Abstract
Keywords
Introduction
Biomaterials of natural origin such as proteins or polysaccharides are being widely studied because they do not release cytotoxic substances during their degradation, 1 allowing their use in medical areas such as tissue engineering. The regeneration of bone tissue in the last decades has been based on three-dimensional scaffolds with structures similar to the extracellular matrix of bone. 2 Natural polymers have been used on these structures due to their biocompatibility, biodegradability, and bioactivity. 3 Such is the case of chitosan that is a non-toxic material which has functional groups of hydroxyl, carboxyl, and amines that helps the bondage with biological particles that promotes the regeneration of bone, 4 allowing its functionalization by mixing other polymers or compounds to improve their properties such as the case of poly (DL-lactic-co-glycolic) (PLGA).5–6 PLGA contains groups such as lactic acid and glycolic acid monomers. 7 The mixture of chitosan with PLGA produces compounds that exhibit complementary properties. The low mechanical resistance of the chitosan is improved by the PLGA presence and this compound can be used as a vehicle for the supply of drugs and proteins. Additionally, PLGA and chitosan are Food and Drug Administration (FDA) approved.8–10
To obtain a better osteogenic activity and biocompatibility in the polymeric compounds, extracts of natural plants can be added, which help to enhance the bioactivity with the formation of hydroxyapatite on the surface of biomaterials.
11
In this work,
The
Materials and methods
Allium cepa extract
The
Synthesis of scaffolds
Different scaffolds were prepared: Chitosan (Ch), PLGA (P), Chitosan + PLGA (ChP), Chitosan +
The lyophilized composites were immersed in absolute anhydrous ethanol (J.T. Baker) for 12 h at a temperature of −4°C. The ethanol was removed by decantation. Then, they were immersed in a 0.5% w/w NaOH (JT Baker) solution for 12 h at −4°C. Subsequently, the NaOH solution was removed and samples were allowed to dry. To cross-link the samples, a solution of genipin (98% CBC) 10 millimolar was prepared using concentrated ethanol, which was added to the composites for 10 min and five times rinsed with ethanol. 25 The pH of the rinse water was measured to ensure NaOH removal and final neutralization. Finally, samples were lyophilized for 12 h.
In vitro bioactivity
To evaluate the apatite mineralization capacity of the scaffolds, a simulated body fluid (SBF) containing 1.5 times the concentration of ions in human blood plasma was used. This solution was prepared according to the methodology described by Oliveira et al. 26 Samples of 0.5 cm × 0.5 cm were immersed in 5 mL of SBF (pH of 7.4) in an incubator at 37°C during a period of 7, 14, 21, and 28 days. The SBF fluid was weekly changed in all samples. At the end of each period, the samples were rinsed with deionized water and dried. Three samples were made for each condition.
Characterization
The scaffolds morphology was analyzed using a Scanning Electron Microscope (SEM JEOL JSM-7000F). An energy dispersion system EDS 7557 INCA Oxford Instruments was used to analyze the dispersion and distribution of apatite particles and to detect the concentration of calcium and phosphorus in the surface after being exposed to the SBF during different periods of time, determining the Ca/P ratio. The average pore size was measured using the Scandium Universal SEM Imaging Platform software. At least 300 pores per sample were measured. The chemical characterization was performed by FTIR (Nicolet 6700, Thermo Scientific, USA). All spectra were recorded using 100 scans and 16 cm−1 resolution. All the samples were scanned within the range of 550–4000 cm−1.
Results and discussion
Figure 1(a)–(e) shows the obtained morphology of the prepared scaffolds where a porous structure is observed in all materials. Figure 1(a) and (d) show similar pore sizes 101 ± 40 and 106 ± 48 μm respectively, (see Table 1). No significant differences were found. Although there are not reported results of this type of material, studies were found where the pore size of chitosan increases with the addition of

SEM images of scaffolds: (a) Ch, (b) P, (c) ChP, (d) ChAC, and (e) ChPAC.
Average pore size and porosity distribution of the different scaffolds.

With the objective to improve the bioactivity, the
Figure 2 (a–e) shows the scaffolds surfaces obtained after their exposure in the SBF solution during 7, 14, 21, and 28 days. It is observed that after the first week, there is apatite crystals formation. Precipitated particles can be seen on the surfaces of the different scaffolds. According to the SEM image, these precipitates cover more samples surface within the pass of the time. In Figures 2(a) and 2(c), the formation of crystals is attributed to the salts contained in the SBF solution, due to the ability of the chitosan to contribute to this growth. Deposition of the apatite layer in the scaffolds can be attributed to the presence of deprotonated and negatively charged carboxyl groups (−COOH), 14 which can cause electrostatic attraction of Ca+2 ions from the SBF, as an initial step for the nucleation that is completed with PO4−3 ions, which leads to spontaneous growth and transformation into apatite crystals similar to the natural bones growth through the consumption of Ca+2, PO4−3, OH−, and CO3−2 ions from the SBF solution. 32 However, the presence of the crystals at the sample surface, according to SEM images, is less evident after 7, 14, and 21 days in the ChP scaffold (Figure 2(c)) concerning that of Ch scaffolds (Figure 2(a)). This is because the PLGA has low surface bioactivity during the first weeks (Figure 2(b)), so when mixed with chitosan slows down this mineralization process. Similar results have been reported when PLGA is added to calcium silicate and bioactive glass.33,34 It was found that the bioactivity is lost, but it helped to obtain better mechanical resistance.

SEM Mineralization of apatite in vitro in 7 (1), 14 (2), 21(3), and 28(4) days in SBF. (a) Ch, (b) P, (c) ChP, (d) ChAC, and (e) ChPAC.
When the extract of

EDS energy dispersion analysis of

A scheme of the mechanism of apatite formation. Calcium and phosphate ions contained in the SBF solution begin to form prenucleation crystals, these iones and prenucleation crystals are attracted to the polar surface groups of the scaffolds, depositing biological apatite on the surface.
Previous studies have shown that the silanol (Si–OH) radicals and organosulfur compounds help to have a surface susceptible to the nucleation and growth of calcium phosphate in the presence of simulated body fluid. Their polar groups increase the affinity of the silicate ions, which provide specific sites for the apatite nucleation, simultaneously joining the calcium and phosphate ions, which leads to an accumulation of the ions on the surface, inducing the formation of apatite or precursors of it (Figure 4).
37
The same mechanism is proposed for this work, where the compounds that form the sulfur and chlorine contained in the simulated fluid, form the polar groups on the scaffold surface and attract the ions of Al and Si contained in the
Ca/P atomic ratio of the surface of HA after soaking time in SBF.

Figure 5 shows the EDS obtained for the ChAC and ChPAC scaffolds in the first and last week, where calcium and phosphorus are identified. The presence of these elements is related to the formation of calcium phosphate phases. 35 There are also elements such as carbon, oxygen, sodium, aluminum, chlorine, and magnesium, which are part of SBF preparation. As it is well known, Martel et al., not only the calcium ions promote the apatite formation but also other elements such as chlorine, silicon, aluminum, and sulfur in the system are consumed forming carbonated apatite that enhances the rapid formation of the biological apatite evidenced by the Ca/P ratio (previously mentioned) obtained in the scaffolds. 22

Analysis by dispersion of energy (EDS) in a time of 7 and 28 days: (a) ChAC and (b) ChPAC.
In previous studies, it has been reported that during the process of formation of apatite layers on the surface of scaffolds containing large amounts of Ca and P, Ca can be replaced by OH, CO3−2, Na+, K+, and Mg+2 ions of the solution, obtaining biological apatite.
41
In this work, the obtained Ca/P values (from 1.6 to 1.67) on the surface of the scaffolds are very close to the real bone Ca/P ratio, so it is considered that the
The FTIR spectra of the obtained scaffolds are shown in Figure 6. The characteristic bands of chitosan are observed at 1665 cm−1 (stretch of C=O), 1587 cm−1 (NH flexion), 1379 and 1433 cm−1 (flexion of COOH), 1157 cm−1 (CO bonds), and 1030 cm−1 (COC stretch).
40
While for the PLGA, there is a stretching of the COC band at 1175 cm−1, the CH bands are present at 1390 and 1440 cm−1 and at 1760 cm−1 the band C=O is found.
41
When PLGA is added to Chitosan, it is observed that the intensity of the COOH and NH bands decreases, but the bands increase when the

FTIR of the scaffolds obtained prior to the contact of SBF.
Figure 7 shows the FTIR spectra of the scaffolds after immersion in the SBF during a time of 7, 14, 21, and 28 days. Analyzing ChAC and ChPAC scaffolds, the presence of PO4−3 and CO3−2 groups can be observed from the first to the fourth week (Figure 7(a)) on both scaffolds,
43
where the formation of phosphate and carbonate crystals is favored. These results support the capacity for biomineralization observed by SEM where it was observed that the surface is completely covered with calcium phosphate crystals after 21 days. The band present at 936 cm−1 is related to the symmetric stretching vibrations of PO4−3 and the peaks at 1020 and 1081 cm−1 are attributed to the asymmetric stretching vibrations of PO4−3.
31
Also, the characteristic bands of CO3−2 were observed at 1453 cm−1, which are due to the elongation of C–O by the substitution of carbonate ions in the structure of apatite.
39
Some authors reports showed similar results where the intensity of the peaks appearing at 1060 and 1100 cm−1 is attributed to the Si–O–Si stretching vibration,
40
which after a prolonged time in contact with SBF serve as apatite promoters, which grows in successive layers and completely covers the surfaces of the samples. The peak at 950 cm−1 is characteristic of the Si–OH stretch vibration of the silica phase,
40
this decrease with the immersion time in SBF, as the biological apatite is formed. The PLGA scaffold shows the same hydroxyapatite characteristics band. Only, in the third week, the PO4–3 group band increases. This is in agreement with the SEM images, where the low capacity of the PLGA for bioactivity is observed. The behavior of chitosan remains constant from the first week since the representative bands of PO4−3 and CO3−2 groups can be seen. They present only a slight increase in intensity in the third week. In the ChP sample infrared spectrum, it is observed that the intensity of the bands of the phosphate and carbonate groups in the first week decreases concerning the spectrum of chitosan alone, but this increases in the second and fourth week, which coincides with the SEM images obtained. Regarding to the ChAC scaffolding, it can be observed that in the second and fourth week, the bands of the groups PO4−3 and CO3−2 are intensified, which could indicate that the extract of
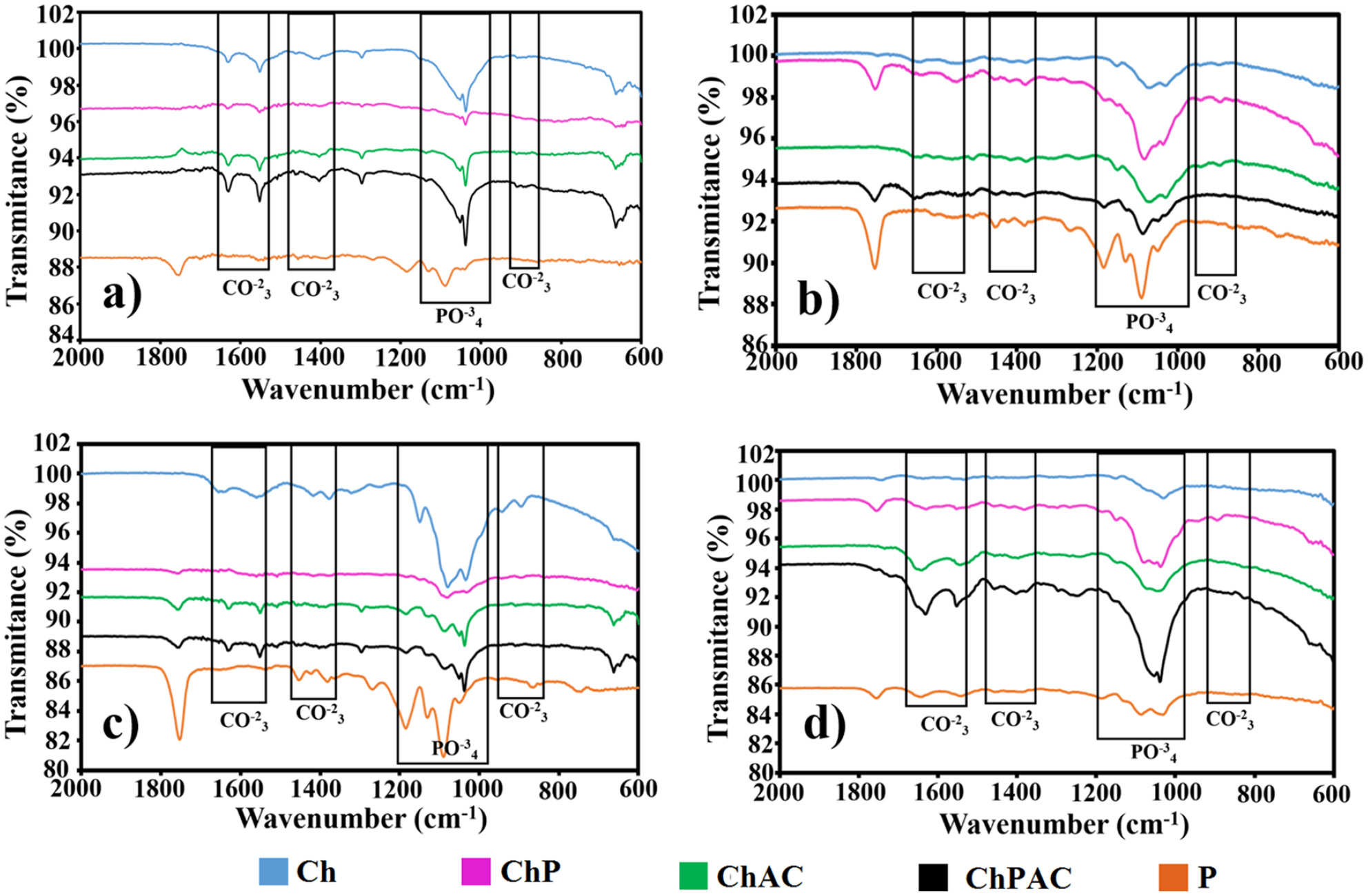
FTIR of the scaffolds after 7 (a), 14 (b), 21 (c), and 28 (d) in contact with SBF.
Conclusion
Three-dimensional scaffolds base chitosan/PLGA/
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the financial support of the Mexican Public Education Secretary (SEP) through the Teacher Professional Development Program (PRODEP) and Technology (CONACyT) through project SEP-CONACyT CB 2015-01-252439.
