Abstract
The implants are increasingly being a part of modern medicine in various surgical procedures for functional or cosmetic purposes. The progressive use of implants is associated with increased infectious complications and prevention of such infections always remains precedence in the clinical settings. The preventive approaches include the systemic administration of antimicrobial agents before and after the surgical procedures as well as the local application of antibiotics. The relevant literature and existing clinical practices have highlighted the role of antimicrobial coating approaches in the prevention of implants associated infections, although the applications of these strategies are not yet standardized, and the clinical efficacy is not much clear. The adequate data from the randomized control trials is challenging because of the unavailability of a large sample size although it is compulsory in this context to assess the clinical efficacy of preemptive practices. This review compares the efficacy of preventive approaches and the prospects of antimicrobial-coated implants in preventing implant-related infections.
Introduction
The use of implants in clinical settings such as catheters, prosthetics, and various other devices have altered the concept of medicine, especially during the previous few years. However, these implants are considerably associated with increased risk of infection and certainly, implant-associated infections are the most common and severe complications accompanied by biomaterials usage. In the USA, the infections associated with devices account for approximately 26% of the healthcare-associated infections.1–3 The orthopedic implants are of particular focus as these remain in the body, therefore their infections are mainly challenging. The associated infections often lead to prosthetic device failure that involves the replacement of implants and usually cause chronic or relapsing disease.1,2 Moreover, the infections associated with the orthopedic implants are difficult to diagnose including the identification of causative agent and its drug susceptibility profiling and are often difficult to treat due to the antimicrobial resistance and persistence. 4
The implants associated infections involve composite interactions between biomaterials, the pathogens, and the immune responses of the host to both. In the absence of a foreign body, the contamination of tissues by opportunistic pathogens is regularly and naturally cleared by the immune defenses of the host. However, in cases of implant-associated infections, the local tissue response triggered against the biomaterial including inflammation, reaction to the foreign body, and granulation tissue formation that ultimately leads to fibrous encapsulation. 5 This provides a place of immune depression, which influences the microbial colonization of implant and a continuous source of infection.6,7 Additionally, the biomaterial provides a substrate for the adhesion and biofilm formation as the bacterial adhesion is the initial step that paves the way for the colonization and infection of the implants. Several studies have reported that the bacterial pathogens can form microcolonies and arrange themselves in biofilms, which protects and helps them to persist in the host, therefore the adhesion and formation of biofilms are essential in the pathogenesis of these infections. 8 For instance, a considerably few Staphylococcus aureus cells are required to infect in the presence of foreign material at the surgical site in comparison to the surgery in absence of any implant materials as seen in animal models. Furthermore, the implants permit not only the pathogenic microbes, for instance, S. aureus but also enable other bacterial species to thrive for example Staphylococcus epidermidis, which was earlier considered as a normal flora.7,8
Medical implants in use
It is estimated that more than 500,000 types of medical devices have been entered the market globally. The invasive devices including the indwelling and/or implantable medical devices just characterize a minor portion of these. On the other hand, the use of devices, for example, vascular and urinary catheters and cerebrospinal shunts is much more, just about hundreds of millions. The studies have reported that the number of cardiovascular electronic devices that are implanted globally has exceeded a million/year whereas the approximately 10 million procedures of the dental implant are performed.9,10 In the USA, the procedures of hip and knee arthroplasty have exceeded 1 million each year. Moreover, about 5–10 million women at present have undergone artificial breast implant procedures. 11
The infection risk differs with the type of device used, the extent of invasiveness, and the anatomical site and duration of insertion/application. The invasive devices especially those which are inserted through body orifices into the human body and are in contact with the mucous membranes favor the entry of bacterial species from the outer environment. Some of these invasive devices include tracheal tubes and cannulae, urinary catheters, fixed dental prostheses, and contact lenses. The endotracheal tubes offer a channel for the passage of microorganisms, which results in a greater risk of pneumonia. 12 However, the current classification system has distinguished the above-mentioned devices from the invasive implants and devices that breach the skin and the mucous membrane and are inserted into body tissue. These devices are widely used in neurosurgery, cardiovascular surgery, orthopedic surgery, plastic surgery, ophthalmology urology, gynecology, and dentistry. The common examples include spinal and neurological stents, cardiovascular stents, catheters, pacemakers, and heart valves, prosthetic joints, bone cement, and artificial ligaments breast implants, intra-ocular lenses, penile implants, urogynecological mesh surgical implants, and dental filling materials. 7 Therefore, these biomaterials are implanted in almost all anatomical sites of the body for the treatment of various medical disorders, and they interact with nearly all kinds of tissues. It is considered as a foreign body wherever this surgically invasive implant is placed and even a minor tissue response changes the immune defenses at the implantation site and is vulnerable to bacterial infection even by less pathogenic and opportunistic pathogens. The critical adverse effects that is, morbidity and mortality are usually observed in patients with permanent or long-term implants, the implants in critically ill patients, and the life-supporting implants into the vital organs such as the brain and heart. Although, almost all invasive devices, particularly the devices which are in contact with the bloodstream can cause sepsis. 7
Infections associated with implants
The implants associated infections generally initiate bacterial contamination during the surgical procedures. Usually, the infections following an orthopedic implant develop within 3 months of the surgical procedure and are known as early postoperative infections. The delayed infections develop between 3 and 24 months following surgery whereas the infections after 24 months are known as late infections.2,13 These late infections are usually asymptomatic infections that can be symptomatic far ahead or the new infections that can reach the implant through a hematogenous route.13–15 The more recent classification discriminates the early infections as those infections which are apparent during the first month of surgery and acute hematogenous infections for which the duration of symptoms lasts for up to 3 weeks from the chronic infections and persist for more than 3 weeks. The removal of damaged tissue and retention of the implant is often suitable only for early infections and acute hematogenous infections.13,14
The Gram-positive cocci mainly the Staphylococcus aureus, Staphylococcus epidermidis, and enterococci have been isolated from the infections associated with orthopedic prosthetics. The Gram-negative bacteria including Escherichia coli, Pseudomonas aeruginosa, and Proteus mirabilis are not frequently involved in infection whereas the anaerobic bacterial account for approximately 4% of infections. 16 These causative agents differ on the site and type of the implant and also depended on the time since the surgical intervention. 17 Moreover, this is suggested that the virulent microbes are usually involved in early infections through contamination of the surgical site such as S. aureus, and the delayed infections are mainly associated with the microorganisms having lower virulence. The hematogenous infections that occur in later stages instigate from those bacterial species which are involved in skin, dental, urinary, and respiratory tract infections. 7
The patients with orthopedic implants, the infections are usually caused by Staphylococci. A study reported the various pathogens associated with the patients undergone arthroplasty and differences between the geographic area were observed, however, S. aureus was the most frequent pathogen in the USA while S. epidermidis was somewhat more common in European countries. 18 These disparities might be due to various factors, for example, the local prevalence of a particular pathogen and the extent of antibiotic resistance in consort with the prophylactic and aseptic measures used. 7
Bacterial adhesion to the surfaces of the implant
The adhesion of bacteria to the surface of implants is mainly divided into two stages. In the first stage, the attachment is unspecific and reversible while and second stage of attachment is specific and irreversible. During the initial adhesion to the abiotic surfaces, the attachment is normally unspecific, however, the adherence with the living tissues specifically involves the adhesion or lectin-based interactions. 7 The exposed surfaces of biomaterials are immediately covered by the extracellular matrix (ECM) proteins and immunoglobulins as soon as these are flooded with physiological fluids. Likewise, the proteins from the blood or interstitial fluid are coated on the implant surface within moments and this progression is dependent on the surface chemistry of the implant.7,19 The adhesins are therefore the foremost means of bacterial adherence and attachment to the surface of the implant inside the human body. Multiple mechanisms have been reported in staphylococcal species for the attachment and formation of biofilms that ultimately lead to chronic implant-associated infections.8,20
Adhesion to uncoated abiotic surfaces
The nonspecific forces come into play for the initial attachment of bacteria to the abiotic surfaces and bacteria act like colloidal microparticles.21,22 The bacterial cell appendages for instance pili and pilus-like structures and nanofibers serve as adhesins. In some bacteria, the nanofibers facilitate cell adhesion to the abiotic surfaces and are therefore associated with biofilm formation. 7 The bacterial species-specific proteins such as the autolysins AtlA in S. aureus and AtlE in S. epidermidis facilitate the binding of bacterial cells with abiotic surfaces. In S. epidermidis, AtlE contributes to the attachment to abiotic surfaces for instance polystyrene and biomolecules that is, vitronectin. The AtlA, however, is an enzyme that is cleaved into catalytically active proteins, a glucosaminidase, and an amidase that binds with the matrix proteins such as fibronectin, fibrinogen, and vitronectin. Therefore, AtlA principally facilitates the S. aureus to adhere to implants that are already coated with the host matrix proteins. Moreover, AtlA was also reported to contribute to the formation of biofilms in S. aureus and Enterococcus faecalis through its autolytic activity and the E. faecalis strains were found defective in biofilm formation after AtlA deletion.7,23
Adhesion to biotically coated surfaces
The adhesion of bacterial cells with ECM principally ensues by specific binding of bacterial adhesins (piliate and non-piliate) to the host proteins. The S. aureus has a range of adhesins, including cell wall anchored components as well as secretable adhesive molecules that are ionically linked with the cell wall. These adhesins are usually multi-functional and not only facilitate adhesion to the ECM proteins but are also involved in the modulation of host immune response and mediate the internalization of bacteria into the host cells.24,25 Collagens, fibrinogens, and fibronectins are the key matrix proteins that serve as ligands for bacterial adhesins. Collagen is a family of more than 20 matrix proteins that are the structural components of tissues. The bone matrix is rich in bone sialoprotein and type-I collagen. In orthopedic implants, the biomaterials interface with the bone tissues, therefore, S. aureus strains having the ability to adhere to bone sialoprotein and collagen are more virulent. 26
Fibronectins: the homo-dimeric glycoproteins are linked by disulfide bonds. The soluble fibronectin present in plasma is secreted by hepatocytes whereas the fibroblasts secrete fibronectins in the interstitial spaces and are associated there into insoluble polymers of high molecular mass. Fibronectins and fibrous collagens attach and promote the adhesion of cells and adjust the cell shape. The molecular nature of the binding between S. epidermidis and fibronectin has been studied and was found that S. epidermidis bound with the carboxy-terminal domain and this interaction was inhibited in the presence of heparin. The synthesis of fibrinogen occurs in the hepatocytes and is comprised of three pairs of non-identical chains. The Staphylococci binds with fibrinogen that is considered an important factor in catheter-associated infections as compared to orthopedic implant infections. 7
The term “race to the surface” was introduced over the past years to designate the competition between the contaminating microbes and the host cells to inhabit the surface of biomaterials. The key to success is a prompt integration of the biomaterials into tissues for most implants, and it is suggested that rapid integration is also critical to prevent bacterial colonization. If bacteria adhere to the surface before the tissue repair the host defenses are unable to avoid colonization and formation of biofilms.21,27 The in vitro models offer mechanistic understandings of the interactions between the biomaterials, bacteria, and host cells; however, they usually focus on a single cell type and shorter observation times.28,29
Biofilm formation
The bacteria usually infecting the implants are not single, sparsely distributed, or adherent cells but are aggregated and adhered tightly to the surfaces of the implant in form of biofilms and are enclosed in a profuse milieu of the extracellular matrix as shown in Figure 1. These biofilms are accountable for the persistence of implant-associated infections, the recalcitrance of infection to therapy, and are a continuous source of bacterial spread. Moreover, the host immune response and antimicrobial agents are often futile against the bacteria present in form of a biofilm, therefore leading to chronic inflammation. Furthermore, the higher cellular density and diversity in biofilms help to increase the chance of horizontal gene transfer among microbes, and conjugation ensues more frequently between the biofilm communities as compared to the planktonic cells.5,30

Schematic diagram represents bacterial colonization on the implant surface. Planktonic bacterial state undergoes phenotypic transformation and develops microcolony. Adherent cells of microcolony divide rapidly and synthesize extracellular polysaccharides/matrix to generates a mature biofilm. Bacterial cells of biofilm undergo microevolution and develop a resistant biofilm. The first stage of the competition phase is an important and vulnerable step in antibiotic-resistant biofilm development. It can be targeted by the coating of the implant with antiadhesive substances (a), antibacterial peptides (b), host cells (c), or antibiotics (d).
The clinical need for antimicrobial-coated implants
The establishment of biofilms on the biomaterials leads to a wide array of chronic infections that are tough to eradicate and have been involved in increased mortality rates.31,32 The immunocompromised hospitalized and elderly patients are at greater risk of acquiring implants associated infections that is nearly 60% of hospital-acquired infections. Although, the innovative medical implants and devices are increasingly being common and are an essential part of clinical care, however, due to their extensive the modern medicine is facing a double challenge: the pathogenesis of bio-film producing clinically important microbes and the problems linked with the growing burden of infection risk posed by multidrug drug-resistant. 33 The variety as well the number of these medical devices that are currently in use including the temporary urinary and vascular catheters to the orthopedic devices has increased over time thereby, the peril of acquiring device associated infections have considerably increased owing to the physical and chemical structure of foreign bodies that every so often encourages the adherence of microbes. 34 Several sources have been described for the transfer of these causative agents involved in such types of infections including distal local infections, hands of the surgical team, patient’s mucous membranes and skin, contaminated medical equipment and devices, and contact with other patients or the family members following a surgical procedure. 35 The major bacterial pathogens involved in biofilm formation on medical implants are summarized in Table 1.
The major bacterial pathogens involved in biofilm formation on medical implants.

The device-related infections generally progress in a definite order that includes the host’s proteins adsorption on the surfaces of implants as a first step which is mainly influenced by the roughness, hydrophobicity, chemical composition, and porosity of the surface. The concentration and composition of the proteins, pH, and salt concentrations also influence the protein adsorption process. 36 This stage is of utmost importance for the microbial adhesion that follows by the proliferation and modulation of their metabolisms and gene expression and consequently growth as a biofilm on the surfaces of the implant. 37 The aseptic procedures and suitable protocols for antibiotic prophylaxis are desired during operative procedures to avoid the antibiotic treatment of biofilms that are already established or the removal of devices as the last option. Additionally, the epidemiological investigation of outbreaks and the quarantine of infected patients as well as the colonized personnel by multidrug-resistant strains as standard procedures that have been applied in hospitals as a preventive measure for biomaterial-related infections. 35
The foremost problem is that it is difficult to avoid such infections entirely despite the sternest hygiene measures and thus the emerging approaches to avert them by inhibiting bacterial adherence or by their killing are increasingly being investigated. A potential way that has increasingly gained importance over time to fight these infections is the development of anti-infective biomaterials which can either thwart the microbial adhesion or microbial killing in the adjacent areas. 36 Specifically, the until now tested anti-infective biomaterials are capable of hindering protein adsorption and early microbial adhesion, hampering the colonization and biofilm formation of microbes, expressing bactericidal action at the interface of devices with host tissues, delivering bactericidal substances at an active concentration, interfering with the functioning of bacteria and disruption of the structural integrity of pathogens in a single form or the complex association of biofilms.38,39 Generally, the anti-infective coatings can prevent bacterial adhesion and are either antimicrobial or antifouling that is capable to kill microbes upon their contact with the device surfaces or able to prevent the accumulation of microbes by repellent properties and interfere with the biofilm formation as shown in Figure 2.35,36

The key attributes of an ideal bioimplant. It can promote host cellular growth and proliferation (a) and must be compatible with the host organism (b). Coating of antibacterial peptides (c), antibacterial agents (d), and antiadhesive agents (e) can inhibit the development of bacterial biofilms.
Preventive strategies for implants associated infections
Implants coatings with antimicrobial agents
During the past few years, the biomaterials that possess antimicrobial activity have got a strong upsurge. Therefore, FDA-approved biomaterials with anti-infective activity have considerably increased during the past decade which specifies the necessity for substitutes to the systemic use of antibiotics that usually come across the problem of poor penetration into the biofilm. The antibacterial biomaterials are the broadest subcategory at the moment as compared to the antifungal biomaterials among the anti-infective biomaterials to cope with the medical device-associated infections. 35 Most of these biomaterials especially those which are by now available in the market specifically provide antimicrobial activity through drug release as well as non-releasing-based methods. The various preventive strategies are summarized in Figure 3.
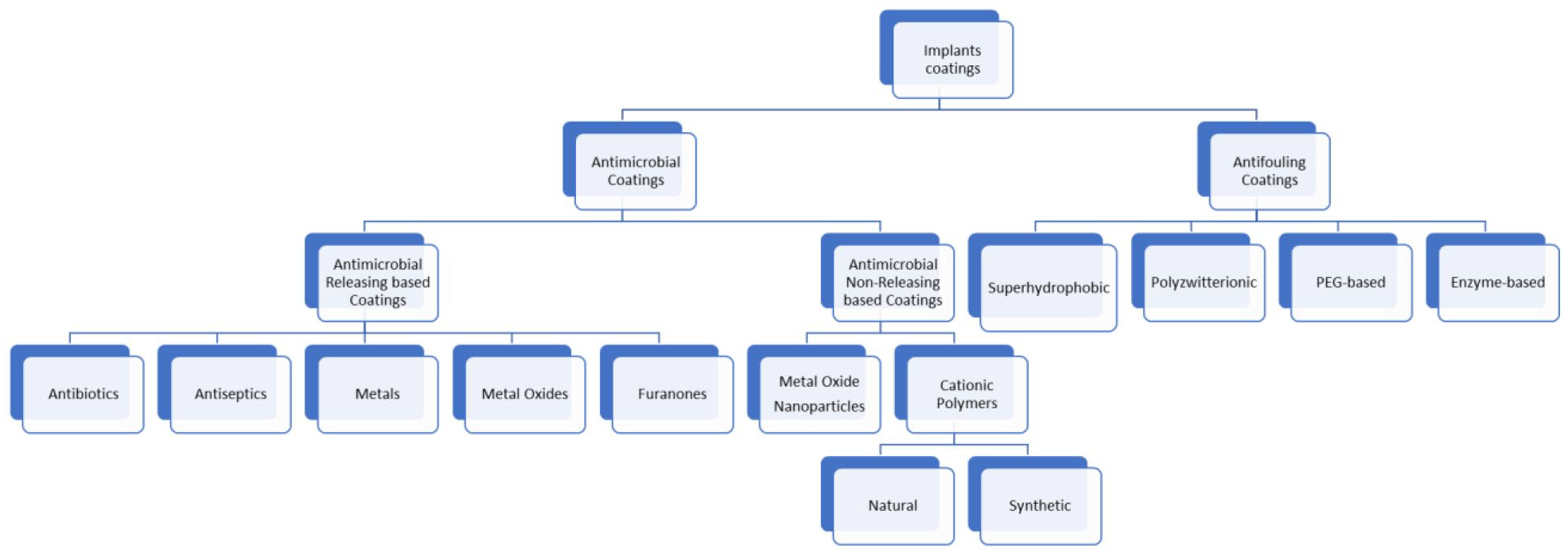
Classification of antibacterial strategies for implant protection.
Antimicrobial coatings based on drug-releasing
The drug released-based coating is an interesting method to reduce the bacterial adhesion that is mainly based on the coatings which release a different type of antibacterial agents. These antibacterial molecules are coated onto the biomaterials by impregnation, physical adsorption, conjugation, or complexation. These active coatings are intended to serve as carriers of antimicrobial compounds which are released in higher concentrations in the surrounding media and are therefore transported to the attached bacterial cells after implantation to constrain the initial colonization and avoid formation the biofilm formation on implanted surfaces.40,41
The antibiotics like vancomycin or gentamicin have been extended used for the local delivery of antibiotics by incorporating these antibiotics in bone cement which are used for fixing the prosthetic implants.45–48 However, several recent studies have reported that these antibiotic-loaded bone cements were not found efficient.49,50 The possible alternate method to this bone cement is controlled coatings for antibiotic release that is based on biodegradable constituents. The cementless prostheses are increasingly being popular especially now in hip arthroplasty and are designed for a prosthesis. 51 The biodegradable coatings on the surface of medical devices holding antibiotics favor the slow release of antibiotics upon the degradation of superficial layers that reaches the interface of the device surface and tissues. The kinetics of the release of antimicrobial agents is equal to the kinetics of the degradation for that particular coating. For example, biodegradable implant coatings loaded with gentamicin were developed by using polylactic-co-glycolic acid or poly(d,l-lactide) polymers as a biodegradable carrier and were found very effective. The gentamicin-containing carriers were applied on the surface of titanium implants that considerably decreased the rate of infection and drastically enhanced the recovery following infection compared to the systemic use of gentamicin.52–54
Titanium alloy substrate was coated with a thin sol-gel coating loaded with vancomycin for the prevention and treatment of bone infections. The antibiotic coating drastically decreased the number of S. aureus cells in the rat osteomyelitis model. Moreover, there was a strong correlation between the release of vancomycin and the rate of degradation of the sol-gel film.55,56 The combinations of antibiotics that is, rifampicin and fusidic acid and of local antiseptic that was loaded on poly(d,l-lactide) was found effective to kill S. aureus in vitro as well as and in a rabbit bone infection model. 57 The K-wires that were coated with poly(d,l-lactide) loaded with single or combination of two antibiotics that is, gentamicin, ciprofloxacin, cefoxitin, colistin, or daptomycin were found successful to fight the mixed infections. The results established that the antibiotics alone as well as in combination were released with an initial burst and exhibited antibacterial activity in a dose-dependent manner. 58
During the previous few years, mesoporous and grit-blasted materials for example hydroxyapatite was used for the loading of gentamicin and tobramycin.59,60 Various other antibiotics including cephalothin sodium, amoxicillin, carbenicillin sodium, and vancomycin hydrochloride have been incorporated in hydroxyapatite coatings and tested for their release and efficacy. The studies have highlighted the potential of antibiotic-loaded hydroxyapatite coatings in a marked reduction in the adhesion and biofilm formation by pathogenic bacterial species and a comparatively high loading of antibiotics was observed in addition to the higher efficiency of antibiotic releasing capacity.35,61
Most of the coating strategies for orthopedic implants are also investigated for dental applications. The drug-loaded hydroxyapatite coatings have been considered as an effective and practical solution for the decontamination of surfaces in dental implant surfaces. Instead of gentamicin, tobramycin was used because the gentamicin showed less affinity for the inner ear and have ototoxic effects. 60 The chitosan and silica loaded with antibiotics have also been used as dental implants, however, they are not found much useful owing to the finite lifetime of antimicrobial drug composites in degradable coatings and resulted in peri-implantitis after few years. On the other hand, the FDA-approved and clinically acceptable polycaprolactone were recognized for its improvement in the longevity of the used antibiotics due to the degradation rate and release of antibiotic for a longer period. The polycaprolactone can delay the release of tobramycin from the allograft bone as this polymer can serve as a releasing rate-controlling membrane. 35 Among the other potential candidates, hydrogels are a useful option for controlled antibiotic release. The ceftriaxone and vancomycin modified poly-ethylene glycol hydrogel coatings for titanium dental implants displayed exciting antibacterial action against methicillin-resistant S. aureus. 62
For urinary catheters and central venous catheters, antibiotic-releasing coatings have been introduced in medical practices. 63 The antibiotics in combination with antiseptics such as rifampicin and minocycline or chlorhexidine and silver-sulfadiazine have been coated on the external and internal surfaces of catheters and their efficacy was analyzed in various clinical trials. These trials established the efficacy of antimicrobial-coated catheters in the improvement of the outcomes of catheter-associated bloodstream infections and reduced the colonization of the catheter. Moreover, the antibiotic coating was found more effective in terms of antimicrobial effect as compared to the antiseptic coatings.64,65 The economic aspects were analyzed for the use of antibiotic-coated catheters and the antiseptic-coated catheters and were compared with the standard ones and were concluded that the coated implants were cost-effective in healthcare settings. 66
A study reported the anti-staphylococcal substances for the coating of catheters although these have not been used in medical practices. The polyurethane with gentamicin, fosfomycin, ciprofloxacin, and flucloxacillin was thoroughly examined for their release rates, morphological features, and bacterial colonization to foresee and appreciate their antimicrobial effects. A fast-initial rate of release was observed for ciprofloxacin, whereas gentamicin showed a more constant release behavior. These polyurethanes exhibited a slow and sustained release of the gentamicin and bacterial colonization was efficiently inhibited. 67 Different functionalized polyurethanes were synthesized for the improvement in the affinity between the polyurethane surfaces and antibiotics. The establishment of polymer/antibiotic interactions controlled the extent of loading in the system as well as the release rate that resulted in a prolonged antibiofilm ability as evident from the scanning electron microscope micrographs of the untreated and treated (with tigecycline) surfaces of polyurethane. For biofunctionalization, the antibiotic-loaded composite mixed with the albumin nanoparticles were used.68,69
In contrast, some studies do not agree with the counteracting or preventive effect of chlorhexidine releasing dental coatings against caries. A systematic review reported that there is uncertain evidence regarding the effect of chlorhexidine in the prevention of dental caries in children and adolescents. Moreover, the experiments were performed using chitosan, tetracycline (20%), or chlorhexidine digluconate (0.02%) coatings for titanium showed that tetracycline inhibited the growth (95%–99.9%) of Staphylococcus epidermidis and Aggregatibacter actinomycetemcomitans (up to 7 days) without cytotoxicity to the cells whereas the chlorhexidine was active against the pathogens for only 1–2 days with a 56%–99.5% inhibition with toxicity to the human cells. 73
For urinary catheters, the Ag coating is the best choice amongst the antimicrobial coatings of choice and several meta-analyses have reported the effectiveness of using urinary catheters coated with Ag alloy in reducing the asymptomatic bacteriuria and urinary tract infections in the catheterized patient compared with the Ag oxide-coated catheters.76,77 A study compared the Ag alloy-coated catheter with a nitrofurazone-coated catheter in vitro and found nitrofurazone-coated ones as a better choice according to the bacterial cell count that was detached from the catheters through sonication. 78 In a multi-center randomized controlled trial in the UK, the results of three types of impregnated catheters were compared including standard polytetrafluoroethylene, Ag alloy-coated, and standard polytetrafluoroethylene catheters. The symptomatic urinary tract infection rates were found lowest in the nitrofurazone group followed by the Ag alloy group and the polytetrafluoroethylene group. 79
The central venous catheters (CVCs) coated with Ag have also been used that included sole Ag-impregnated CVCs as well as CVCs impregnated with Ag, platinum, and carbon (iontophoretic). Agion® (Sciessent: Wakefield, MA, USA) was the first antimicrobial technology-coated catheter based on the impregnation of Ag zeolite in the CVC polymer matrix. The clinical trials have assessed the efficacy of Ag-coated CVCs, however, they failed to prove their usefulness in reducing the rate of bacterial colonization among the patients in intensive care units (ICUs) that were catheterized compared to the standard CVCs.80,81 The platinum, Ag, and carbon impregnated CVCs were synthesized in combination with Polyurethane with the brand name Oligon®. The randomized, controlled trial with 206 patients admitted to ICU showed that the incidence of colonization and catheter-associated bloodstream infections using Oligon catheters was 29% as compared to the control catheters that is 29%. 82
The combination of Ag with chlorhexidine has achieved increased efficacy. In the first generation of chlorhexidine and silver-sulfadiazine, the antimicrobial agents were coated on the external surface of CVCs. The short-term effects of these CVCs were overcome during the manufacturing of second-generation chlorhexidine and silver-sulfadiazine-coated CVCs having a three times higher concentration of chlorhexidine and with both the surfaces (internal and external) coated with these antimicrobial agents. 83 In a clinical trial among 363 ICU patients, the rate of colonization was 3.7% lower in the chlorhexidine and silver-sulfadiazine coated CVCs group as compared to the group of patients having untreated catheters. 84 Currently, a wide variety of silver-containing wound dressings are also available including Actisorb Silver (Johnson and Johnson, USA), Silverlon (Argentum Medical, USA), and Acticoat (Smith and Nephew, UK) are few of these. These antimicrobial-containing dressings were found very useful for the successful management of wound-associated infections. 85
The efficacy of Ag nanoparticles (NPs) seems to be better attributable to multiple mechanisms including the release of Ag ions, the production of reactive oxygen species, and the damage of the cell membranes directly. 86 A study reported the wound protection from E. coli and S. aureus and healing effects in an animal model using hydrogel embedded with Aloevera and AgNPs. 87 In a study, the CVCs surfaces were treated with a polydopamine layer that induced the formation of Ag-NPs as a homogeneous coating comprised of spherical and well-separated NPs having a size of 30–50 nm that possessed antibacterial activity against S. aureus. 88 The AgNPs along with heparin were embedded with a copolymer to obtain a bifunctional coating on the surface of CVCs with antithrombotic and antimicrobial properties. The synergistic action was observed between heparin and AgNPs as the coating was found to inhibit the adhesion of S. aureus. 89
The usnic acid; a benzofuranone secondary metabolite in lichen was impregnated into polyurethane that was unable to inhibit S. aureus during the initial attachment, however, was capable to kill the attached bacteria thereby preventing the biofilm formation. The P. aeruginosa was able to form biofilms on the polyurethane surface loaded with usnic acid, but the morphology of the biofilm was altered as compared to the biofilm growth on the untreated polyurethane surfaces. These findings suggest the role of usnic acid in interfering with the signaling pathways of P. aeruginosa. The studies also reported the capacity of furanone to decrease the adhesion of S. epidermidis and slime production on various biomaterials when adsorbed to polyvinyl chloride, polyurethane, polypropylene, polyethylene, polytetrafluoroethylene, and silicone.35,93 Tympanostomy tubes were fabricated from the polymers including ethylene-vinyl acetate and polyurethane and antimicrobial agents that is, ciprofloxacin and usnic acid was inserted into the eardrum (tympanic membrane) and were tested for antimicrobial activity against S. aureus, P. aeruginosa, Streptococcus pneumoniae, and Haemophilus influenzae as well as drug release kinetics. The kinetic curves studies have revealed that the concentrations of the released drug concentrations were greater than the MICs during the first 6 days of elution. 94
It is generally understood that the furanones can exhibit disparities in their antibacterial activity that depends mainly on the bacterial species. In two study in the same year, three brominated furanones was used to apply on the PVC surface and were ineffective to stop the biofilm formation by S. aureus but one of the furanone was found effective to inhibit the biofilm formation by E. coli.95,96 Various studies have reported the use of furanones after loading them onto the microparticles and nanoparticles. Specifically, the halogenate furanone was combined with biodegradable NPs made up of poly(D/L) lactic acid (PDLLA) to construct an antibacterial coating for titanium implants. This antibacterial coating was found to inhibit the S. aureus all over the 60 days of study. This period is considered long enough to prevent implant-associated infections during the initial stages of device implantation.97–99
The antibacterial activity was assessed for nitric oxide-releasing xerogel coated on a silicone elastomer of medical-grade with a sol-gel-derived film capable to store nitrous oxide in vivo. The subcutaneous implants of these silicone materials into rats with a prior administration of S. aureus colony showed a marked (82%) reduction in the rate of infected implants which suggests the potential application of nitric oxide as biomaterial coating for the prevention of such infections. 102 The nitric oxide modified xerogel coatings were demonstrated to reduce the adhesion of P. aeruginosa by approximately 90% and to remove the previously adhered bacteria. 103
The Low concentration nitric oxide-releasing coatings were developed to produce mono-filament polypropylene meshes. These coatings exhibited a considerable bactericidal effect on the biofilms produced by S. aureus, coagulase-negative staphylococci, P. aeruginosa, and E. coli in vitro, however, their beneficial effects in vivo using mouse model were not evident from bioluminescent imaging following the subcutaneously implanted meshes. 104 The nitric oxide-releasing films fabricated by diazeniumdiolate-doped PLGA poly(lactic-co-glycolic acid), which is established as a promoter as well as controller of nitric oxide release were developed which were further encapsulated in a silicone coating. These films showed a 98.4% reduction in S. aureus biofilm biomass and 99.9% of E. coli following incubation in a bioreactor at 37°C or room temperature for 1 week. 105
Non-releasing methods
The non-releasing methods for antimicrobial coating are based on the coating materials which can counteract bacterial adhesion and avoid the formation of biofilms when microbes gain access to the coated surface. These coatings are mainly based either on the polymer which themselves possess antimicrobial or the metal-oxide NPs. 35
Antimicrobial polymer-based coatings
The antimicrobial polymers are mainly known as polymeric microbicidal agents if are produced by the polymerization of the repeating units of the bioactive molecules or referred to as biocidal polymers if the entire molecule is bioactive. 35 The polymerization of antibiotics for the synthesis of polymeric biocides is mainly achieved to diminish the toxicity of antibiotics, however, a decrease in the activity often occurs as seen in the case of polymerization of vancomycin and polyethylene glycol (PEG)-methacrylate that gives rise to a polymer which showed six-fold less activity compared to the vancomycin itself. 106 Additionally, the polymeric biocide is only active when the linked antibiotic is released from the carrier in the majority of cases which makes these non-releasing methods uncertain. 107 The biocidal polymers are fascinating and recently have got considerable attention because of their advantages as chemically stable, non-volatile, and are unable to permeate via skin in human beings and animals. Therefore, biocidal polymers can lessen environmental concerns and have a longer lifetime. The majority of these are polycationic and capable to bind with membrane proteins of microorganisms.35,108
Cationic biocidal polymers
The cationic polymers are widely being used in the development of approaches for gene delivery and drug delivery. These are divided into natural and synthetic cationic polymers and bear a net positive charge. Upon exposure to the cationic polymers, the adsorption and penetration of the agents occur into the microbial cell wall followed by their reaction with the cell membrane (protein or lipid) leading to membrane disassembly. The leakage of low molecular weight matter, the degradation of nucleic acids and proteins, and ultimately the cell wall lysis may also occur. 35
From the antimicrobial perspective, the chitosan has been modified by combining the quaternary ammonium group or by embedding small molecules or short polymeric onto the chitosan backbone to make it more effective. The chitosan modification using quaternary ammonium compounds has improved its water solubility as well as antibiofilm ability as evident from studies. 111 The covalent joining of chitosan to various surfaces was also developed using carboxymethyl chitosan and pristine chitosan attached to titanium and titanium alloy substrates and was found more effective in killing pathogenic microbes and preventing bacterial biofilm formation. The bacterial number on the modified titanium substrates was lower than on uncoated controls as observed in these experiments. The surface of medical-grade silicone which was treated with polydopamine and grafted with carboxymethyl chitosan exhibited more than 90% reduction in the adhesion of Proteus mirabilis and E. coli and the biofilm formation by both bacterial species under static as well as dynamic conditions. 112
The antimicrobial spectrum following the covalent linkage of chitosan with various polymer surfaces including polypropylene, polyethylene, and polyethylene terephthalate was observed in various studies. The chitosan addition to the low-density polyethylene showed clear inhibition halos areas for S. aureus and E. coli. 113 The chitosan or its derivatives are linked with various anionic polymers particularly different polysaccharides such as hyaluronic acid, heparin, pectin, alginate, and kappa-carrageenan for the development of multilayered films having endured antimicrobial properties. The chitosan along with other anionic polymers self-assemblies was considered as an ultrathin nanocoating for its antibacterial and antifouling properties. It was observed that the multilayers films of chitosan in association with kappa-carrageenan have better anti-adhesive potential against enterococcal cells as compared to the chitosan layer alone.114,115 Moreover, the polyurethane surface coated by chitosan and a lentinan-based polyelectrolyte considerably inhibited the growth of P. aeruginosa. The ultrathin protective coating having antibacterial and antifouling properties for glass and silicone prepared from the chitosan derivative using layer by layer deposition method were resistant to the adsorption of human plasma proteins and other plasma components as well as biofilm formation by S. aureus.116,117
Polyethyleneimine (PEI); synthetic cationic polymers
Polyethyleneimine (PEI) is a synthetic cationic polymer that is most widely used because of its low price and commercial availability from several sources. The interest in PEI is mainly related to its high positive charge density that turns it into an effective antibacterial and antifungal agent. 118 Conversely, the clinical applications of PEI are limited due to its severe cytotoxicity, poor biocompatibility, and low compatibility with blood. 119 However, easy degradable low molecular weight linear PEI derivatives having disulfide bonds, PEI’s with imine linkers, and folate-PEG-derived PEI were produced to decrease toxicity and improve biodegradability. The PEI derivatives were compared to their unmodified counterparts and were found to possess more antibacterial activity. 120
The immobilized hydrophobic PEI-based biocidal coatings have been developed especially for the surface of glass slides. It was found that these PEI-based coatings were 100% lethal against E. coli and S. aureus strains. The polyethylene or glass slides that were dipped into the N,N-dodecyl, methyl-PEI were shown to disrupt the cell membranes of E. coli and S. aureus, probably through the leakage of total cellular proteins.121,122 Additionally, the effect on the outcome of this application by the mode of application and concentration of the hydrophobic PEI and its toxicity on the mammalian cells was investigated after 1 year. These N,N-dodecyl, methyl-PEI were also used to produce stainless steel and titanium surfaces that were resistant to biofilm formation by the S. aureus strains and were evaluated in vitro and in vivo experiments using orthopedic fracture material. 123 Acinetobacter baumannii; an emergent nosocomial pathogen was also used to determine the antimicrobial action of this cationic N,N-dodecyl, methyl-PEI, and the surface coated with hydrophobic PEIs were found to be bactericidal especially after 30 min following a bacterial spray onto the dry surfaces but regarded as less bactericidal when bacterial strains were used in form of single drops.124–126
N-hexyl, methyl-PEI was also evaluated to find its potential to thwart the bacterial colonization on the surface of the medical device. The S. aureus and E. coli strains were used strains to assess the bactericidal action of N-hexyl, methyl-PEI coating on the amino-glass slide. Approximately, a 109-fold decrease in the live bacterial count was observed and 100% surface-adhered bacteria were with no toxic activity on monkey kidney cells. 127 The PEI chains alkylated with the bromohexane were coated onto the polyurethane ureteral stents as a permanent antimicrobial coating. These PEI were designed as a brush-like structure and exhibited strong activity against E. coli, Klebsiella pneumoniae, and P. mirabilis species with no cytotoxicity toward the mammalian cells. 128 The nanoparticles of Quaternary ammonium-PEI were combined in the wound dressings to apply following maxillectomy in head and neck cancer patients. These dressings resulted in excellent bactericidal activity for a wide range of bacterial pathogens without conceding the biocompatibility properties. 129
The cationic silicones were developed and tested for their antimicrobial spectrum. The in vitro and in vivo activity of silicone rubber coated with a disinfectant that is, ammonium chloride was evaluated against S. aureus, S. epidermidis, E. coli, and P. aeruginosa. The viability of the adherent staphylococci was reduced from 90% to 0% whereas the Gram-negative bacteria were reduced from 90% to 25%. 135
Coatings based on metal-oxide nanoparticles
The photoactive coatings are based on materials that are capable to produce reactive oxygen species (ROS) in the presence of visible or ultraviolet radiations. The generation of this ROS comprises electrons that have undergone promotion to their excited conduction band. These electrons can then react leading to the production of superoxide anions hydroxyl radicals which can damage the organic biomolecules, including proteins, carbohydrates, lipids, and the DNA/RNA resulting in bacterial cell death. The various metal-oxide nanoparticle, such as titanium dioxide, copper, and zinc oxides have been studied for their potential to inhibit the microbes regarding their photogenerated induced oxidative stress. The nanoparticles are better in the generation of ROS compared to their bulk counterparts possibly because of the increased surface area which provides a better interface for UV irradiation. 136
Titanium dioxide (TiO2) is the commonest photoactive material used for biomedical applications and has been used to coat various substrates including stainless steel, glass, and different medical devices.137–139 The TiO2 has widely being used mainly for dental and orthopedic implants, owing to its reported capacity to improve the adhesion and proliferation of osteoblasts in vitro as well in vivo animal models. 140 Several chemical and electrochemical approaches have been used for coating TiO2 on the medical implants and the sol-gel technique as found as the most useful. The TiO2 coating over the orthodontic wires using the sol-gel method has been shown to improve the anti-adhesive capacity of the wires against Streptococcus mutans in addition to the bactericidal activity against Porphyromonas gingivalis and S. mutans. 141 Likewise, the coating of external fixation pins being used in the surgical procedures with TiO2 increased their mechanical strength as well as an effective antibacterial activity against Staphylococcal species. 142 The sol-gel approaches also allow easy assimilation of metal ions for example copper or silver molecules having intrinsic antimicrobial characteristics. The copper-containing sol-gel-based TiO2 coatings over titanium plates were used for the prevention of implants associated infections following surgical procedures. These coatings were found effective in decreasing the number of bacteria that is, S. epidermidis strain having biofilm-forming potential. Moreover, the bacterial growth was also reduced in the neighboring media because of the release of Cu ions as confirmed in vitro and animal models.143,144 The Cu-TiO2 nanotube coatings were developed with varying copper contents in a study. The 1% copper-TiO2 nanotubes considerably decreased a load of viable adherent bacteria as compared to the untreated titanium surfaces and prevented the bacterial adhesion for additional 48 h, however, were found more effective against E. coli compared to S. aureus. 145
The silver (Ag) ions or nanoparticles were also used in combination with TiO2 to increase the antimicrobial activity. The multilayered heparin/chitosan coating comprising of TiO2, and Ag-NPs was used to deposit onto the polyethylene terephthalate. The antimicrobial activity was tested against E. coli and the assays were performed in the dark and under UV light (low intensity). The TiO2-Ag multilayer coating exhibited short-term antimicrobial activity in the dark as well in the low-intensity UV light, though the long-term properties were found in the UV light only. 146 In a more recent study, the Ag-TiO2 film coatings onto the titanium surfaces exhibited the bacteriostatic effect against S. aureus and E. coli and found that the antimicrobial activity against these microbes was enhanced due to the release of Ag ions from the coating. 147 The TiO2 was also coated onto the silicone catheters which showed excellent bactericidal activity against E. coli and onto the PLGA wound dressings obtaining an interesting antibacterial effect against both S. aureus and E. coli with sufficient survival rate of fibroblasts and keratinocytes. 148 The overall antibacterial mechanisms of nanoparticles are shown in Figure 4.

Biological effects of nanoparticles on the bacterial cell.
Antifouling coatings
Antifouling coatings are widely being introduced for their repellent properties toward microbes or to affect the architecture of biofilms. The repellent coatings mainly involve the use of hydrophilic polymers, PEG, zwitterionic materials, or super-hydrophobic material with low surface energy materials having a nanoscale surface topology. The coatings affecting the architecture of biofilms are mainly based on enzymes immobilized on the surface which either interfere with the bacterial QS or can degrade the biofilm matrix.149–151
The antifouling properties of PEG are frequently studied which are mainly linked with its hydration as well as steric hindrance effect, thus PEG is the most widely used polymer for biomaterial surfaces. 152 The length of polymer chains and the surface packing density mainly affect the antifouling properties of PEG as the short PEG chains ⩽1000 Da applied onto the polyurethane surfaces were unable to reduce the adhesion of S. epidermidis. In contrast, the longer PEG chains ⩾3350 Da was found to minimize the bacterial adhesion. 43
The poly-zwitterionic coatings have got considerable attention during the previous few years as an alternative to the PEG-based antifouling materials having higher in vivo stability. The poly-zwitterionic coatings have been widely investigated for their resistance ability to bacterial adhesion. 151 The antifouling properties of poly-zwitterion coatings over the polymer surface are also related to the formation of a hydration layer acting as a physical barrier to avoid the protein and bacterial adhesion like that of the PEG. 150 Though, the binding of water molecules is stronger due to the ionic interactions in the case of poly-zwitterionic materials. The surface packing is much important in deciding the strength of surface hydration and self-assembled monolayers were found as the most efficient. The phosphorylcholine-based self-assembled monolayers were found to reduce the adhesion of E. coli by 90% in an in vitro model. 153 Various studies have reported the activity of zwitterionic coated surfaces for the prevention of bacterial adhesion and inhibition of biofilm formation of different bacterial pathogens. 35
Antibacterial enzyme coatings
The antibacterial activity of enzymes has recently been evaluated in addition to the inactivation of degradation of biofilm. Certain enzymes may possess antimicrobial activity through hydrogen peroxide production which is toxic to the invading microbes. The hydrogen peroxide mainly destroys the bacteria through disruption of protein synthesis and bacterial cell membranes as well as oxidation of thiol groups and oxygen scavengers. 154 The glucose oxidases and cellobiose dehydrogenase (CDHs) are some of the examples of hydrogen peroxide producing enzymes that can significantly lessen the growth of bacteria, therefore are widely implicated in the food industry.154,155 The in situ approach based on the antibacterial activity of CDH based on hydrogen peroxide production by Myriococcum thermophilum using cellobiose or other exopolysaccharides from the biofilms was assessed. The inhibition of bacterial growth and biofilm formation was observed on silicone catheters for the common urinary pathogens namely the E. coli, S. epidermidis, S. aureus, P. aeruginosa, Stenotrophomonas maltophilia, P. mirabilis, and Acinetobacter baumannii in presence of cellobiose. The combination of CDH with glycoside hydrolases was found to enhance the antibacterial and antibiofilm activity further by releasing a large amount of hydrogen peroxide which resulted in a substantial decrease in biofilm formation on the surface of urinary catheters. Additionally, the urinary catheters functionalized with CDH resisted the biofilm formation by S. aureus under static as well as dynamic conditions.154,156 The Myeloperoxidases, haloperoxidases, and lactoperoxidases are other groups of antibacterial enzymes that use hydrogen peroxide to oxidize halide/pseudohalide. The glucose oxidase and horseradish peroxidase incorporated in electrospun fibers of polyurethane inhibited the growth of S. aureus and E. coli. However, these enzymes may contribute toward the undesirable degradation of biochemical compounds in the adjacent areas of implants. 157
In addition to the hydrogen peroxide producing enzymes, few other types of enzymes have been reported that can kill bacteria through different mechanisms. An antibacterial enzyme such as Lysostaphin which acts on the Staphylococcal cell wall has been immobilized over a wide range of surfaces and was reported to kill S. aureus clinical strains. 158 The nanotube conjugates comprised of Lysostaphin and carbon were developed which were found to decrease the protein adsorption and adhesion of bacteria to the surfaces. 159 Another antibacterial enzyme that is, lysozyme which degrades the cell wall of bacterial pathogens were incorporated into the coatings for the inhibition of planktonic and immobilized growth of bacteria. The constructs made of negatively charged gold nanoparticles combined with lysozyme inhibited bacterial growth.160,161 Though the antibacterial strategies based on enzymes are quite promising, the enzyme activity following immobilization on the surfaces as well as stability and efficacy inside the host are the foremost apprehensions for their applications in clinical settings. 162
Antimicrobial peptides
Antimicrobial peptides (AMPs); the oligopeptides which possess a broad-spectrum antimicrobial activity especially against bacterial, fungal, and parasitic pathogens have got significant interest during the past few years. 163 The AMPs interact with the bacterial cell membrane and disrupts the membrane or causes pore formation that ultimately leads to cell death. The AMPs disrupt the bacterial membranes in a nonspecific manner, therefore, the likelihood of acquisition of AMPs resistance is significantly low among the bacterial species. A wide range of antibacterial surfaces and materials using AMPs have been developed.154,164 For example, the polyurethane catheters with AMP-brush coating prevented bacterial adhesion and inhibited bacterial growth in a mouse model. 173 The hydrogels having a strong adherence capacity to a different surface or medical devices were developed using an AMP that is, Cateslytin which displayed a strong antibacterial activity and was found as biocompatible. 165
A broad-spectrum AMP that is, Tet213 was coated onto the titanium surface that limited the biofilm formation of S. aureus and Porphyromonas gingivalis and exhibited constant antimicrobial activity. 166 A firm antimicrobial coating was developed using cecropin-melittin functionalized with gold nanoparticles which showed bactericidal activity against various multidrug-resistant bacteria. 167 Numerous studies have reported the application of antimicrobial peptides for the coating of medical implants as shown in Table 2. However, the major problems associated with the AMPs are their uncontrolled toxicity, higher cost of manufacturing at a large scale, their cytotoxic effects on the eukaryotic cells, and degradation by the host proteases have limited their practical applications. 168
Application of antimicrobial peptides in the development of antimicrobial coated medical implants.

Conclusion and future prospects
Implant-associated infections are a stern medical problem. The implanted material provides a base for bacterial pathogens, which have an assemblage of mechanisms to be effective in this environment through biofilm construction, immune evasion, the formation of persister cells, and antimicrobial resistance. The prevention of implant-associated infections is important and the data of the related risk factors that favor the onset of such infections could help to avoid such microorganisms during preoperative or postoperative periods. The antimicrobial releasing coatings are probably the most extensively studied to prevent the medical implants associated with infections. Many of these have been established in animal models as well as clinical trials and in many cases have been able to provide a shield against infection development. Regardless of the positive results in some studies, these coatings are not applicable in clinical settings due to the rising concerns regarding their long-term effectiveness and the emergence of multidrug-resistant microorganisms. Consequently, alternative approaches to overcome or avoid these impediments have been considered, few of those seem to deserve better attention in the forthcoming years. Predominantly, the application of implants with either super-hydrophobic coatings or inherently microbicidal polymers might achieve long-term safety free from the menaces of drug resistance. Although their efficacy as an anti-biofilm material has been experimentally established, these innovative anti-biofilm coatings have not been reproduced in the clinical trials, probably due to the absence of standardized procedures for analyzing the reproducibility of results obtained from in vitro studies. Therefore, the anti-biofilm activity of the diverse and novel coatings established recently during in vitro studies along with the translational research needs to be conducted further to validate the most promising results for their possible applications in clinical settings. The pursuit for advanced anti-infective biomaterials may encourage well-founded anticipations to overcome the infective complications which are associated with surgical procedures or the long-term use of medical implants.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
