Abstract
Background
The aim of this study was to investigate the effectiveness of different remineralization agents by quantitative light-induced fluorescence digital Biluminator™ (QLF-D).
Methods
Artificial caries lesions were created, and the teeth were divided according to the tested materials: (i) distilled water, (ii) acidulated phosphate fluoride (APF), (iii) Curodont Repair (CR), (iv) ammonium hexafluorosilicate (SiF) and (v) ammonium hexafluorosilicate plus cetylpyridinium chloride (SiF + CPC). After treatment procedures, each of the samples was placed in artificial saliva. After demineralization and 1 and 4 weeks of remineralization procedures, fluorescence loss and lesion areas were measured with QLF-D. Data were statistically analyzed (α = 0.05).
Results
The fluorescence values of the demineralized enamel specimens treated with the various agents differed significantly compared with pretreatment values for both 1 and 4 weeks (p<0.05). At 4 weeks, the highest fluorescence gain was calculated in the CR, APF and SiF groups compared with the control (p<0.05).
Conclusions
APF, SiF and CR groups yielded greater remineralization ability than SiF + CPC and control groups.
Introduction
Dental caries is a bacterial-driven process that depends on the balance between plaque fluid and tooth structure. When pathological factors (salivary dysfunction, fermentable carbohydrates, cariogenic bacteria etc.) became predominant, alternative approaches are necessary to promote remineralization (1, 2).
One of the most effective ways to prevent caries is the use of fluoride applications (3–4–5). The anticaries activity of fluoride can be attributed to its inhibition of the demineralization processes, enhancement of enamel remineralization and effect on the biological activities of cariogenic microorganisms (3–4–5). Several different fluoride agents, such as acidulated phosphate fluoride (APF), sodium fluoride, stannous fluoride and amine fluoride, have been used clinically (3, 4, 6–7–8). Although the anticaries effect of these fluoride-containing products has been known for some time, ongoing studies are attempting to develop new fluoride-based materials that are more effective.
One of those materials, ammonium hexafluorosilicate (SiF) solution, has been suggested as an alternative fluoride-containing anticaries agent (9–10–11–12–13). A limited number of studies has indicated that SiF treatment does not produce tooth staining; upon application, a large amount of precipitates forms at the tooth surface, and the acidity of the solution causes etching of the hard tissues of the tooth and reprecipitation of calcium and phosphate ions as silica-calcium-phosphate crystals (9–10–11–12–13). In addition, it has been emphasized that the antibacterial activity of the SiF solution can be increased with the addition of several antibacterial agents, such as cetylpyridinium chloride (CPC) (13). However, there are a limited number of studies about SiF solution in the literature, and prior to clinical use, a more detailed evaluation must be done on the remineralization activity of the material with or without antibacterial agents.
Although the anticaries ability of fluoride has a known benefit, high concentrations of fluoride mainly increase the remineralization of the outer enamel and decrease the demineralization of the inner enamel, frequently resulting in a superficial remineralization layer (10, 14). Accordingly, other anticaries agents still need to be assessed, which could include a useful material for enhancing the remineralization of caries lesions.
In recent years, an alternative nanotechnology for remineralization has been developed for the repair of the enamel matrix by using self-assembling peptides (P11−4; Curodont Repair [CR]) (15). Studies have shown that peptide treatment significantly increases the mineral content of the lesion body and inhibits mineral loss (15–16–17–18). P11-4 induces hydroxyapatite nucleation and initiates crystal formation (15–16–17–18). Based upon these data, it is thought that these materials could be useful in the management of incipient caries lesion progression. However, no comparative studies have been performed with self-assembling peptides, and the current literature provides limited information on the remineralization capacity of the material.
To assess the treatment's efficacy and accurately measure changes in the lesion progress, a variety of assessment and quantification analysis techniques can be used. Ideally, the evaluation method should be accurate, reproducible, noninvasive and simple to use (19, 20). A quantitative light-induced fluorescence (QLF) imaging system (Inspektor™ Pro; Inspektor Research Systems BV, Amsterdam, The Netherlands) is a tool that can be used for the quantitative assessment and longitudinal monitoring of mineral changes in the early stages of caries (20–21–22–23–24). Recent studies have indicated that QLF is a noninvasive, objective, highly sensitive, reliable, repeatable, reproducible and accurate diagnostic tool (20–21–22–23–24). The quantitative light-induced fluorescence digital Biluminator™ (QLF-D) is an upgraded version of the first product, which has been upgraded to enhance these characteristics of the QLF (24). Although there are a lot of studies in the literature using QLF to evaluate the remineralization/demineralization process, only a limited number of studies have been performed with QLF-D. Therefore, the purpose of this study was to determine the remineralization potentials of the 3 different fluoride-containing topical agents and a self-assembling peptide for regenerating demineralized smooth surfaces by QLF-D. The tested null hypothesis was that there were no statistically significant differences in terms of the remineralization effect for the different materials tested.
Methods
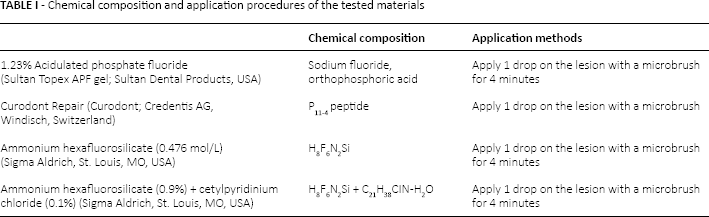
In the study, 4 remineralization agents were tested: APF, CR, SiF and ammonium hexafluorosilicate plus cetylpyridinium chloride (SiF + CPC). Furthermore, 2 different treatment periods (1 and 4 weeks) were assessed. The chemical composition, manufacturer and application procedures for the tested materials are presented in Table I. The present study was approved by the medical ethics committee of Izmir Katip Celebi University (report no. 2013/204).
Chemical composition and application procedures of the tested materials
Sample preparation
Extracted human mandibular molars free from clinically visible abnormalities were cleaned of debris and stains with pumice, stored in a 1% thymol solution at 4°C, and used within 1 month of the extraction. The teeth were thoroughly rinsed and examined under a stereomicroscope (Olympus SZ61; Olympus Optical Co., Tokyo, Japan); any tooth with defects, erosions or microcracks on its enamel surfaces was excluded. The soft tissue and calculus were removed mechanically from the tooth surfaces with a periodontal scaler. The roots of the tooth were removed 2 mm below the cementoenamel junction using a water-cooled diamond disk (Buehler Inc., Lake Bluff, IL, USA). The coronal part of each tooth was longitudinally sectioned in the mesiodistal direction, providing enamel slabs. Each enamel slab was embedded horizontally on self-cured acrylic resin (Vertex Dental, Zeist, Netherlands) in prefabricated cylindrical molds keeping the buccal surface exposed and cured overnight. The surface of the tooth slabs was polished using 800-, 1200- and 2400-grit silicon carbide paper to obtain a flat and smooth enamel surface. The flattened buccal/lingual surfaces of the specimens were bonded with a standardized strip to create a 2.5-mm-diameter window in the middle.
Following the preparations of the specimens, artificial carious lesions were created on the buccal surface of each tooth in an acidified gel system for 7 days. The gel was prepared by adding 100 mmol/L sodium hydroxide, 100 mmol/L lactic acid and 6% w/v hydroxyethyl cellulose. The final pH value was 4.5, and final viscosity was 100 cP (25). After a 7-day demineralization, the specimens were cleaned of the demineralization solution with deionized water. Following the caries lesion formation, the strip was removed from the specimen, and a circular artificial caries lesion with a diameter of 2.5 mm was obtained from each slab.
Initial QLF analyses
At the end of the demineralization phases, the mean fluorescence loss for each enamel slab was assessed with the QLF-D Biluminator™. The QLF-D was fixed to the QLF-D in vitro stand to position the specimens with the same angle and camera position. Video-repositioning software of the QLF system was used to capture the images when the correlation was higher than 0.9. Before the measurements, the specimens were dried for 5 seconds with an air syringe. Additionally, the measurement was performed for each enamel slab after 15 minutes to dry the specimen equally. The relative fluorescence loss (ΔFinitial) was calculated as the pretreatment (baseline) fluorescence value. During the analysis, a contour was drawn around the artificial caries lesion area using the white spot wizard. The contour of the area was marked on the original sound enamel. The difference between the sound enamel (reference area) and the lesion area was calculated, and quantitative results were obtained for the following parameters: mean fluorescence loss over the lesion (ΔF%) and area of the lesion (square pixels). Specimens with an initial baseline ΔF of 20-25 were chosen for the experiments to allow standardization.
Treatment procedures
After baseline QLF measurement, a total of 75 enamel specimens were randomly divided into 5 groups (15 enamel slabs each) according to the tested materials and 1 of the following treatments was administered: (i) distilled water (control), (ii) APF, (iii) CR, (iv) SiF or (v) SiF + CPC. While SiF and SiF+CPC solutions were laboratory solutions, they were prepared following the previous study protocols (11, 13). After the application of treatment agents according to the manufacturers’ instructions (Tab. I), each of the samples was placed in containers independently, containing 10 mL artificial saliva (25) (MgCl26H2O, K2HPO4, KCl, calcium lactate, fluoride, sodium carboxymethylcellulose, methyl-4-hydroxybenzoate, pH 7.2), and all samples were placed in an incubator at 37°C with 5% CO2 for 1 and 4 weeks. The artificial saliva was renewed daily.
Posttreatment QLF analyses
The mean fluorescence loss and area of the lesions were calculated at 1 and 4 weeks after the treatment and saliva immersion procedures, as described in the initial QLF analysis step.
Statistical Analysis
To determine the fluorescence changes, the initial and posttreatment results (1 and 4 weeks) of each group and the differences between the 1- and 4-week treatments were compared using paired sample t-tests. Intergroup comparisons were performed with a multivariate analysis of variance (p<0.05), followed by Tukey's test. All statistical analyses of the data were conducted using SPSS statistical software version 21.0 (SPSS Inc., Chicago, IL, USA), with an alpha value of 0.05 selected as the level of significance.
Results
The results of the QLF measurements in each group at the different time intervals are presented in Table II. The fluorescence values of the demineralized enamel specimens treated with the various agents differed significantly compared with the pretreatment values (p<0.05). The mean values for fluorescence gains between the baseline and posttreatment (1 and 4 weeks) are presented in Table III. The fluorescence gains of the APF and SiF groups were significantly higher than those of the control group after 1 week (p<0.05). However, no significant differences were observed among the CR and SiF+CPC groups when compared to the controls (p>0.05). At 4 weeks, the highest fluorescence gains were calculated in the CR, APF and SiF groups compared with the control (p<0.05). In addition, there was significant interaction between all of the materials’ applications and the time intervals (1 and 4 weeks). All materials showed more fluorescence gain at 4 weeks compared with 1 week. The lesion areas of all study groups differed significantly compared with the pretreatment values, at the end of the 1-week and 4-week remineralization periods (p<0.05).
Fluorescence loss (ΔF%) and lesion area in each study group, at baseline and end of 1-week and 4-week remineralization
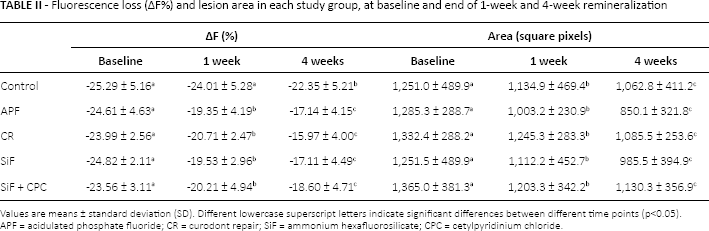
Values are means ± standard deviation (SD). Different lowercase superscript letters indicate significant differences between different time points (p<0.05).
APF = acidulated phosphate fluoride; CR = curodont repair; SiF = ammonium hexafluorosilicate; CPC = cetylpyridinium chloride.
Fluorescence gains (ΔF%) between the study groups for different time intervals
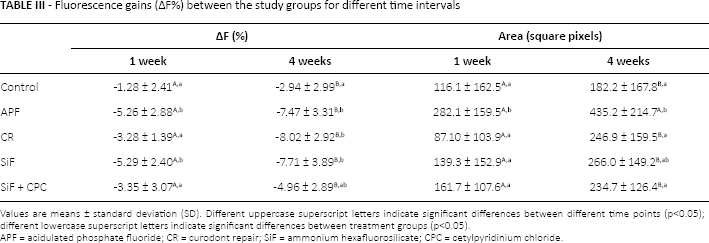
Values are means ± standard deviation (SD). Different uppercase superscript letters indicate significant differences between different time points (p<0.05); different lowercase superscript letters indicate significant differences between treatment groups (p<0.05).
APF = acidulated phosphate fluoride; CR = Curodont Repair; SiF = ammonium hexafluorosilicate; CPC = cetylpyridinium chloride.
Discussion
QLF is a visible light system that can quantitatively monitor caries lesion progression or regression in a nondestructive way (24, 26). Previous studies have tested the QLF method in in vitro experiments with transverse microradiography, optical coherence tomography, scanning electron microscopy (SEM), energy dispersive X-ray spectroscopy, surface microhardness tests and digital photography (27, 28). These studies reported that QLF could be used to monitor the progression or regression of enamel demineralization. QLF-D, a newly developed device, is based on the same principle but with an enhanced light source and filter system compared with the old version. Although there are numerous studies in the literature using QLF to evaluate the remineralization ability of different materials, limited studies have been performed with QLF-D. Therefore, in this study, the quantitative, light-induced fluorescence digital Biluminator™ system was used to evaluate the remineralization capacity of the different treatment agents.
During the QLF analyses, a standardized technique should be used to eliminate some interference factors, such as environmental conditions, operational motion and degree of dehydration (26). In the present study, all QLF images were captured in a dark room with the standardized settings, after all of the enamel specimens were dried totally and equally. All image analyses for each group were analyzed by 1 examiner, whose approach was calibrated prior to the study (intraexaminer kappa value was 0.83). Additionally, with the QLF-D, unlike the old version, an in vitro stand was developed to capture the images of the samples always in the same position and same angle. Also, in the current study, a QLF-D in vitro stand and video-repositioning software were used to eliminate the interference caused by the position of the specimens and operator-dependent variables.
According to the results of this study, there were significant differences between the initial and posttreatment QLF measurements, and all of the experimental groups had significant regaining of fluorescence and reduction of the lesion area. With regard to fluorescence gain, CR was more effective than the control and SiF + CPC treatments, and it was as effective as APF and SiF. A few studies have assessed the remineralization capacity of CR, and the results obtained here were consistent with those of previous reports (15–16–17–18). Takahashi et al reported that CR application is effective in preventing enamel demineralization, has the ability to enhance enamel remineralization and can be considered as a microinvasive approach for preventing enamel demineralization (16). Brunton et al suggested that the treatment of early caries lesions with CR was safe and that a single application was associated with significant enamel regeneration, presumably by promoting mineral deposition within the subsurface tissue (17). Jablonski-Momeni et al evaluated the remineralization efficacy of P11-4 with a fluorescence-based caries diagnostic device and SEM and found that CR application created visual and microscopic changes in artificially created enamel lesions (18). The researchers suggested that in situ and in vivo studies should be designed to assess the efficacy of CR over the long term and in comparison with other preparations. The self-assembling peptide was designed to form fibrils at a low pH and to be monomeric in solutions with a higher pH. These peptides play an important role in the mineralization process, where negatively charged surfaces and phosphorylated serine residues attract Ca2+, initiate crystal formation and control the orientation and elongation of the hydroxyapatite crystals (15, 18). Unlike fluoride solutions with a low pH, peptides have no solvent effect on dental hard tissues. These low-pH solutions etch the surface of the enamel and cause quick precipitation on the enamel surface due to the formation of Ca fluoride (CaF2) (27). However, following the application of the CR, accumulation of saliva components in the skeletal structure was realized slowly. This mechanism may explain the relatively low fluorescence gain after the 1-week treatment with CR and the high fluorescence gain after the 4-week treatment with this agent in the present study.
In the current study, the APF and SiF solutions were also more successful than the control group after both 1 and 4 weeks. The results of the present study are consistent with those of many studies in the literature, which have indicated that APF is an effective cariostatic and caries-prevention agent (6, 7, 27, 29). The remineralization capacity of the APF solution is caused by high fluoride content and the acidic structure of the material (6, 27). The low pH of APF gel may result in etching of surface enamel and increased surface roughness soon after its application. Then the CaF2, created by the Ca, loses this erosive tissue and F sourced from the APF solution, causing a rapid increase in the hardness of the enamel (6, 27). The high fluorescence gain after the 1-week treatment in the current study supports this idea. Similar to our results, Kim et al suggested that APF is an effective material in reducing the depth of the incipient caries lesions (30). Researchers explained that the dissolving effect of the material and formation of CaF2 was due to calcium from the teeth, as well as penetration of fluoride into the enamel structure. Lee et al confirmed that the remineralizing efficacy of 1.23% APF gel application was found to be as successful as 5% fluoride varnish, and it significantly decreased the fluorescence lesion area compared with the control group and the group with iontophoresis using 2% sodium fluoride (27). In accordance with these studies, the findings of the current study indicate that a single APF application and 1- to 4-week saliva immersion provide significant fluorescence gain and reduce the area of the lesions, which denoted the remineralization capacity of the material.
According to the results of the study, another material that induced the greatest amount of enamel remineralization was SiF. In an in vitro study, Hosoya et al evaluated the effects of the SiF solution on different layers of demineralized enamel (10). They detected a high concentration of Ca and P ions in the deepest layers of the enamel and suggested that the solution penetrated the deepest layers of the lesions. In accordance with the findings of Hosoya et al, the high fluorescence gain obtained in the present study with SiF indicates that the material has a great remineralization capacity on demineralized enamel specimens. Similar to APF, SiF is an acidic solution and dissolves the upper layer of enamel after a single application and leads to the formation of firmly bound fluoride (9, 10). Firmly bound fluoride is not easily washed away in the oral environment, plus it forms fluoridated apatite, which contributes to increasing the acid resistance of teeth. SiF also contains silica, and silica-containing compounds cause rapid silica-calcium-phosphate precipitation and induce apatite formation (9, 10). These mechanisms may explain the high fluorescence gain obtained after the application of SiF in the present study.
In addition to evaluating the effect of SiF alone in the current study, the addition of CPC to the content of the material was also assessed. CPC is an effective antibacterial agent. It exerts an antibacterial action by combining with negatively charged proteins on bacterial cells and destroying their cell membrane by disturbing the electric balance (13). A previous study reported that the addition of CPC to SiF solution significantly increased the antibacterial activity of the agent (13). However, no previous studies have compared the effect of a combination of CPC and SiF on remineralization activity versus that of SiF alone. According to the results of the present study, SiF+CPC solution induces the remineralization of the artificial caries lesion; however, no significant difference was found when compared with the control group. This study clearly demonstrated that addition of CPC adversely affected the activity of SiF.
In the present study, the remineralizing efficacy of 3 different fluoride-containing topical agents and a self-assembling peptide was evaluated using QLF-D. Based on the results of the study, the null hypothesis was rejected since differences in the remineralization capacities were observed among the different materials tested. CR, APF and SiF were found to be quite successful at the end of the 4-week treatment periods. Although this study does not simulate exactly the conditions of the oral environment, the results provide useful information on the remineralization effectiveness of the different agents.
Conclusions
All of the agents used in the study aided the remineralization of initial caries lesions.
CR, APF and SiF exhibited similar remineralization abilities. These findings suggest that different materials enhance the remineralization of artificial caries lesions via different mechanisms.
CR is a promising remineralization agent that has been proven to be an anticaries agent similar to fluoride-based solutions, and it seems to be suitable for clinical use.
Footnotes
Financial support: No grants or funding have been received for this study.
Conflict of interest: None.
