and we should have compared the TNF system before and during treatment. Third, some HT received medications, such as statin (7/89 cases) and antiplatelet agent (5/89 cases), which may have decreased their TNF-α levels. Some HT might have accepted lifestyle modifications, such as a vigorous physical activity and a low salt diet, which have the potential to ameliorate chronic inflammation associated with hypertension. Fourth, earlier studies indicate that the absolute value of plasma TNF-α concentrations is usually extremely low (in the order of pg/mL). It is thus too rough to evaluate the activity of the TNF network only on basis of TNF-α concentration (1, 34). In this study, TNF-α levels were correlated strongly with sTNFR1, sTNFR2 levels and only moderately with chronic inflammation in HT. Although the TNF-α concentration was unexpectedly lower in HT than NT, higher sTNFRs levels positively correlating with CRP and HOMA-R indicated that the TNF network activity was enhanced in HT. The correlation between TNF-α and sTNFRs was preserved in HT treated with ACEI, ARB, or CCB while it was hidden in HT treated with diuretics. While ACEI, ARB, and CCB might all modulate the disturbed TNF network into the same direction, diuretics might modulate the TNF-related parameters differently.
Antihypertensive medications had differential effects on insulin resistance. First, our results suggest that ACEI and ARB improve insulin resistance, but not by improving the activated TNF system and chronic inflammation. Second, pretreatment SBP and HOMA-R were significantly higher in HT taking CCB. Since CCB has been reported to be neutral or beneficial to insulin sensitivity in hypertension (35), a selection bias towards the higher pretreatment SBP levels might contribute to insulin resistance associated with HT taking CCB. Third, pretreatment DBP and HOMA-R were significantly higher in HT taking diuretics and the sTNFR2 levels tended to be higher in HT taking diuretics. These results indicated that a selection bias towards the higher pretreatment DBP levels might contribute to insulin resistance associated with HT taking diuretics. Otherwise, diuretics could worsen insulin resistance through the increased sTNFR2 levels.
In our preliminary study, we compared the differences in TNF-α, sTNFR1 and sTNFR2 levels before and during the treatment with ARB. While TNF-α and sTNFR1 levels were similar between the 2 timepoints, sTNFR2 levels were slightly but significantly decreased during ARB treatment. Therefore, the substantially higher sTNFRs levels in HT compared to NT were not considered as the effect of ARB treatment (21st Scientific Meeting of the International Society of Hypertension, 2006, Abstract).
Limitations
The main limitation of this study is its cross-sectional nature. We can thus not draw conclusions about the causality of associations and correlations; a further prospective investigation is required for clarifying these causal relationships. Additionally, the findings we obtained from NT and HT cannot be generalized to females (as female hormones may influence the TNF system, especially during menopause) or stage 1 hypertensive male subjects. In addition, we adopted different inclusion criteria of age for NT and HT as to reduce the effects of age on the discordant blood pressure levels. However, the different inclusion criteria might add a confounder to our analyses. The association of HT with higher sTNFR2 levels and insulin resistance can be partially attributable to higher BMI in HT.
In conclusion, a disturbed TNF system and insulin resistance were associated with treated stage 2 and 3 HT. The disturbed TNF system was more strongly correlated with lower eGFR and only moderately correlated to chronic inflammation compared to insulin resistance in HT. Although ACEI or ARB have beneficial effects on insulin resistance, they have no substantial impacts on the disturbed TNF system and chronic inflammation. Future studies should focus on exploring an antihypertensive agent with a clinical potential to improve chronic inflammation by modulating the disturbed TNF system.
Footnotes
Acknowledgments
We thank Mai Iida and Naoko Ohara for assisting us with sample collection.
