Abstract
Purpose
The aim of the present in vitro study was to evaluate the leakage observed for 2 different microbial species at the level of the implant–abutment (I-A) interface, and the marginal fit and size of microgap at the I-A interface in 2 different implant connections.
Methods
Ten specimens of each group were tested. The inner parts of 5 implants per group were inoculated with 0.1 μL of a viable Enterococcus faecalis suspension and 5 implants per group with Aggregatibacter actinomycetemcomitans. All of the vials containing the control specimens were incubated at 37°C under aerobic condition for E. faecalis and 37°C in presence of 5% CO2 for A. actinomycetemcomitans. They were maintained for 14 days, and the possible penetration of bacterial suspension into the surrounding solution was determined by the observation of turbidity of the broth. The I-A interface was evaluated for size of microgap and measured under SEM. Five implants of each group were evaluated. The marginal fit between implant and abutment was measured at 8 random locations in each assembly, under different magnifications at the interface.
Results
No leakages through the I-A interface were demonstrated for either type of connection evaluated. The microgap values of all I-A interfaces ranged from 0.008 to 2.009 μm; the differences between the 2 systems were statistically significant.
Conclusions
The present study demonstrated that a good marginal fit of implant components seemed to be able to prevent bacterial leakage.
Introduction
The levels of success obtained by the different methods of prosthetic rehabilitation using osseointegrated implants have reached elevated heights (1), but despite this, the 2-stage implant systems have still serious drawbacks. Bacteria can migrate into and out of the internal cavity of the implants. The implant–abutment (I-A) interface in the implant system with screwed abutment showed presence of microorganisms (2-3-4). The gaps and cavities of the I-A interface can act as a bacteriological reservoir and lead to inflammatory reactions of the peri-implant soft tissues (5). The positioning of the I-A interface at the alveolar bone level has been found to be associated with significant inflammatory infiltrate and bone loss, when compared with complete absence of an interface (6). These observations suggest that an inflammatory stimulus arose at the I-A interface, and there was a causal relationship between the degree of peri-implant inflammation and the extent of alveolar bone loss. Various authors (6, 7) have suggested that this increase in the content of inflammatory cells may be due to the adhesion and the proliferation of bacteria at the level of the I-A interface. The position of the I-A interface can have clinically important implications, due to the fact that aesthetic demands favor the placement of implants in a more apical position (8). This fact could promote inflammation and bone loss, perhaps gingival recession, and therefore aesthetic failure. Implants placed at the level of the bone crest or at a subcrestal level, showed a higher bone loss when compared with implants placed in a supracrestal position (9-10-11). Moreover, differences in the responses may exist for implants whose abutments have not been manipulated during the healing phase (9). The loss of stability of the connection screw is a complication that can damage the interfaces of the implant components, facilitating the contamination of its internal parts by microorganisms (12, 13).
Even though complete prevention of microbial penetration into the internal part of the implants has not been demonstrated in vitro, conical Morse taper connections have shown the most favorable results (14-15-16-17-18-19-20). The inclusion of polymer washers in the implant systems has only decreased, but not eliminated, the bacterial contamination (21). Hermeticity was demonstrated by cemented-retained I-A assemblies (18). From a mechanical standpoint, discrepancies and microgaps between components were unavoidable when different parts were fitted together. Nonetheless, there have been concerted efforts to achieve a tight I-A connection. The purpose of this in vitro study was to evaluate the migration, over a period of 14 days, of 2 different microbial species from the inside to the outside of the I-A assembly, and the marginal fit and size of microgap at the I-A interface, in 2 different implant connections.
Materials and Methods
A total of 20 implants (Sweden & Martina S.p.A., Padova, Italy) were used in this in vitro study. Both implants presented a conical Morse taper interface: Group 1 – Kohno Straight implants (4.25x13 mm) with their respective prosthetic components (straight, preformed posts, complete with the screws required for fastening them onto implants); Group 2 – Kohno TG DES implants (4.25x15 mm) and the prosthetic components (directly screwed posts). The implants had a conical internal connection, anchored by means of an internal hexagon of ample dimensions at the base of the cone.
All implants and prosthetic components were standard manufactured sterile samples. All other materials utilized in the experiment were sterilized inside surgical bags with the use of an autoclave. Images of I-A interfaces were obtained from 5 implants of each group under scanning electron microscopy (SEM). The purpose of these images was to allow a quantitative analysis of the width of the marginal gap between the prefabricated components. In addition, 10 specimens of each group were tested in the microbiological experiment.
Microbiological examination
After several trials, 0.1 μL was determined to be the ideal quantity of bacterial suspension for inoculation in both implant systems. Two different bacterial sizes were used.
The inner part of 5 implants, for each group, was inoculated with 0.1 μL of a viable
SEM analysis
Five implants of each group were selected. Each sample was washed in phosphate-buffered saline (PBS), dehydrated in ascending series of alcohols (50%, 75%, 95%, 100%), allowed to dry on absorbent paper for 48 hours, and then observed with a scanning electron microscope (Philips XL30CP, The Netherlands). The marginal fit between implant and abutment was measured at 8 locations (rotating in 45-degree increments) in each assembly with different magnification to the outer parts of the interfacial zone.
Statistical Analysis
The total number of implants per group exhibiting bacterial colonization of the microgap was reported. For description of SEM data, mean values and standard deviations (SD) were calculated. The differences between the groups were statistically analyzed using Mann-Whitney test. Statistically significant differences were considered to be a p value <0.05.
Results
Table I shows the number of assemblies showing bacterial contamination in the nutrient solution over the 14-day observation period. At the beginning of the study, both groups of implants and abutments analyzed were equally sterile, and therefore comparable from a statistical point of view.
BACTERIAL LEAKAGE IN IMPLANTS WITH DIFFERENT IMPLANT–ABUTMENT CONNECTIONS INOCULATED WITH
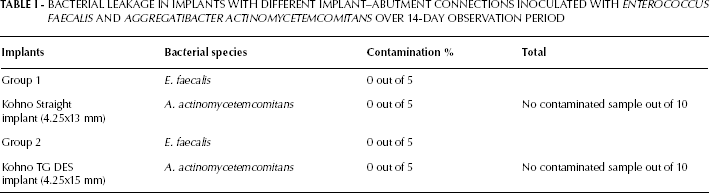
In the internal hexagon implants (Group 1), no bacterial contamination was found in I-A assemblies seeded with
Table II shows the means and standard deviations of the microgap values of I-A interface. The values ranged from 0.008 to 0.044 μm for Group 1 and from 0.008 to 2.009 μm for Group 2. Statistical analysis revealed significant differences between the 2 implant systems.
MICROGAPS OF IMPLANTS AND COMPONENTS EVALUATED UNDER SCANNING ELECTRON MICROSCOPY

Values are means ± SD.
The SEM pictures of each I-A marginal fit at different magnifications are shown in Figures 1 and 2.

Scanning electron micrographs of the marginal fit of implant–abutment (I-A) interfaces of Kohno Straight implant (Group 1).

Scanning electron micrographs of the marginal fit of implant–abutment (I-A) interfaces of Kohno TG DES implant (Group 2).
Discussion
The investigation of microgaps at the I-A interface is important because it seems reasonable that microbial colonization is either hindered or helped by the marginal fit of the I-A connection. Several authors have shown that 2-piece implants present a microgap between the implant and the abutment that has been shown to cause microbial leakage (5, 18, 19, 20, 22). These gaps may represent a bacterial reservoir that could interfere with peri-implant tissue health (4, 23). In the present study, the mean size of microgaps ranged from 0.008 to 0.044 μm for Group 1 and from 0.008 to 2.009 μm for Group 2, and the difference was statistically significant. Previous studies (5, 22, 24, 25) have shown that this gap was about 1.0 to 49.0 μm, depending on the system; Boeckler et al (26) cited clinically acceptable marginal gaps varying between 30 and 200 μm. The bacterial leakage can be altered by mechanical loading, a factor that might favor a higher movement of bacteria at the interface. Concerning the potential threat of microbial colonization, this investigation also addressed the question of whether bacteria would penetrate the I-A interfaces of the systems used in this study. No degrees of leakage through the I-A interface were demonstrated for either type of connection evaluated in the present study. Similar results have been found by other authors, who have demonstrated the presence of very low levels of bacterial contamination in Cone Morse connection implants (5, 15, 18-19-20, 27). The Cone Morse taper internal connection seems to be able to resist bacteria penetration more. Previous work from our laboratory showed that in this type of connection (18-19-20), the bacterial contamination occurred quite late during the course of the experiment (on the 22nd day), while the contamination was always earlier in the butt-joint connection implants. On the other hand, implants with Morse taper connections have been proposed as an alternative to external-hexagon implants with the goal of better stability (reduced micromovements) of the components and less bacterial leakage at the I-A interface (28). Better stability of the I-A assembly could be related to a more precise adaptation of the different components, and to the use of suitable torque forces (27, 28). The present investigation confirmed these studies.
Conversely, in an in vitro study, comparing bacterial infiltration in 2 implants with different Morse taper conical connections (Bicon and ANKYLOS), it was found that 20% of the implants showed evidence of bacterial leakage following incubation, all on the second day (27). Jansen et al (5) found after 14 days of incubation, contamination in 50% of the ANKYLOS and in 69% of the Astra implants. Location of the microgap near the alveolar crest could also be responsible for more than 1 mm of bone loss, reported in the first year of implant loading (3). The microgap has been found to have a significant influence on crestal bone levels around 2-piece implants (29). The degree of bacterial penetration in a specific implant system is probably a multifactorial condition depending on the precision of fit between the implant and abutment, the degree of micromovement between the components, the torque forces used to connect them and the loading forces when the implants are in use (3).
In the present study, different types of bacteria were used.
In conclusion, the marginal gap between implants and abutments may not have been large enough to produce leakage and consequent bacterial growth. However, the closing of implant abutment screws at the manufacturers’ recommended closing torque values could help to minimize microgaps and the potentially adverse effects of microleakage. Therefore, good marginal fit of implant components, as observed under SEM, seemed to be able to prevent bacterial leakage.
