Abstract
Purpose
The purpose of the study was to evaluate the surface characteristics of demineralized enamel after treatment with pastes containing casein phosphopeptide–amorphous calcium phosphate (CPP-ACP) or casein phosphopeptide–amorphous calcium fluoride phosphate (CPP-ACFP) and to compare their efficacy with that of 0.05% NaF.
Methods
Following formation of the artificial carious lesion, enamel slabs were divided into 4 groups (CPP-ACP, CPP-ACFP, 0.05% NaF and control) and submitted to a chemical caries model. Remineralization potential was examined using scanning electron microscope, energy-dispersive spectroscopy (EDS) and microhardness test. Scanning electron microphotographs were analyzed for area, minimal, maximal and mean diameter, perimeter, roundness and number of enamel defects and percentage of tooth surface affected by defects.
Results
Treatment with 0.05% NaF partly reduced the appearance of enamel defects when compared with irregular demineralized enamel. Treatment with CPP-ACP or CPP-ACFP resulted in occlusion of defects which produced more flattened enamel surface. Image analysis revealed reduction of the dimensions of the defects in the 3 experimental groups. Treatment with CPP-ACFP decreased the number of enamel defects when compared with demineralized enamel. The EDS analysis did not show differences in Ca/O, P/O and Ca/P ratios between the groups (P>0.05). Microhardness test revealed significant effects of CPP-ACP and CPP-ACFP (P<0.05).
Conclusions
Pastes containing CPP-ACP or CPP-ACFP showed potential to remineralize enamel surface lesions.
Keywords
Introduction
Contemporary minimally invasive approaches to the treatment of dental caries are based on the assessment of caries risk and control of caries as an infectious disease (1). It is well known that early diagnosis and arresting of the caries process can delay operative treatment, and the possibility of remineralizing early enamel lesions has been clearly demonstrated (2). However, although various techniques for early caries detection, risk assessment and caries control have progressively developed in recent decades, there is still no consensus on caries management.
Conventional caries prevention methods are primarily based on applications of fluoride. For over 40 years, numerous fluoride preparations have been available to the public to inhibit caries and promote remineralization. Topical fluorides have been recommended for the noninvasive treatment of incipient enamel subsurface lesions, especially if the risk factors of poor oral hygiene and cariogenic diet are also present (3).
Apart from fluoride, there must be sufficient calcium and phosphate ions present before the enamel is able to remineralize. It has been shown that for every 2 fluoride ions, 10 calcium and 6 phosphate ions are required to form 1 unit cell of fluoroapatite, so the availability of calcium and phosphate ions can be the limiting factor for remineralization after the topical application of fluoride (4). Casein phosphopeptides (CPP) have the ability to stabilize calcium and phosphate ions in solution in the form of amorphous calcium phosphate (ACP) (5). The CPP-ACP nanocomplex incorporates into dental plaque, where it is claimed to act as a reservoir of calcium and phosphate ions and helps plaque fluid to maintain a state of super-saturation with respect to enamel, thus depressing enamel demineralization and enhancing remineralization (6). Recently, the addition of fluoride to the CPP-ACP nanocomplex has been proposed, and a novel amorphous calcium fluoride phosphate (ACFP) phase was shown to exhibit superior remineralization (7, 8).
Although the anticariogenic potential of CPP-ACP has been studied comprehensively, little is known about the effect of the nanocomplex on enamel surface. The purpose of this study was to evaluate surface characteristics of demineralized enamel after treatment with pastes containing CPP-ACP or CPP-ACFP, and to compare their efficacy with that of 0.05% NaF.
Materials and Methods
Institutional ethics committee approval was obtained for the study. Ten buccal or lingual surfaces obtained from sound extracted third molars were used in the present study. After extraction, soft debris and periodontal ligaments were removed, and each tooth was inspected for cracks, hypoplasia and white spot lesions, and stored in physiological saline at +4°C until used for experiments. Buccal and/or lingual surfaces were separated using a water-cooled diamond-impregnated low-speed saw (Isomet Low Speed Saw; Buehler, Lake Bluff, IL, USA). Surfaces other than buccal/lingual enamel were coated with an acid-resistant varnish. Prior to the experiment, each enamel slab was divided into 2 parts. One half of each specimen was used for scanning electron microscopy (SEM) and energy-dispersive spectroscopy (EDS). The other half of the specimen was embedded in self-cured acrylic resin (Palavit G; Heraeus Kulzer GmbH, Hanau, Germany) using a cylindrical Teflon mould, leaving enamel surface exposed. Enamel surfaces were ground wet using 600, 1,500 and 2,000 grit silicon carbide paper (3M, St. Paul, MN, USA) to expose a flat surface for the microhardness test. To produce an artificial carious lesion, enamel slabs were immersed in a demineralizing solution (pH 4.0) for 3 days. The demineralizing solution contained 100 mmol/L lactate, 3.0 mmol/L CaCl2, 1.8 mmol/L KH2PO4 and 1 wt% carboxymethylcellulose (9).
Following formation of the artificial lesion, each slab was divided into 4 parts to form 4 experimental groups (n=10): (i) CPP-ACP (Tooth Mousse, containing 10% CPP-ACP; GC Int., Tokyo, Japan), (ii) CPP-ACFP (MI Paste Plus, containing 10% CPP-ACP and 0.9 mg/g fluoride; GC Int.), (iii) 0.05% NaF (Curasept ADS 205; Curaden International AG, Kriens, Switzerland) and (iv) control demineralized enamel. One of 4 specimens made from the original individual sample was distributed into each test group.
Enamel samples were submitted to a chemical caries model in which enamel was subjected to a number of demineralization and remineralization cycles. Demineralizing solution used in the cyclic model contained 2.0 mmol/L CaCl2, 2.0 mmol/L KH2PO4 and 75 mmol/L acetate (pH 4.3) (10). Remineralizing solution contained 1.2 mmol/L CaCl2, 0.72 mmol/L KH2PO4 and 30 mmol/L KCl (11). The pH of the remineralizing solution was adjusted to 7.0 with potassium hydroxide (KOH). The experimental design included 2 demineralization/remineralization cycles a day during 10 days: immersion of enamel samples in 10 mL of demineralizing solution, twice daily (at 9 AM and 9 PM) for 30 minutes; topical application of the CPP-ACP or CPP-ACFP for 3 minutes, or 0.05% NaF for 60 seconds (simulation of the suggested instructions for use); washing in distilled water for 10 seconds; immersion in 100 mL of remineralizing solution (12).
Remineralization potential was evaluated using SEM, EDS and microhardness test. Enamel samples were coded and examined blindly. For the SEM/EDS analysis, specimens were mounted on aluminium stubs and sputter-coated with gold (Polaron SC502 Sputter Coater; Fisons Instruments, East Grinstead, UK). Surface morphology was examined with SEM (Jeol JSM-5800; Jeol Industries Ltd., Tokyo, Japan) at different magnifications. Elemental analysis was performed with EDS detector (INCAx-sight; Oxford Instruments, Oxfordshire, UK) at ×200 magnification to determine Ca/P, Ca/O and P/O ratios. SEM microphotographs were subsequently analyzed with the image analysis technique. Three photographs of each specimen were first viewed randomly using Adobe Photoshop CS3 software. Areas of enamel loss of different sizes and shapes were marked as enamel defects by 2 calibrated investigators independently. Kappa interexaminer score was 0.93, and disagreements were resolved by discussion. Enamel defects were then analyzed with image analysis software (Image-Pro Plus for Windows). The calibrated images were measured for area, minimal, maximal and mean diameter, perimeter, roundness and number of enamel defects and percentage of tooth surface affected by defects. For the microhardness test, 3 indentations were made in each specimen using a Vickers microindenter under a load of 100 g during 15 seconds (Leitz DURIMET I; Leica Microsystems GmbH, Wetzlar, Germany). Indentations were evaluated with a metallographic microscope (Epival Interphako; Carl Zeiss GmbH, Jena, Germany) at ×12.5 magnification, and the Vickers hardness number (VHN) was calculated.
Descriptive statistical analyses were primarily implemented. Friedman's analysis of variance (ANOVA) was used for the intergroup comparative analyses. The level of significance was set at P<0.05, and the data were processed using the statistical software SPSS for Windows.
Results
SEM microphotographs are shown in Figure 1. Incipient carious lesion showed irregular, roughened appearance with several enamel defects all over the surface (Fig. 1a, b). In 3 experimental groups, different morphology of the enamel surface could be observed. Treatment with 0.05% NaF partly reduced the appearance of enamel defects (Fig. 1c, d). On the other hand, treatment with paste containing CPP-ACP (Fig. 1e, f) or CPP-ACFP (Fig. 1g, h) resulted in occlusion of defects which produced more flattened enamel surface with less apparent defects.
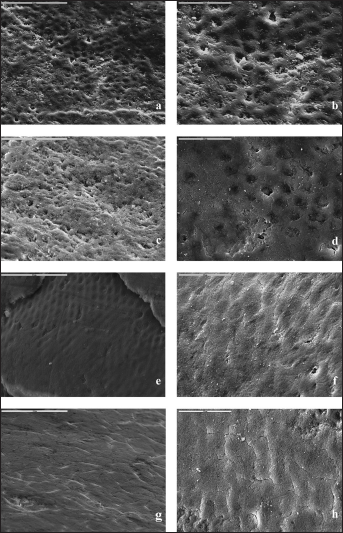
Scanning electron microscopy microphotographs of the enamel showing irregular surface of the incipient carious lesion with numerous defects (a: ×1,000, b: ×2,000), surface after treatment with fluoride with partially occluded defects (c: ×1,000, d: ×2,000), surface after treatment with casein phosphopeptide–amorphous calcium phosphate (CPP-ACP) (e: ×1,000, f: ×2,000) or casein phosphopeptide–amorphous calcium fluoride phosphate (CPP-ACFP) (g: ×1,000, h: ×2,000) resulting in significant alterations of enamel appearance, with occlusion of defects.
Image analysis revealed reduction of the defects' dimensions in the 3 experimental groups (Tab. I). In addition, treatment with CPP-ACFP reduced the number of enamel defects when compared with demineralized enamel (Tab. II). The percentage of defects in the surface examined was 1.82% for demineralized enamel, 1.13% for 0.05% NaF, 0.39% for CPP-ACP and 0.30% for CPP-ACFP.
CHARACTERISTICS OF ENAMEL DEFECTS

Values are means ± SD.
CPP-ACFP = casein phosphopeptide–amorphous calcium fluoride phosphate; CPP-ACP = casein phosphopeptide–amorphous calcium phosphate.
Significant differences between the values marked with the same symbol (P<0.05, Friedman's ANOVA).
NUMBER OF ENAMEL DEFECTS (per mm2)

Values are means ± SD.
CPP-ACFP = casein phosphopeptide–amorphous calcium fluoride phosphate; CPP-ACP = casein phosphopeptide–amorphous calcium phosphate.
P<0.05 (Friedman's ANOVA) between the groups marked with the same symbol.
The EDS analysis (Tab. III) did not show differences in Ca/P, Ca/O and P/O ratios between the treatment groups (P>0.05, Friedman's ANOVA). Fluorine was detected in 1 sample in the CPP-ACFP group.
EDS ANALYSIS OF ENAMEL SURFACES
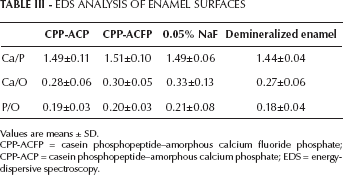
Values are means ± SD.
CPP-ACFP = casein phosphopeptide–amorphous calcium fluoride phosphate; CPP-ACP = casein phosphopeptide–amorphous calcium phosphate; EDS = energy-dispersive spectroscopy.
Microhardness test revealed significant effects of pastes containing CPP-ACP or CPP-ACFP (Tab. IV). No differences in VHN values were noted between the 3 experimental groups (P>0.05, Friedman's ANOVA).
ENAMEL MICROHARDNESS

Values are mean VHN ± SD.
CPP-ACFP = casein phosphopeptide–amorphous calcium fluoride phosphate; CPP-ACP = casein phosphopeptide–amorphous calcium phosphate; VHN = Vickers hardness number.
P<0.05 (Friedman's ANOVA) between the groups marked with the same symbol.
Discussion
In vitro studies are the commonly used as testing models in dental research. The major advantage of in vitro models is the ability to perform single-variable experiments in a highly controlled environment (13). The pH cycling system (chemical caries model), such as that employed in the present study, has become the model of choice to evaluate the caries process and caries preventive measures. Although it is not possible to completely simulate the complex biological aspects of caries, laboratory models are still of great importance for caries research.
Substrates used for in vitro caries models comprise enamel or dentine slabs. To reduce specimen variability and to achieve more reliable results, it has been recommended to use single sections of caries lesions (13). Therefore, in the present study, each demineralized enamel slab was divided into 4 parts which were randomized into 3 experimental and 1 control group. When evaluating novel materials, it is recommended to include a proven agent in the experimental design. The remineralizing potential of fluoride mouthrinses has been clearly described (12), so 0.05% NaF mouthrinse was used in this study as a well-known and the most commonly used everyday remineralizing agent.
Although the anticariogenic potential of CPP-ACP nanocomplex has been documented in laboratory, animal and human in situ caries models, thus far there has been no study concerning surface characteristics of demineralized enamel after treatment with CPP-ACP/CPP-ACFP. Oshiro et al (14) evaluated the effect of CPP-ACP paste on the demineralization process in sound enamel. Field emission-SEM revealed significant demineralization of nontreated samples, which was more pronounced with the longer test period. On the other hand, enamel treated with CPP-ACP paste twice a day showed slight morphological changes during the 28-day experimental period. When examined with atomic force microscope, CPP-ACP paste was shown to reduce the depth of the interprism cavities formed during the acid attack (15).
The image analysis technique permits reliable measurements and can produce detail and objective data through assessment of a great number of variables (16). The present study showed significant alterations in enamel surface morphology after treatment with paste containing either CPP-ACP or CPP-ACFP, with a reduction in enamel defects' size. These results can be regarded as indicators of enamel remineralization and are consistent with findings that CPP-ACP and CPP-ACFP enable growth of partially demineralized hydroxyapatite crystals (7).
The EDS analysis showed a slight increase in Ca/P ratio in all treatment groups, with the highest values in the CPP-ACFP group. Reynolds (17) also reported an increase in Ca/P ratio in demineralized enamel after 10-day treatment with various CPP-ACP solutions, which was attributed to the mineral deposition in hydroxyapatite form. In the present study, fluorine ions were detected in only 1 sample. However, this result should be treated with caution. As a semiquantitative analytical method, EDS exhibits certain limitations, especially for the analysis of the elements present in relatively low concentrations. We performed the standard examination under 20 kV acceleration voltage. Under these operating conditions, EDS has a detection limit of approximately 0.1% mass fraction of an element (18), and fluoride content of enamel is nearly at or below this level (19). Therefore, it can be presumed that it was not possible to detect fluorine ions although they might have been present at the surface of the enamel.
It has been acknowledged that mineral gain or loss in enamel as a result of demineralization and remineralization can be measured as microhardness change (20). There is no standard condition for enamel and dentin microhardness testing. Indentation microhardness testing with either Vickers or Knoop indenter has been used for the measurement of hard tooth tissues' microhardness. It has, however, been proposed that the Vickers test is more useful in enamel microhardness studies (21). In the present study, positive changes in surface microhardness were recorded after treatment with paste containing CPP-ACP or CPP-ACFP. Rehder Neto et al (22) evaluated the effect of different calcium-containing remineralizing agents on enamel microhardness. They reported a significant effect of the tested agents when compared with control demineralized enamel, with calcium sodium phosphosilicate being the most efficient. In addition, similar responses were noted in the CPP-ACP and CPP-ACFP groups, which is in accordance with the results of the present investigation.
In an in vitro study, Cochrane et al (7) demonstrated that CPP-ACFP solutions produced greater remineralization than the CPP-ACP solutions, especially at pH values of 5.5 and below. Kumar et al (23) also reported that paste containing CPP-ACP exhibited greater remineralization potential when used in combination with fluoridated toothpaste than when used alone. The difference in remineralization between CPP-ACP and CPP-ACFP was attributed to the presence of fluoride and its capacity to improve generation of fluoroapatite. Although SEM/EDS and microhardness analyses in the present study did not show differences between the CPP-ACP and CPP-ACFP groups, the fact that the paste containing CPP-ACFP reduced the number of enamel defects could be regarded as an indication of its greater remineralization potential.
Within the limitations of the present study, pastes containing CPP-ACP or CPP-ACFP showed the potential to remineralize enamel surface lesions. The remineralization effects of CPP-ACP and CPP-ACFP were found to be comparable, and greater than the remineralization effect of 0.05% NaF.
