Abstract
The current study was conducted to test the early osteogenic potential of strontium (Sr) doped titanium-aluminum-vanadium (Ti-6Al-4V) alloy using an in vitro cell culture experiment. Thirty Ti-6Al-4V alloy sheets were sandblasted and etched with large grit acid. Of these, 10 alloy sheets represented group I, 10 sheets doped with Sr using a hydrothermal process represented group II, and 10 sheets also coated with Sr-supplemented hydroxyapatite using a hydrothermal process represented group III. The surfaces of the three groups were characterized by scanning electron microscopy (SEM), energy dispersive spectroscopy (EDS), and X-ray diffraction (XRD). The three groups were also compared in terms of their water contact angle, protein adsorption, and Sr ion release profile. Further, bone marrow mesenchymal cells (BMSCs) obtained from the femora of 10 Sprague Dawley rats were used for the in vitro cell culture experiment, and the viability of the cultured cells was evaluated using the MTT assay and confocal microscopy; in addition, their osteogenic potential was assessed using alkaline phosphatase ALP activity. For statistical analysis, data were analyzed using one-way analysis of variance (ANOVA; IBM SPSS statistics 23), and p < 0.05 was considered statistically significant. SEM images revealed that the three groups had surface roughness, and EDS and XRD revealed the success of incorporating Sr to their surfaces. Group III had the best contact angle, protein adsorption, and Sr ion release rate. The cell culture also revealed that the surfaces of the titanium alloy sheets in group III were the most viable and had the best osteogenic potential; however, there was no statistically significant difference between groups I and II. In Conclusion, Sr alone was not able to improve the osteogenic potential of titanium alloy surfaces.
Introduction
Strontium (Sr) has been found to improve the osseointegration and surface mechanical properties of titanium implants, decrease bone resorption, and provide antimicrobial resistance.1,2 Various methods exist for adding Sr to the surface of a titanium alloy2–5—for example, micro-arc oxidation 3 ; electrochemical deposition 4 ; laser ablation at low pressure 5 ; loading to titanium nanorods synthesized by electrochemical anodization 6 ; alkali-heat treatment and magnetron sputtering 7 ; incorporation into fluorohydroxyapatite 8 ; plasma-spraying of Sr-reinforced hydroxyapatite 9 ; spin coating using 1% strontium nitrate (Sr(NO3)2) solution 10 ; surface application of organic–inorganic hybrid sol-gel coatings 11 ; heat sintering of Sr powder 12 ; or adding it hydrothermally by immersing the samples in a solution containing Sr followed by heating. 13 These coating methods revealed that Sr coating had an antimicrobial effect on Staphylococcus aureus, candida, and E. coli3,4 as well as improved the corrosion resistance of the titanium alloy4,9 and increased its microhardness.5,9,12 In addition, the long-term Sr ion release was found to promote osteointegration through improved and sustained osteoblastic cell differentiation.7,8,14
In vitro studies have found that Sr enhanced bone marrow-derived mesenchymal stem cells (BMSCs) migration, osteogenic differentiation, and gene expression, and promoted their angiogenic factor CD31expression, thereby leading to improved angio-osteogenesis, all of which improved the bone-to-implant contact.15,16 Sr was also found to improve early cellular attachment by enhancing the expression of cell attachment binding proteins (such as integrins); reduce intracellular oxidative stress; and enhance osteogenic potential as proved by the increased of alkaline phosphatase activity, collagen secretion, and osteocalcin production along with the suppression of osteoclastic activity and inhibition of adipogenesis through the suppression of adipokines expression.17–20 A confocal laser scanning microscope confirmed these findings through the study of cell behavior and morphology, which showed better early spreading, adhesion, proliferation, and osteogenic differentiation.21–28
Further, the histomorphometric analysis of bone-to-implant contact in in vivo studies revealed that Sr accelerated bone apposition, 28 increased peri-implant bone volume, 29 and increased bone-to-implant contact, which led to greater pull-out strength30,31 in normal and osteoporotic animal models; this, together with the ability of Sr to alleviate inflammation (as proved by immunohistochemistry results), made Sr surface coating a good candidate for use on implants in osteoporotic patients.32–35
However, there were a few other studies that did not support the abovementioned results, as Sr configuration (SrTiO3) was found to have inferior osteogenic function 36 and no effect on the proliferation or differentiation of BMSCs when incorporated in a calcium-strontium-zinc-phosphate (CSZP) coating. 37 A study that compared magnesium (Mg) to Sr, it was found that Mg had stronger osteogenic potential in vivo when implanted in rat tibia and a stronger effect on the same animal BMSCs as compared to Sr. 38 Another study, in tibiae of rats, compared a test group of sandblasted and acid-etched (SLA) Sr-coated titanium implants, and a control group of SLA implants with no coating, using histological analysis and mechanical removal torque tests—did not find significant differences between the study groups 39 ; this result was further confirmed by the findings of an additional study, using the same animal model, that implanted Sr-ranelate-coated titanium implants, compared them to implants with no coating, and also there was no histomorphometric analysis differences in the bone formed around the implants in the two groups after 2 and 6 weeks of healing. 40
Accordingly, the current work aimed to study the osteogenic role of Sr, where the Sr was incorporated into titanium-aluminum-vanadium (Ti-6Al-4V) alloy surfaces; moreover, uncoated titanium alloy surfaces and titanium alloy surfaces coated with hydroxyapatite supplemented with Sr were compared using in vitro studies.
Materials and methods
Ethical approval was obtained from the relevant institutional review board to conduct this study (IRB# Dent 85-24). Thirty sheets of Ti-6Al-4V of 10X10 mm length and width, and a thickness of 1 mm, polished manually by 2000 grits of SiC papers for approximately 5 min each; then, they were ultrasonically cleaned in a sequence of acetone, ethyl alcohol (70%), and distilled water for 15 min. To render the surfaces of the titanium alloy sheets rough, they were sandblasted and treated with large grit acid-etched implant surface (SLA) using combined acid etching of H2SO4 (48% Merck) and HCl (37%, Merck) at 60°C for 1 h; thereafter, they were cleaned of the acid traces by rinsing in alcohol and distilled water and were then sandblasted and cleaned again using the same protocol, before they were air dried.
The 30 titanium alloy sheet samples were divided into three groups: group I—10 SLA uncoated titanium alloy sheets; group II—10 SLA titanium alloy sheets that underwent hydrothermal treatment in a solution of Sr (OH)2 (Sigma-Aldrich, USA) to add Sr to their surface, thereby yielding SLA-Sr surfaces; and group III—10 SLA titanium alloy sheets hydrothermally treated with a hydroxyapatite (HA) solution supplemented with Sr2+ ions at a temperature of 200°C for 12 h, thereby yielding SrHA (Ca9Sr(PO4)6(OH)2) surfaces.
Surface coating characterization was conducted using scanning electron microscope (LEO 1450VP), energy dispersive spectroscopy (EDS), and X-ray diffraction (XRD, D8 Advance, Bruker, Germany) to study the surface elemental composition. The three groups were also compared in terms of their water contact angle (ca), which was measured using a goniometer; affinity for protein adsorption; and strontium ion release profile, which was determined by inductively coupled plasma-mass spectroscopy after 3 and 7 days of culture.
In vitro cell culture experiments were conducted using third passage BMSCs obtained from the femora of 10 Sprague Dawley rats. The titanium alloy sheets in the three groups were first immersed for 24 h at 37°C in Dulbecco’s Modified Eagle’s Medium (DMEM; Figure 1(a)) to facilitate the culturing of the BMSCs (Figure 1(b)) using a density of approximately 2 × 104 cells/mL in a CO2 incubator; moreover, the viability, attachment, and proliferation of the cell on titanium alloy sheets were assessed using an MTT assay (MTT, Sigma, USA).

(a) Titanium sheets immersed for 24 h in DMEM at 37°C and (b) the cell culture procedure.
To study cellular adhesion in each group, the cells were washed thoroughly with PBS, fixed for 10 min in 4% paraformaldehyde (PFA), and then for 15 min in 0.1% Triton™ (X-100 in PBS), and finally stained with DAPI-FITC-phalloidin for an additional 5 min. The confocal laser scanning microscope (CLSM, FV3000, Olympus, Japan) was used to image the red fluorescence of the cytoskeletal F-actin and the blue fluorescence of the cell nuclei. To study the osteogenic potential of Sr, the BMSCs were cultured on the titanium alloy sheets in the absence of osteogenic induction supplements; thereafter, the alkaline phosphatase (ALP) staining and activity were observed using a stain kit (BCIP/NBT ALP) after 3 and 7 days of culture.
All the resulting data were analyzed using one-way analysis of variance (ANOVA; IBM SPSS statistics 23), and differences were considered statistically significant at p < 0.05.
Results
Samples surface characterization
Figure 2(a) depicts an SEM image of group I (SLA) with distinct acid etching and sandblasting roughness. In Figure 2(b), the effect of incorporating Sr on the surfaces of the titanium alloy sheets in group II (Sr-SLA) is noticeable, as the surfaces are still rough; however, these became smoother at the nanometer level.
Figure 3 depicts the surface of the titanium alloy sheets in group III after hydrothermal coating with SrHA. It is evident that the surface is dense, rough, and characterized by rounded edges and polygonal structures (which are believed to be the hydroxyapatite crystal coalesced due to the incorporation of Sr); however, the surface is still rough with nano-sized pores.
In Figure 4(a), the EDS reveals the surface elemental composition. Sr ions were successfully doped onto the surfaces of the titanium alloy sheets in groups II and III, and the EDS spectrum seen in Figure 4(b) reveals the even and homogenous distribution pattern of Sr, with a percentage of 1.1 ± 0.1 weight percent (wt%) in group II and 1.28 ± 0.1 wt% in group III. The difference in wt% between groups II and II was statistically significant (p ˂ 0.05) and could be explained by the substitution of Sr2+ for Ca2+ (Table 1). In addition to EDS, XRD was used to investigate the crystalline phase structure of the surfaces of the titanium alloy sheets in the three groups. As evident in Figure 5, group I had Ti, O, Al, and V and Sr was added to these elements in group II; however, group III had HA with more Sr than that in group II, as identified by the diffraction peaks for HA-Sr.
Statistically significant differences (p < 0.05) were found between the water contact angles of the surfaces of the titanium alloy sheets in the three groups, as depicted in Figures 1 and 2. In group I (SLA), the surface contact angle was 10.43° ± 0.29°; in group II (Sr-SLA), the surface contact angle was 8.73° ± 3.09°; and group III (SrHA), the surface contact angle was 6.63° ± 3.16°. However, all these values indicated a high degree of hydrophilicity; this implied better wettability, which could be attributed to increased surface roughness (Figure 6(a)).
A protein adsorption test revealed that the surfaces of the titanium alloy sheets in group III (SrHA) (p < 0.05) adsorbed significantly more protein (15 ± 1.4 µg/cm2) than those in groups I (SLA; 3 ± 1.01 µg/cm2), and II (Sr-SLA; 2 ± 1.06 µg/cm2); however, no significant differences, in protein adsorption, were detected between groups I and II (Figure 6(b)).
Group III had a significantly higher Sr ion release profile on both the third and seventh days (Figure 6(c)). On the third day, the Sr released in group II (2.56 ± 0.07 mg/L) was lower than that in group III (8.6 ± 1.2 mg/L); by the seventh day, the total amount of Sr released from group II was approximately 11.1 ± 24.0 mg/L, while that from group III was 27.53 ± 2.0 mg/L.

(a) SEM image of the SLA treated titanium sheets. (b) SEM image of group II (Sr-SLA), with Sr added to the SLA treated titanium sheets surface resulting in a smoother surface, however, with rough nodular structures, and porosity on a sub-micron scale.

SEM image of group III (SrHA), shows the surface of the Sr doped hydroxyapatite, with no cracks, indicating the success of the slow hydrothermal coating process, which created a dense, rough coating, exhibiting rounded edges polygonal structures, and sub-micron scale porosity.

(a) EDS of groups II and III surfaces and (b) EDS spectrum of Sr in groups II and III.
Surface elemental composition as determined by EDS.


XRD patterns of groups I, II, and III. The XRD demonstrated the characteristic peaks of strontium in groups II and III at different angles due to the difference in surface crystalline structure.

(a) Water contact angle, (b) protein adsorption, (c) strontium ion release profile. Data represent the mean ± SD (columns with whiskers) of 3 tested groups; *p < 0.05.
Cell viability, attachment, proliferation, and osteogenic potential
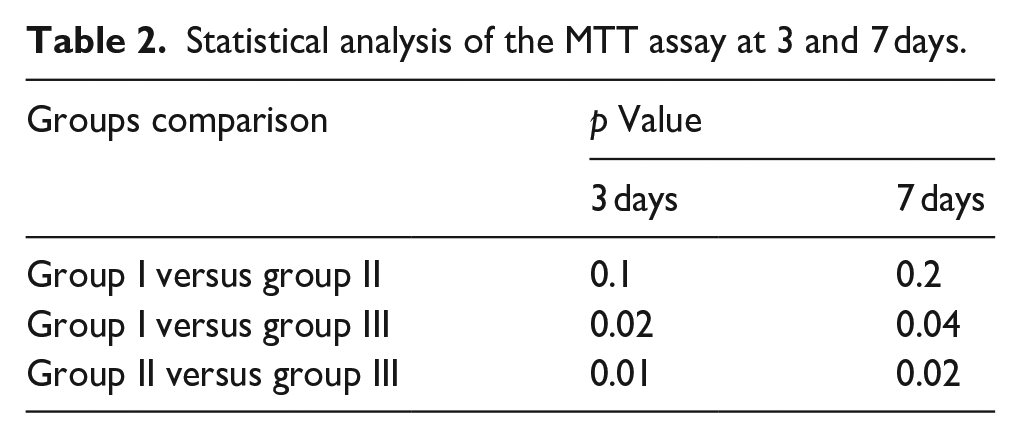
Based on the MTT assay results, presented in Figure 7 and Table 2, after 3 and 7 days of seeding the BMSCs, no cytotoxicity was detected for any of the surfaces of the titanium alloy sheets in the three tested groups, however, there was a significantly increased cellular spread, proliferation, and viability on the surfaces of the titanium alloy sheets in group III as compared to those in groups I and II.
After 7 days of culture, the CLSM images, depicted in Figure 8(a) and (b), revealed that the cells on the surfaces of the titanium alloy sheets in group III had significantly (p < 0.05) more adherence, higher density, clearer nuclei, and more distinct cytoskeletons and membranes as compared to the cells cultured on the surfaces of the titanium alloy sheets in groups I and II, which did not differ significantly from each other (Table 3).
On the third and seventh days of culture, the ALP activity on group III sheets revealed more pronounced dark purple staining, as compared to that in groups I and II, however, no significant difference was found between groups I and II as related to their ALP activity (Figure 9(a) and (b), and Table 4).
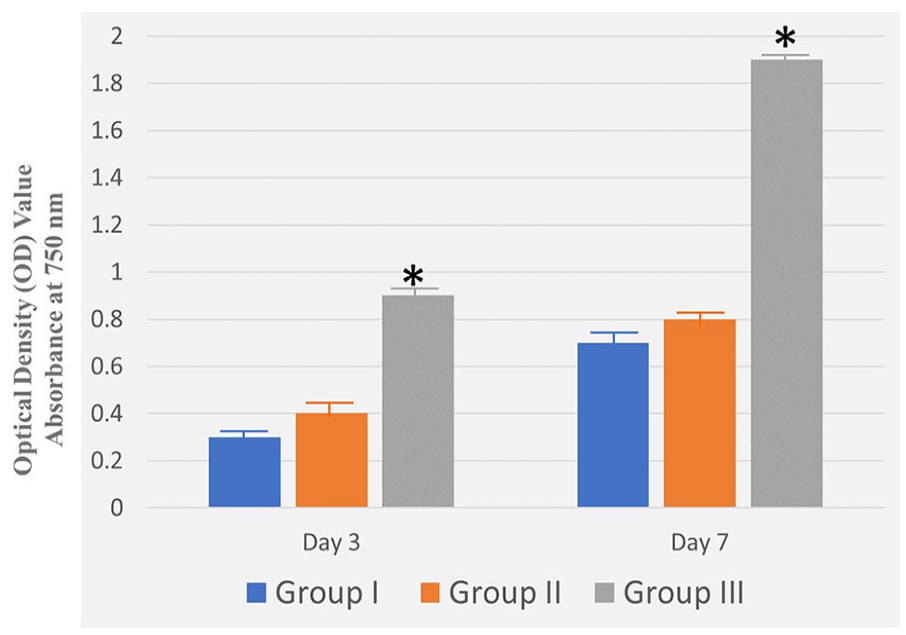
Cellular viability on the surfaces of groups I, II, and III based on the MTT assay results. Data represent the mean ± SD (columns with whiskers) of 3 tested groups; *p < 0.05.
Statistical analysis of the MTT assay at 3 and 7 days.

(a) Confocal laser scanning microscope (CLSM) images of BMSCs cultured on group I, II, and III surfaces. (b) Fluorescence of the F-actin area stained in each group at 3 and 7 days. Data represent the mean ± SD (columns with whiskers) of three tested groups; *p < 0.05.
Statistical analysis of F-actin area stained in each group at 3 and 7 days.


(a) Alkaline phosphatase (ALP) staining. (b) The ALP activity of BMSCs cultured on group I, II, and III after 3 and 7 days of culture. Data represent the mean ± SD (columns with whiskers) of three tested groups; *p < 0.05.
Statistical analysis of ALP activity in the three groups at 3 and 7 days.

Discussion
Considering the different sample preparation techniques used to add Sr to titanium alloy surfaces, several studies used the hydrothermal method—which is similar to the technique used in the current study—and concluded that Sr was able to improve the osteogenic potential of the titanium surface.6,7,10–12 In addition to the hydrothermal method, other preparation techniques led to the same conclusion—for example, micro-arc oxidation,3,30,31 magnetron sputtering,7,14,21, and evaporation-induced self-assembly method. 18 However, a few other preparation techniques did not report any osteogenic potential of Sr, such as atmospheric plasma spraying, 36 phosphate chemical conversion (PCC) technique, 37 magnetron sputtering, 39 and the evaporation-induced self-assembly method (used in our previous study on the long-term osteogenic effect of Sr, which could not detect any statistically significant difference between the Sr-doped surface and control surface of titanium, employing a histological analysis 40 ). Similarly, the current study did not find any significant difference between groups I (the non-coated titanium alloy surfaces) and II (the Sr-doped titanium alloy surfaces) in terms of improving cellular attachment, proliferation, and osteogenic differentiation.
However, in this study, when Sr was included in the hydroxyapatite coating of surfaces in group III, statistically significant results were documented with regard to improved cellular adhesion, spread, and osteogenic differentiation as compared to that in groups I and II. This finding was in agreement with those of several other studies that included Sr in the hydroxyapatite,5,9,13,17, fluorohydroxyapatite, 8 or Sr and Mg into the hydroxyapatite. 4 Other studies reported the success of Sr in combination with other trace elements, such as silver, silicone, 3 zinc, magnesium, 33 calcium, phosphorus, cobalt, and fluoride 30 ; however, Sr was found to have lower osteogenic potential than Mg. 38 indicating that Sr needs to work in concert with other materials or elements to enhance the osteogenic potential of BMSCs.
Comparing the results of the current study to that of other studies, it was found that the acid etching and sandblasting provided roughness that was similar to that attained in Zhou et al.15,20 and Geng et al. 26 ; this was useful for the attaching of BMSCs, as reported by Zhang et al., 38 micro-mechanical interlocking for the SrHA coating, and decreasing the water contact angle that improved the wettability and protein adsorption. However, this study found that the incorporation of Sr in the surfaces of the titanium alloy sheets in group II made their surfaces smoother; in group III, this incorporation smoothened the surface of apatite crystals, but the surface was solid with no cracks, a finding that could be attributed to the slow hydrothermal process that created a dense surface similar to that found by Tam et al. 13
Tam et al. 13 and Zhou et al. 20 also used EDS and XRD to study the chemical composition of the surfaces. Similarly, the current study reported a successful incorporation of Sr onto the surfaces of the titanium alloy sheets in groups II and III, with more Sr in group III than that in group II. According to Zhang et al., 38 the release of Sr ions probably enhanced the biological response of the BMSCs. Moreover, the results of this study found that the release of Sr ions in group III was significantly higher than that in group II, which was probably due to the higher content of Sr in group III and its easier release from SrHA than from Sr bound to the surfaces in group II. In agreement with the results of Tam et al., 13 the MTT assay and cell culture study of the current study revealed that the cells in group III (SrHA) exhibited a more biocompatible pattern than the Sr surfaces in group II, which did not differ from the uncoated surfaces in group I; this finding was in contrast to that of Zhou et al., 15 who found that the cells on the Sr surfaces had more activity than those on the control surfaces. In addition, Zhou et al. 15 and Geng et al. 26 reported a more osteogenic potential of Sr surfaces, as indicated by the ALP activity, than those on the control surfaces; however, the current study did not yield such a result since group II did not statistically differ from group I, as reported by the ALP activity assessed by the ALP BCIP-NBT staining assay on the third, and seventh days after culture. Based on the findings of the current study, we were unable to confirm the osteogenic potential of Sr due to its inability to enhance osteogenic potential in group II as compared to group I, however, in respect to the contradicting findings of other mentioned studies, consideration to the method of Sr doping to the surface, and the amount of its release must be given in future studies to confirm its osteogenic potential.
In conclusion, the current study did not find any statistically significant differences between the Sr-treated titanium alloy surfaces in group II as compared to the untreated titanium alloy surfaces in group I; however, the Sr-osteogenic potential was significantly improved when added to hydroxyapatite in group III as compared to that in groups I and II. These findings are in contrast to those of several previous studies that reported the osteogenic potential of Sr. Though, it is worth mentioning that the current study had several limitations, which We encourage other researchers to overcome in the future if they decide to conduct further studies on strontium, for example, the use of different preparation techniques to add strontium to the surface titanium implants, with or without other elements. Additional surface characterization techniques could have also been used, like Raman Spectroscopy or Fourier transform infrared spectroscopy (FTIR). Different cells such as dental pulp stem cells, or MC3T3-E1 osteoblasts, could be used to further confirm the cellular attachment, proliferation and differentiation patterns, together with different cellular bioactivity characterization techniques, such as quantitative real-time polymerase chain reaction assays, and relative gene expression of Runx2, COL-1, ALP, OPG, OPN and OCN. Finally, in spite of the results of the current study, further studies are suggested to evaluate other advantages of strontium, such corrosion resistance, or antibacterial effect. Also, in vivo experiments of strontium doped implants, that could be characterized with were micro-CT, and HE staining to study bone area, bone–implant contact and removal torque values of such implants.
Conclusion
Taking into consideration the limitations of the current study, the following conclusions were drawn; The hydrothermal process succeeded in adding Sr to the titanium alloy surfaces in group II, and in the hydroxyapatite coating of III, based on the findings of the EDS and XRD. Strontium alone was not able to recruit more cells to the titanium alloy surfaces, and was unable to improve their osteogenic differentiation, as compared to the control surfaces; however, its osteogenic potential was improved when added to hydroxyapatite in group III.
Footnotes
Acknowledgements
The authors also would like to thank Dr. Yasmine Wahby for help with the cell culture and its characterization techniques. The researchers would like to thank the Deanship of Graduate Studies and Scientific Reasearch for financial support (QU-APC-2025).
Author contributions
All authors contributed to conception, study design, data collection, analysis, and interpretation, and drafted and critically revised the manuscript.
Data availability statement
The data will be available upon the request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
