Abstract
Background
The specific biological need of patients frequently becomes obvious just in the intraoperative setting. We hypothesized that a double-layer patch approach that allowed rapid attachment to an implant surface would represent a potential solution for technically challenging intraoperative personalized local drug delivery.
Methods
Dexamethasone-loaded poly[(rac-lactide)-co-glycolide] (PLGA) microparticles were embedded within a polyvinyl alcohol (PVA) patch that was attached to metal implant surfaces by in situ polymerization of alkyl-2-cyanoacrylates (CAs). Hydroxyapatite (HA) nanoparticles were also embedded in the PVA patch.
Results
Very rapid dexamethasone-release profiles were observed from the PLGA microparticles / PVA patches. The incorporation of HA nanoparticles into the PVA enabled control of CA penetration within the patch, and improved significantly its attachment, while no interference with the drug release was observed.
Conclusions
Double-layered patches with 1 layer for drug delivery and 1 as gluing interface could represent a solution for safe and controlled local drug delivery from implant surfaces or other, even biological, materials. The technology platform presented here opens the opportunity for personalized medicine by allowing local administration of drugs with customized release based on an intraoperative application.
Introduction
A number of clinical situations require local administration of drugs to enable or reinitiate healing (1), treat infections (2) or block pain associated with surgical interventions (3). Examples with a great need in health care are infections associated with implants (4), delayed healing situations in fracture treatment (5) and prolonged phases of pain following surgical treatment (6). The clinical problems involved are diverse, and there is no simple solution that likely suits all indications (7). In many cases, it would be highly desirable to intraoperatively customize the implants to enable local and controlled drug delivery for personalized patient treatment – e.g, in cases of total knee replacement surgery or tibia fractures. The existing medical device combination products approved by the US Food and Drug Administration (FDA) (8), as well as the current clinically approved methods to locally deliver drugs to bone injuries as scaffold-grafting materials, demineralized bone matrix, injectable biomaterials or implant coatings, do not meet such criteria. For example, bioabsorbable patches have been proposed for prevention of air leaks in pulmonary surgery (9), and as a safe hemostatic method against soft tissue bleeding (10). On the other hand, multilayered patches have been used in transdermal drug delivery, and polyvinyl alcohol (PVA) appears to be one of the useful polymers for such devices (11).
Because of these shortcomings, new intraoperatively customized implant coating strategies, such as sleeves, sticky strips and sticky drug-delivery patches, have been proposed (12). The use of polycyanoacrylates has already been tested and shown to be a very promising approach for rapid attachment of PVA-based drug-delivery patches to implant surfaces (13). It was shown that alkyl-2-cyanoacrylate (CA) polymerization can be initiated by water molecules absorbed by the PVA patch. PVA is a hygroscopic polymer with a semicrystalline morphology, which can be manipulated by physical methods such as freeze–thawing cycles (14). Cyanoacrylate adhesives have been extensively used in various clinical applications such as in thoracic, gastrointestinal, neurological, cardiovascular, ophthalmological and vascular as well as cartilage- and bone-grafting procedures (12), and it has been suggested they could be used to control the rate of drug delivery (15). At present, CAs have limited FDA approval for topical use as alternatives to sutures (16).
The shear strength of such PVA-based coatings depends on the type of CA, the time the patch is pressed against the implant surface, as well as the amount of humidity absorbed by the PVA patch (13). The CA monomers were shown to have penetrated and polymerized within the PVA without showing any negative effect on the bioactivity of embedded molecules and without fundamentally altering the release pattern of bovine serum albumin as a model protein (13). The combination of CA with the PVA patches also proved to be noncytotoxic in vitro (13).
Poly[(rac-lactide)-co-glycolide] (PLGA) particles are well-established carrier systems for various types of drugs and are suitable for modifying the release patterns of those drugs. A drug-delivery approach with dexamethasone-loaded PLGA microspheres and freeze–thawed PVA composite has already been tested for coating of sensors (17). In the current study, drug-loaded PLGA microparticles with burst dexamethasone release were embedded within a PVA patch to check for a possible retarding effect on the drug-release profile when the patch was attached over a surface by a CA adhesive. However, such an implant coating strategy still has the risk of highly reactive CA monomer penetration within the patch and possible interaction with the embedded drugs (13). Therefore, an additional approach for controlling the diffusion of CA monomers into the patch during the attachment procedure was required.
Hydroxyapatite (HA) nanoparticles have already been used to influence the morphology of PVA-based matrices and produce bioactive nanocomposite polymer hydrogels (18). Even though the dispersion of HA in polymer matrices can be a critical challenge (19), the formation of hydrogen bonding between the HA nanoparticles and PVA can allow a uniform distribution (20). The colloidal solution comprising PVA and HA nanoparticles has been reported to be processed as nanofibers (21) as well as membranes (22). For HA/PVA composite membranes, a reduced hydrophilicity due to the highly crystalline HA nanoparticles and their potential in guided bone regeneration have been reported (22). Therefore, we hypothesized that the addition of highly crystalline HA nanoparticles in the PVA patch would improve its properties, particularly by acting as a barrier to any unnecessarily deep penetration of CA monomers in the patch and subsequent polymerization in the proximity of the loaded drugs. At the same time, the immediate patch attachment to the metal surfaces, its solid attachment to implant surfaces and the drug-release profile should not be altered.
Accordingly, the influence of HA nanoparticles on the CA penetration within the patch, any potential patch detachment after swelling, the shear strength and the influence on the model drug-release profile were characterized. Double-layered PVA/HA patches were prepared with a maximum thickness of 60 µm, which, as a future platform, could accommodate drug-delivery particles of an approximately similar size. In this case, 1 layer (the drug-free layer) served as glue contact layer which came into contact with a CA adhesive and represented a barrier layer for penetration of CA monomers. A second layer (the drug-loaded layer) could be designed to allow the desired drug-release profiles that would not be influenced by the gluing to the implant surface. Ideally, the patch should stay attached to the implant surface covering it during the entire drug-release phase, followed by complete removal once it is no longer required. For this reason, materials with extended disintegration time were used: a particle-embedding PVA material in combination with a slowly degrading HA that reduced patch swelling and served as a barrier for CA monomer penetration. The PVA/HA combination could be especially useful with orthopedic implants, while other biomaterial combinations might also be used by this platform according the required indications.
Materials and Methods
Preparation of PVA patches with incorporated dexamethasone-loaded PLGA microparticles
Microparticles from PLGA with a glycolide content of 50 mol%, ethyl ester end-groups, a weight average molecular weight (Mw) of 24 kDa and a polydispersity (PD) of 2.1 (Resomer RG 503) were loaded with dexamethasone (micronized; Fagron, Barsbüttel, Germany) in an oil-in-water emulsion procedure (23). Briefly, 1 mL of 19 wt% PLGA solution in methylene chloride was supplemented with 28 mg of dexamethasone by sonication and emulsified in 2 mL of a 1 wt% solution of polyvinyl alcohol (Mw ∼31,000 kDa, DH ∼88 mol%; Mowiol 4-88; Kuraray) by rotor stator homogenization (24,000 rpm; Ultra-Turrax T 25; IKA). This was followed by solvent evaporation, lyophilization and characterization of particle size (d0.5 = 2.3 µm, span = 3.1; Mastersizer 2000; Malvern) for a final dexamethason payload of ∼84 µg/mg microparticles (encapsulation efficiency 84.2% ± 3.7%).
To prepare microparticle-loaded patches, 80 mg of microparticles with burst dexamethasone release was dispersed by pipetting into 10 g of 5 wt% PVA in aqueous solution (Mw ∼47000, DH 98.0-98.8 mol%; Mowiol 6-98; Sigma-Aldrich, Germany) (Fig. 1). The dispersion was then incubated on a tube roller (30 rpm) for 15 minutes at room temperature and followed by casting in the mold. To prepare dexamethasone-loaded PLGA microspheres/PVA composite films (17) with increased crystallinity (14), the films were treated by 3 repetitive cycles of freeze–thawing (2-hour freeze at -20°C/1-hour thaw) and then drying at room temperature. In contrast, to prepare nontreated films, the cast PVA/microparticle solution was dried at room temperature until a film was obtained. The obtained dry and brittle films (PVA/PLGA/dexamethasone) were then carefully punched to obtain standardized patches (“P8”) with a diameter of 8 mm. Then the PVA/PLGA/dexamethasone patches were glued over the sandpapered Titanium alloy-Synthes disks (to ensure that the patches would stay attached in a wet state) by 1 µl poly(n-butyl cyanoacrylate) (PBCA) after 1 minute of humidity exposure and 1 minute of pressing (13).
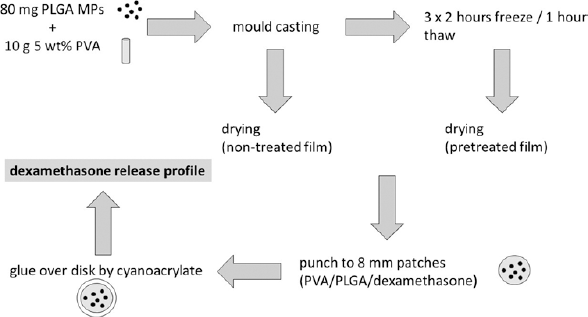
Work flow for preparation of polyvinyl alcohol (PVA) patches with incorporated dexamethasone-loaded poly[(rac-lactide)-co-glycolide] (PLGA) microparticles (PVA/PLGA/dexamethasone). MPs = microparticles.
Overview of pretreatment of samples for characterization (dexamethasone release not included)

HA = hydroxyapatite; PBCA = poly(n-butyl cyanoacrylate); PVA = polyvinyl alcohol.
Six samples of each group were each placed in 2 mL phosphate-buffered saline (PBS) (×1) and incubated in an oven at 37°C -38°C. Nonglued patches and microparticles alone served as control release groups. At days 1, 3, 5, 7, 14, 21 and 28, 1 mL of the PBS was collected and stored at -20°C, while 1 mL of fresh PBS was added to the samples. To evaluate the total amount of dexamethasone loading in the patches, they were dissolved in 2 ml of dimethyl sulfoxide (DMSO) in an ultrasound bath for 10 minutes. Then, 100 µL of the DMSO fraction was mixed with 900 µL of 93% ethanol for polymer precipitation and used for high-performance liquid chromatography (HPLC).
The amount of the released dexamethasone from the patches (glued or not), from the microparticles alone and from the dissolved patches was quantified by HPLC (1200 series HPLC; Agilent Technologies). On a 125-4 RP-18 column (LiChroCART® 125-4, LiChrospher® 100, 5 µm; Merck, Darmstadt, Germany), isocratic separation was performed at 30°C with a flow rate of 1 mL/min, 10-µL injection volume and 65/35 mixture of 0.1% trifluoroacetic acid in water and acetonitrile as eluents, with detection at 240 nm (retention time 4.5 minutes).
Preparation of PVA/HA patches
To eliminate the risk of CA monomer penetration within the patch, HA nanoparticles were incorporated into the PVA. Due to the crystalline nature of HA nanoparticles, which already imparts reduced patch swelling, no pretreatment by freeze–thawing was required. For that purpose, PVA (Mw ∼47,000, DH 98.0-98.8 mol%; Mowiol 6-98; Sigma-Aldrich, Germany) was suspended in water in an ultrasonic bath at 80°C for 5 hours until complete dissolution. PVA films with embedded HA nanoparticles (nanopowder <200 nm BET-equivalent spherical particle diameter; Sigma-Aldrich, Germany) were achieved by dispersing the HA in the PVA solution as described below. The first layer of the double-layered PVA/HA films (133/200 PVA/HA + 300/100 PVA/HA) was prepared by dispersing 200 mg of HA nanoparticles in 6.41 g of distilled water in an ultrasonic bath at 40°C for 15 minutes (Fig. 2). Then 886 mg of 15 wt% PVA solution (corresponding to 133 mg PVA) was added to the HA dispersion, followed by treatment in a ultrasonic bath at 40°C for 30 minutes and incubated on a tube roller for 15 minutes before casting in the mold (depth: 1 mm, area: 60 × 120 mm).
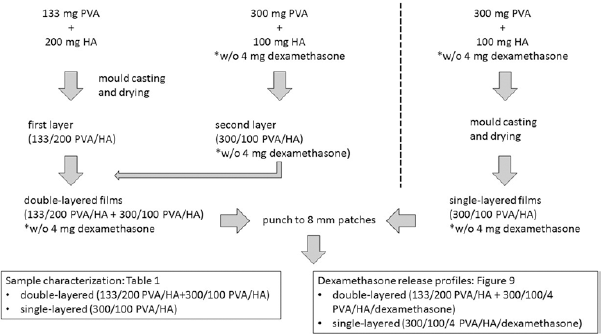
Work flow for preparation of the polyvinyl alcohol (PVA)/hydroxyapatite (HA) patches.
After overnight drying at room temperature and formation of the first layer (133/200 PVA/HA), a second layer of PVA/HA was cast. For this, 7.5 g of dispersion containing 300 mg/100 mg of PVA/HA was prepared according to the above-mentioned protocol and incubated on a tube roller for 24 hours before casting in the mold. Briefly, 100 mg of HA nanoparticles was added to 5.4 g of distilled water. They were then incubated in an ultrasonic bath at 40°C for 15 minutes. Then, 2 g of 15 wt% PVA solution (300 mg PVA) was added to the HA dispersion, followed by treatment in an ultrasonic bath at 40°C for 30 minutes and incubated on a tube roller for 24 hours. The dispersion was cast onto the first layer (133/200 PVA/HA) to achieve double-layered films (133/200 PVA/HA + 300/100 PVA/HA) with a thickness of 58.8 ± 3.9 µm. Thickness values were obtained by a magnetic thickness gage (Magna-Mike, Model 8000).
For single-layer film of 31.7 ± 4.6 µm thickness, a 7.5 g dispersion containing 300/100 of PVA/HA, respectively, was prepared in the same way as for the second layer of the double-layered film (Fig. 2).
PVA/HA films were punched to prepare patches “P8” and “P15” with standardized sizes of 8 and 15 mm diameter, respectively. P8 patches were used for scanning electron microscopy (SEM), Raman microscopy and wet detachment, whereas P15 patches were used for analyzing the shear strength (Tab. I).
For imaging of the patch morphology on a cross-sectional plane, patches were cut with a razor blade, coated with Ir and analyzed by scanning electron microscopy (Gemini Supra™ 40 VP; Carl Zeiss NTS).
Attachment of PVA/HA patches to test implant surfaces
Metallic implant surfaces, as they are used in trauma implants, were selected as test sample metal surfaces for the present analyses. The attachment capability of patches on these test implant surfaces has been described previously (13). Briefly, anodized titanium disks with an upper cylindric part 12 mm in diameter and 3.5 mm in height (gluing part), a lower cylindric part 15 mm in diameter and 3 mm in height (for sample fixation) and surface properties identical to standard osteosynthetic plates were produced and donated by Synthes GmbH, Switzerland. Disks with identical dimensions prepared from anodized titanium Ti-6Al-44 and tumbled/electropolished CrNi 1.4441 steel (donated by AAP Implantate AG, Germany) were used for comparison. Disks with these dimensions were chosen to fit the fixation holders of the pressing device and the tensile tester. They were also able to accommodate the attachment of the P15 patches for shear testing and P8 for the other testing. As CA monomers were used – (i) butyl-CA (Indermil, donated by P.J. Dahlhausen & Co. GmbH, Germany) forming PBCA by in situ polymerization, for patch attachment to the test implant surface, and (ii) methyl-CA (Loctite 496, Henkel; obtained from Conrad Electronics GmbH, Germany, as local distributer) forming poly(methyl cyanoacrylate) (PMCA) as internal control adhesive for shear testing.
Both the pressing device and the preparation steps have been described previously (13). Briefly, the test implant disks were fixed in the holder of a special pressing device (Fig. 3). Then, the PVA/HA patch was exposed to a controlled level of humidity (100% relative humidity) to absorb water molecules and therefore initiate the rapid polymerization of the CA adhesives. Meanwhile, 1 µL of CA adhesive was applied and spread over the desired disk surface, where the patch would be glued (Fig. 3). The humidified PVA patch (P8) was placed using tweezers onto the CA at the test implant surfaces, immediately followed by pressing with a constant weight of 2.5 kg using a silicone disk to allow for homogeneous force distribution (Fig. 3). One minute of pressing and 1 minute of humidity exposure were sufficient for the attachment of the patch. In cases of testing the shear strength, the humidified patch (P15) was placed onto a silicone disk that was fixed in the lower holder of the press. The larger patch diameter assured that the patch covered the whole disk surface to provide a geometrically defined setup for the shear testing. Five microliters of CA adhesive was spread over the disk fixed in the upper platform, which subsequently was pressed against the humidified patch (13).
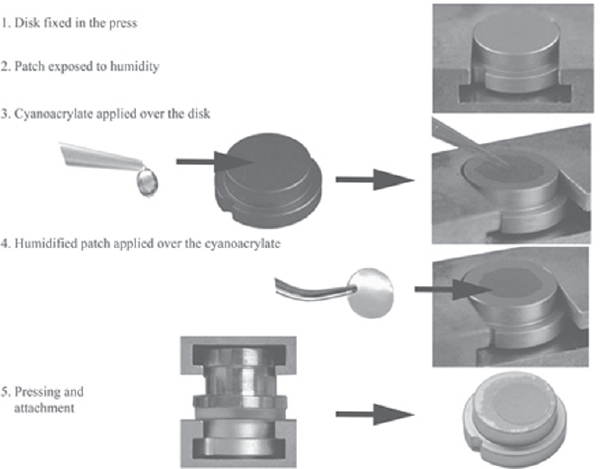
Work flow of coating procedure needed to apply the drug-delivery patch to the test implant surface. Parts of the figure are reproduced from (13) with permission from John Wiley and Sons.
If disks were reused, as done for certain groups, they were cleaned in an ultrasonic bath at 40°C as follows: (i) 15 minutes in acetone 99.5% (Sigma-Aldrich, Germany), (ii) 15 minutes in 2% Micro-90 (Sigma-Aldrich, Germany) and (iii) 15 minutes in ethanol 96%. To examine the wet detachment of the patches from the intact test implant surfaces (Tab. I), each sample was placed in 2 mL PBS (×1) and incubated in an oven at 37°C -38°C.
Shear strength of surface coating
The shear force is an important parameter for the quality of the patch attachment, and the procedure to perform the relevant mechanical test has been described elsewhere in detail (13). Briefly, additional disks were glued to the patch by PMCA (due to its having the highest adhesive strength) to the glued disk/patch construct. The P15 patch was larger than the test implant surfaces to avoid a direct glue interface between the 2 disks. This disk/patch/disk construct was placed in a custom-made socket (Fig. 4, left), which was inserted into a special holder (Fig. 4, right). Using a displacement rate of 1 mm/min by tensile tester (Zwick/Roell Z010, load cell Xforce K, Germany), the shear force and displacement were measured until failure of the construct occurred. The group tested included 6 samples (Tab. I).

Methodology for sample fixation for mechanical testing: the disk/patch/disk construct is placed in the socket (left) which is inserted in the holder (right). Then force is applied from above by a pin to the disk on the right side, while the left part of the device is rigidly fixed, leading to a well-controlled shear testing. Parts of the figure are reproduced from (13) with permission from John Wiley and Sons.
Characterization of PVA/HA patches by Raman spectroscopy
Due to their brittleness, the PVA patches with included HA nanoparticles were analyzed for CA monomer penetration within the patch, without being peeled off from the implant surfaces (Tab. I). Three samples per group were analyzed by depth profiling using Raman spectroscopy: A Bruker Senterra dispersive Raman spectrometer equipped with 2 lasers (785 and 532 nm) was used for that purpose. Using a ×50 magnification for the depth profiling, a step size of 5 µm for 12 steps (resulting in a depth of 60 µm; except the 30-µm samples, which involved 6 × 5 µm steps) was employed. The power of the laser was adjusted to 5 mW at a wavelength of 532 nm, and 128 scans were made with an aperture of 50 µm. For analysis of the Raman spectra, a CH-bond at 2,900 cm−1 for the PVA, a CN bond at 2,250 cm−1 for PBCA and a PO-bond at 963 cm−1 for the HA were used for evaluation by the Bruker OPUS Software. For better evaluation, the spectra were additionally normed on the PVA signal and presented as intensity vs. depth. Nontreated PVA patches and HA powder alone without any CA were used as controls for Raman spectroscopy.
Influence of HA nanoparticles on dexamethasone release from PVA patches
To evaluate the effect of the HA nanoparticles in the PVA patches, on the dexamethasone release, the first layer (133/200 PVA/HA) was prepared as described above. Due to the low solubility of dexamethasone in water (<100 mg/100 mL), 10.6 mg of dexamethasone was dispersed in 2 mL of distilled water by vortex, followed by 15 minutes in an ultrasound bath at 40°C. Then 2 separate PVA/HA dispersions including dexamethasone were prepared in 3 steps as follows: In step 1, 100 mg HA nanoparticles were added in 4.4 g of distilled water, followed by dispersal in an ultrasonic bath at 40°C for 15 minutes. In step 2, 755 µL of dexamethasone dispersion (corresponding to 4 mg dexamethasone) was filled with distilled water until 1 g of weight was reached, and this was added to the HA dispersion. step 3 involved 2 g of 15 wt% PVA solution (300 mg PVA) added to the HA/dexamethasone dispersion, followed by treatment in an ultrasonic bath at 40°C for 30 minutes. This was then incubated on a tube roller for 24 hours before being cast in the mold. One of the dispersions was cast on the 133/200 PVA/HA layer to achieve double-layered (133/200 PVA/HA + 300/100 PVA/HA) film, and the other dispersion was cast alone to achieve a single-layered (300/100 PVA/HA) film. The films were dried and prepared as P8 patches: double-layered (133/200 PVA/HA + 300/100/4 PVA/HA/dexamethasone) and single-layered (300/100/4 PVA/HA/dexamethasone). Then, the patches were glued on the sandpapered Titanium alloy-Synthes disks with 1 µL of PBCA after 1 minute of humidity exposure and 1 minute of pressing. Nonglued patches served as control groups in release studies conducted as described in the section “Preparation of PVA Patches With Incorporated Dexamethasone-Loaded PLGA Microparticles.” To define the total amount of dexamethasone loading in the patches, they were dissolved in 2 mL of distilled water using a lab vortex mixer. The amount of the released dexamethasone from the patches (glued or not) and from dissolved patches was quantified by HPLC as described above in the section “Preparation of PVA Patches With Incorporated Dexamethasone-Loaded PLGA Microparticles.”
Results
Drug-release profile of microparticles embedded in PVA patch
The penetration of PBCA into the PVA patches can have an inhibitory effect on drug release. However, the results showed that the shape of the drug-release profile was not altered by PBCA penetration (Fig. 5). In all cases, a burst drug release was observed at the first day even though different initial amounts were released. The control group of nonglued PVA/PLGA/dexamethasone patches were dissolved almost immediately after their placement in PBS but showed decreased dexamethasone release at day 1 (76.0% ± 2.6% for nontreated, 75.9% ± 38.3% for pretreated patches) when compared with the release from the control group–microparticles alone (89.7% ± 9.2%). Gluing of these PVA/PLGA/dexamethasone patches resulted in even further decreased release during day 1 (57.2% ± 1.6% for nontreated, 60.8% ± 24.5% for pretreated patches). The burst release was followed by a gradual slow release during the observed period of 28 days. The only exception was the glued, nontreated patch group, where the release reached 73.6% ± 2.1% at day 3, and no further release was detected during the 4 weeks of the observation period (Fig. 5).
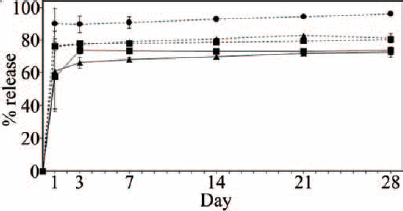
A graphical presentation of dexamethasone release from PVA/PLGA/dexamethasone patches. Nontreated/glued (rectangles with unbroken line); pretreated/glued (triangles with unbroken line); control/nontreated/not glued (squares with dashed line); control/pretreated/not glued (triangles with dashed line); control/microparticles only (circles with dashed line).
Shear strength of coating and influence of HA nanoparticles on PVA patch morphology
The addition of HA nanoparticles to the PVA resulted in milky white patches, and their morphology depended on the HA to PVA ratio. The first layer in the double-layered patch (133/200 PVA/HA) resulted in uniform particle distribution and a compact structure of the layer (Fig. 6A). The second layer (300/100 PVA/HA) showed formation of HA nanoparticle clusters within the PVA. Achieving a uniform distribution of single HA particles or specific HA cluster sizes was not within the scope of this study but may be a subject of future work.

(
The shear strength of the PVA/HA patches was correlated to the thickness of the patch. In particular, the 58.8 ± 3.9 µm thick double-layered patch (133/200 PVA/HA + 300/100 PVA/HA) had a shear strength of 13.0 ± 2.3 MPa, whereas the 31.7 ± 4.6 µm thick single-layered (300/100 PVA/HA) patch showed a significantly lower shear strength of 8.4 ± 2.3 MPa (Fig. 6B).
Patch properties after attachment to implant surface
HA nanoparticles were included in the PVA to prevent the penetration of CA monomers into the patch. The results obtained by depth profiling showed that the CN bond at 2,250 cm−1 was missing in the spectra, indicating that no PBCA was penetrating into the double-layered patch (133/200 PVA/HA + 300/100 PVA/HA). No PBCA was detected at the point of measurement closest to the patch surface that faces the test implant surface – i.e., the side where the CA monomers have been added (Fig. 7A). Also at a greater penetration depth of 60 µm, no accumulation of the CN bond signal was detectable (Fig. 7B).
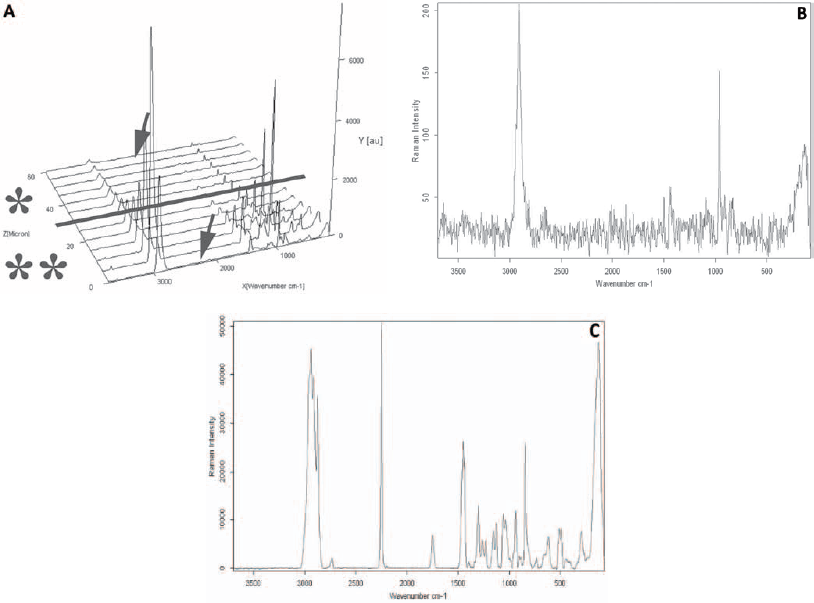
(
Even though no penetration of PBCA was observed within both the double-layered patch (133/200 PVA/HA + 300/100 PVA/HA) and single-layered patch (300/100 PVA/HA), the presence of highly crystalline HA nanoparticles in the PVA resulted in an improved patch attachment to the implant test surfaces maintained in the wet state (Fig. 8). For 80-µm thick PVA patches with no HA, a tendency to detach from the surface shortly after swelling in PBS at 37°C -38°C was observed. The only exception was the group with titanium alloy (AAP), where patches remained attached for more than 4 weeks (data not shown). On the other hand, double-layered patches (133/200 PVA/HA + 300/100 PVA/HA) and single-layered patches (300/100 PVA/HA) detached after swelling, only from the Titanium alloy-Synthes surface. After 7 days of incubation, 2 out of 3 single-layered patches had detached, only from the CrNi steel-AAP (Fig. 8). The rest of the patches (double-layered on Ti-Synthes-multiple use, Ti-AAP, CrNi-AAP, and single-layered on Ti-Synthes-multiple use, CrNi-AAP) remained attached to the surfaces for more than 4 weeks. The thicker PVA/HA patch formulations had a higher tendency to detach (data not shown) and therefore were excluded from further investigation.

(
Influence of HA nanoparticles on dexamethasone release from PVA patches
HA nanoparticles were incorporated into the PVA patch to prevent PBCA penetration and eliminate any potential reaction with incorporated bioactive molecules. And yet, the presence of HA nanoparticles might affect the drug-release profile provided by the patches. For that reason, only pure dexamethasone rather than dexamethasone-loaded PLGA microparticles were included in the patch, which also reduced the complexity of the system. Therefore, dexamethasone was loaded in double-layered (133/200 PVA/HA + 300/100/4 PVA/HA/dexamethasone) and single-layered (300/100/4 PVA/HA/dexamethasone) patches. The results showed that a complete dexamethasone release was achieved from the nonglued patches (double- and single-layered) during the first day. Gluing of these patches resulted in similar drug-release profiles. To be more specific, double-layered patches released 86% ± 20% at day 1, followed by a complete release at day 3. The big standard deviation in this group is probably due to the inhomogeneous dexamethasone penetration into the first layer of the patch during the preparation procedure. On the other hand, single-layered patches showed 92.8% ± 2.6% release during the first day, followed by a gradual and very slow release during the observation period of 14 days (Fig. 9).
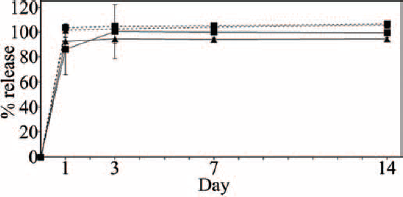
Dexamethasone-release profiles from PVA/HA/dexamethasone patches: double-layered (133/200 PVA/HA + 300/100/4 PVA/HA/dexamethasone)/glued (rectangles with unbroken line); single-layered (300/100/4 PVA/HA/dexamethasone)/glued (triangles with unbroken line); double-layered (133/200 PVA/HA + 300/100/4 PVA/HA/dexamethasone)/not glued (squares with dashed line); single-layered (300/100/4 PVA/HA/dexamethasone)/not glued (triangles with dashed line).
Discussion
Local drug release is known to be capable of enhancing regenerative cascades, serving as a pain killer or preventing secondary infections. However, no strategies for a personalized drug release are readily available, which can be shaped to the patient's needs by the surgeon and which may be used in an intraoperative setting and in conjunction with conventional therapies for critical bone defects such as implants. One possible solution can be a drug-loaded patch that is attached to the implant surface by in situ CA polymerization, which exhibits a controlled drug release depending on the microparticles embedded in the patch. However, the CA monomer penetration and polymerization within such a patch represent a potential risk for drug modification, raising safety concerns, which need to be adequately addressed and excluded.
The goal of the present study was to develop a technology platform that would allow the fulfillment of the above-mentioned criteria using a multilayer patch strategy. The hypothesis was that a multilayer approach would allow, on the one side, restriction of CA monomer polymerization to the first patch layers. On the other side, HA nanoparticles in the whole patch would influence its morphology and improve the patch attachment even in a wet state as a realistic intraoperative and in vivo scenario. In this way, an intraoperative customization would allow a local and controlled drug delivery from the implant surface of interest.
To determine the effect of the penetration of CA monomers within the patch and the potential influence of those monomers on the drug-release profile, PVA patches with no HA present had first to be used. For that purpose, dexamethasone-loaded PLGA microparticles (23) were included in nontreated and pretreated PVA patches. Very rapid release profiles were observed under the selected conditions. This behavior is not characteristic of pure dexamethasone-loaded PLGA microparticles, as we know from other studies (24-25-26). Still, here, a PLGA microparticle batch was selected with a surprisingly fast release pattern, possibly caused by some unknown phenomena that effected drug shielding in the PLGA matrix and allowed easier accessibility by the release medium. Despite the fact that this point may require further attention and optimization in the future when changing to various clinically relevant drugs, it allowed here the evaluation of a potential gluing effect on drug release. Still, the gluing showed a slight effect, as the amount of released dexamethasone in the glued patches was lower when compared with the nonglued control group patches (Fig. 5). Several factors may have contributed to the observed release profile: An interaction of CA and dexamethasone might have occurred, resulting in incomplete drug release in the time frame studied. CA monomers probably penetrated into and polymerized within the patch as shown previously (13), which builds a diffusion barrier to drug release and alters the accessible interface for efficient mass transport processes (15). Probably also the contact angle of the polymer patch and the release medium were affected. This, in addition to the reduced surface after attachment of patches to cover the titanium disk, may explain the observed slower release for the attached compared with the nonattached patches. When changing from model drugs to a specific therapeutic substance in the future, the capability of this technology to release clinically relevant doses at required rates will need to be examined.
The present work illustrates that such a drug-delivery concept can be achieved by a patch, which contains a higher weight content of HA nanoparticles than PVA in the first layer (e.g, the 133/200 PVA/HA layer). In such cases, the layer has a compact structure due to the uniform nanoparticle distribution within the PVA (Fig. 6A). To prepare another layer for drug delivery, a ratio of PVA/HA that contained less HA (e.g., 300/100 PVA/HA) was used. The HA nanoparticles entrapped in this layer of the patch formed clusters (Fig. 6A), which imparted patch stability and allowed drug molecules to diffuse space between the HA clusters.
The results obtained from the shear strength tests revealed that the shear strength of the PVA/HA patches was correlated to the thickness of the patch. In fact, the double-layered patches (133/200 PVA/HA + 300/100 PVA/HA) (Fig. 6B) had values comparable to pure PVA patches of similar thickness (13). On the other hand, single-layered patches (300/100 PVA/HA) showed a significant decrease in shear strength. The lesser thickness of the single-layered patches and their brittleness caused from the presence of HA nanoparticles might be an explanation for this result.
Since the penetrating CA monomers can inactivate bioactive molecules (27) and can also affect their diffusion from the patch, it was of greatest importance to prevent CA penetration throughout the patch layers. The CA penetration across layers could be avoided by the presence of HA nanoparticles that introduced hydrophobicity and seemed to act as a physical barrier for the CA monomer penetration throughout the patch (Fig. 7A). It was initially expected that the CA monomers would penetrate and polymerize within the 133/200 PVA/HA layer because their penetration within the patch seems to be inhomogeneous which may be linked the specific morphology of PVA as a typically semicrystalline material (14). However, the penetration was not only limited by, but seemed to be entirely prevented by, the HA nanoparticles, since the signal even from the analyzed location closest to the lower patch surface was free of signals from the CN bond, as shown by Raman spectroscopy (Fig. 7B).
Even though CA penetration into the patch was strongly impaired, both single- and double-layered PVA/HA patches showed improved attachment to the test implant surfaces in a wet state (Fig. 8) when compared with the PVA patches alone (data not shown). While the PVA/HA patches were detaching from the first-time-use titanium alloy (Synthes), the multiple use of this surface led to different behavior, and the patches did not detach. This is in agreement with the shear strength results, where the new metal surfaces generated lower values than the reused ones, even though the reuse did not have any influence on the micro-roughness (13). This is probably due to the changes in the surface chemistry, and further investigations need to be performed to explain this observation.
The composition of the PVA/HA patches or their gluing should not interfere with the diffusion of the free drug molecules in the drug-loaded patch. To reduce the complexity of the drug-delivery system and determine the effect caused only by the HA nanoparticles, only pure dexamethasone rather than dexamethasone-loaded PLGA microparticles were included in the patch. The single- and double-layered PVA/HA patches were able to achieve complete dexamethasone release in only 1 day (Fig. 9). Furthermore, gluing these patches onto the implant surface only slightly influenced the release profile. For example, with the double-layered patches, complete release was achieved after 3 days (Fig. 9). The main explanation for this effect might be the possible dexamethasone penetration into the first layer of the patch during the preparation procedure. Actually, if a second dispersion that includes dexamethasone is cast over the first layer, then the first layer absorbs water, swells and may partially allow dexamethasone access to this layer. The dexamethasone absorbed by the first layer probably needs a longer time to diffuse out of the patch. On the other hand, the single-layered patches released almost the complete amount during the first day, followed by a gradual and slow release (Fig. 9). That might possibly be due to dexamethasone interaction or entrapment in the HA nanoparticle clusters, which again would result in longer diffusion times for release. Also, the attachment of a patch to cover a surface causes decreased contact of the surface of the patch with the release medium, which may be the most probable explanation for the slower drug release with the attached compared with the nonattached patches. Nevertheless, formulations of this kind can include drug-delivery particles to control the release profiles and can be processed as a single layer or as the second layer of a double-layered patch.
There is a certain risk of CA monomer penetration and polymerization within the patch and their potential interaction with the drugs. Therefore, the use of HA particles to prevent the CA monomer penetration within the patch seems to be indicated to prevent any influence on the drug-release profile, as well as any kind of chemical modification. A multilayer approach as illustrated here can be used to ensure that CA penetration into the patch is limited, so that the second drug-loaded layer provides the desired release profiles.
Conclusions
An intraoperative custom-made implant-coating strategy was proposed that can allow drug delivery by drug-loaded microparticles embedded within a PVA patch after attachment to the implant surface with a CA adhesive. The addition of HA nanoparticles within the PVA patch prevented the penetration of CA monomers through a multilayered patch, simultaneously improving the patch attachment capability in a wet state and providing strong attachment to the implant surface in a dry state. In all cases, fast drug release was observed, which may be further optimized in future studies by using microparticles with a more efficient encapsulation of the therapeutic drug. It is important to note that the patch attachment did not relevantly interfere with the drug-release functionality. That is beneficial when the proposed patch design is a double-layered PVA/HA patch with 1 (drug-free) layer in contact with the CA adhesive and 1 layer containing the drug-loaded particles. For example, this approach could allow local and controlled release of growth factors to stimulate regeneration, allow local administration of antibiotic treatments or serve to provide pain relief at the local side of its occurrence. Thus it would eventually find use in treatment of postoperative pain management and significantly improve patient comfort. Further drug-release and toxicological studies are required to identify the best-suited application of this platform.
Footnotes
Financial support: B.T. was supported by DFG funding from the Berlin School for Regenerative Therapies (GSC 203). We also want to extend our thanks for the generous donations from Synthes GmbH, AAP Implantate AG and P.J. Dahlhausen & Co. GmbH.
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
